Inorganica Chimica Acta, 169 (1990) 23-24 23
Inorganica
Ghimica
Acta
Synthesis and Crystal Structure of o-Phenylene- diaminebis(o-benzoquinonediimine)ruthenium(II) Hexafluorophosphate
HEI-YING CHENG and SHIE-MING PENG*
Department of Chemistry, National Taiwan University, Taipei 10764 (Taiwan)
(Received December 6, 1989)
In our continuing research on the 2nd- and 3rd- row transition metal complexes of o-benzoquinone- diimine [l ,2], we have synthesized a mixed-ligand
*Author to whom correspondence should be addressed.
metal complex, [Ru(opda)(bqdi)z](PF6)2, and report here its crystal structure.
o-phenylene- diamine (opda) (1) semi+-benzoquinone- diimine (s-bqdi) (2) o-benzoquinone- diimine (bqdi) (3)
Reaction of RuC13 with 1 in methanol under air for one day, followed by precipitation with PF,- from a concentrated solution, led to the isolation of the diamagnetic, red-purple compound [Ru(opda)- bqdi)z](PF.&. The pertinent spectroscopic data are: IR, u(N-H) at 342 1, 3308, 3264 and 3115 cm-‘; V(PF,) at 838 cm-’ ; UV-Vis, 544 nm (E = 6.78 X
lo3 M-’ cm-‘), 486 (4.74 X 103), 456 (5.28 X 103). Suitable single crystals were obtained by slow evaporation of a methanolic solution of the complex.
The crystal structure of [Ru(opda)(bqdi)2]2+ is shown in Fig. 1. The Ru atom is bound to the tris-
Fig. 1. ORTEP drawing of the cation [Ru(opda)(bqdi)z12+ with 50% probability. Space group, P21/c: a= 10.012(9), b= 14.135(4), c = 19.609(g) A; p= 113.91(6)“, Z = 4. A total of 1437 reflections were observed with I > 1.500, 352 parameters, togiveR=9.5.R,=7.5%.
24
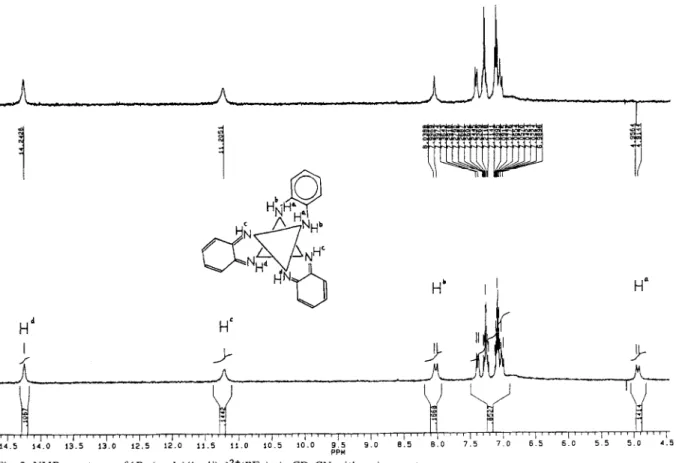
Fig. 2. NMR spectrum of [ Ru(opda)(bqdi)zlz+(PF& in CD&N with assignments.
bidentate ligands in a trigonally distorted octahedron. From the observed bond pattern, ligands A and B are assumed to be o-benzoquinonediimine (short Ru-N bonds, average 2.00 A ; short C-N bonds, 1.30 A ; short C-C bonds conjugate to C-N bonds, 1.31 A), and ligand C is assumed to be o-phenylenediamine (relatively long Ru-N bonds, 2.07 A; long C-N bonds, 1.39 A ; aromatic C-C bonds). The observed bond pattern of the o-benzoquinonediimine ligand in this complex resembles those in [Fen(CN),- (bqdi)]*- [3], [Run(bqdi)(bipy)2]2’ [4] and [Fe- (bqdi)s] ‘+ [ 11. The bond pattern of the o-phenylene- diamine ligand in this complex is comparable to those ;
Q;i(opda)41
*+ [S]
and [Com(opda)(sbqdi),]+9 *
Further diagnostic evidence is the NMR spectrum of the complex, shown in Fig. 2. Four kinds of somewhat broad signals (protons attached to nitrogen atoms) were observed at the 6 values (in ppm): 14.243 (s, 1); 11.205 (s, 1); 8.018 (d,J= 12.6 Hz, 1); 4.935 (d, J= 12.6 Hz, 1). Irradiation of the signal at one doublet resulted in the change of the other doublet to a singlet, confirming that the protons of the amine group are nonequivalent and that there is coupling between them. The assignments of the NMR signals, shown in Fig. 2, are based on the fact that the imiie NH resonances occur at lower field than those of the amine NH2.
An attempt to oxidize [Ru(opda)(bqdi)2]2+ to [Ru(bqdi)3]2c [7] by metal-assisted ligand oxidation [3,8] was unsuccessful.
Acknowledgement
This work was supported by the National Science Council, Taiwan.
References
S. Peng, C. Chen, D. Liaw, C. Chen and Y. Wang, Inorg. Chim. Acta, 101 (1985) L31.
S. Peng, K. Peters, E. Peters and A. Simon, Inorg. Chim. Actu, 101 (1985) L35.
G. G. Christoph and V. L. Goedken, J. Am. Chem. Sot., 95 (1973) 3869.
P. Belser, A. Zeiewsky and M. Zehnder, Inorg. Chem., 20 (1981) 3098.
R. C. Elder, D. Koran and H. B. Mark, Jr.. Znorg. Chem., I3 (1974) 1644.
M. Zehnder and H. LGllige, Helv. Chim. Actu, 63 (1980) 154.
L. F. Warren.Znorg. Chem., 16 (1977) 2814.
V. L. Gocdkcn, J. Chem. Sot., Chem. Commun., (1972) 207.