國
立
交
通
大
學
生化工程研究所
博 士 論 文
Haptoglobin 在生理與生化上之功能角色
Biochemical and physiologic role of haptoglobin
研 究 生:賴以祥
指導教授:毛仁淡 講座教授
Haptoglobin 在生理與生化上之功能角色
Biochemical and physiologic role of haptoglobin
研 究 生:賴以祥 Student: I-Hsiang Lai
指導教授:毛仁淡 講座教授 Advisor: Chair Prof. Simon JT Mao
國 立 交 通 大 學
生 化 工 程 研 究 所
博 士 論 文
A Thesis
Submitted to Institute of Biochemical Engineering College of Biological Science and Technology
National Chiao Tung University in partial Fulfillment of the Requirements
for the Degree of PhD in
Biochemical Engineering
June 2009
Hsinchu, Taiwan, Republic of China
Haptoglobin 在生理與生化上之功能角色
研究生:賴以祥 指導教授:毛仁淡 講座教授 國立交通大學 生化工程研究所 博士班摘要
Haptoglobin(Hp)是當動物受到感染或炎症反應時,血液中濃度會大量增加的一種 急性反應期蛋白質。其最主要功能是可與血液中游離的血紅素形成複合物,而避免溶血 時所造成的氧化壓力及組織傷害。 乳牛之乳房因細菌感染而產生炎症反應時,其牛奶中可發現大量體細胞數與Hp 濃 度上升。但是牛奶中 Hp 形成機制尚未明瞭。本研究發現牛奶中 Hp 濃度與體細胞數目 呈現正相關。進一步經由反轉錄聚合酶連鎖反應,西方轉印法、免疫化學染色法、雙重 免疫螢光染色法及Hp release 實驗進行分析,證明牛奶中 Hp 可經由嗜中性球合成與分 泌。本研究發現,當牛感染乳房炎時,牛奶中活化的嗜中性球會分泌大量 Hp,而利用 Hp 本身具有之抗氧化能力,可避免自由基形成所造成的細胞組織傷害。關於 Hp 之抗氧 化能力,本研究進一步利用Cu2+所誘發的氧化試驗發現各個重組Hp 次單位皆具有抗氧 化能力,可保護低密度脂蛋白不被氧化,並且發現β次單位之抗氧化能力明顯高於 native Hp(3.5 X)、α次單位(10X)與臨床上使用藥物 Probucol(15 X)。此研究結果可提供 未來設計mini-Hp 作為強抗氧化藥物之參考,並且可進一步了解 Hp 其抗氧化能力之機 制。在反芻動物 Hp 研究方面,本研究利用基因選殖分析發現,鹿 Hp 蛋白質之胺基酸 序列與人類 Hp 2 類似,在α次單位內皆具有一段重複序列,並且-SH group 在胺基酸序列上的位置與人類 Hp 2 相同,經蛋白質電泳分析顯示鹿 Hp 蛋白質為 tetramer (α-β)4之 結構。為了進一步了解鹿Hp tetramer 結構,利用已知可辨認人類與鹿 Hp α次單位之單 株抗體(W1)進行分析,結果發現此單株抗體僅能辨認人類 Hp 2 polymers,但卻無法 辨認鹿之Hp tetramer,顯示鹿 Hp tetramer 結構形成後此單株抗體位於α次單位上之抗原 決定位(epitope)無法完全暴露在表面,而導致此抗體無法辨認鹿之 Hp tetramer,研究 結果發現α次單位上胺基酸序列的差異所形成之空間障礙(steric hindrance)在人類與鹿 形成Hp polymer 上扮演重要的關鍵。此外,將鹿與其他哺乳類動物之 Hp 進行親緣關係 與免疫化學之分析,發現鹿 Hp 2 基因可能在 2 千 5 百萬年前就存在。 綜合以上所述,本研究發現牛奶中嗜中性球可分泌大量 Hp,其抗氧化能力可保護 細胞而避免遭受到氧化性傷害。而Hp 之抗氧化功能區域主要位於β次單位上。Hp 胺基 酸序列經由親緣關係分析,發現鹿 Hp 2 基因可能在 2 千 5 百萬年前就存在,其 Hp tetramer 之演化可能在生理方面佔有一定的優勢。
Biochemical and physiologic role of haptoglobin
Student: I-Hsiang Lai Advisor: Chair Prof. Simon JT Mao
Institute of Biochemical Engineering
National Chiao Tung University
Abstract
Haptoglobin (Hp) is an acute phase protein responsive to inflammation and infection. One
of the major functions of Hp is to capture released hemoglobin during excessive hemolysis
and to scavenge the hemoglobin-induced free radicals during oxidative stress.
Hp and somatic cell counts (SCC) are sharply elevated in bovine milk following
intramammary administration of endotoxin or bacteria. However, the sources of milk Hp
responsible for such increase are not fully understood. In this study, our results reveal that
milk Hp concentrations were correlated with SCC. Reverse transcriptase-polymerase chain
reaction, Western blot, immunocytochemistry, double immunofluorescence, and Hp releasing
experiment demonstrate that neutrophils were associated with the biosynthesis and release of
Hp in milk. We propose neutrophils may play an essential role in elevating milk Hp in
addition to previous suggestions that Hp may be derived from mammary tissues and
circulation. During bovine mastitis, activated neutrophils produce significant amount of
reactive oxygen species which may cause tissue damage. Hp is an extremely potent
attenuate such intracellular damage.
With respect to the antioxidant activity of Hp, we demonstrated that the antioxidant
activity was found to be associated with both Hp α and β chains when assessed by Cu2+-induced oxidation of low density lipoprotein. Interestingly, the antioxidant activity of
β chain was extremely potent and markedly greater than that of native Hp (3.5 x), α chain (10 x) and probucol (15 x). The present study provides a potential utility for the future design of
“mini-Hp” in developing a novel potent antioxidant. It may also provide a new insight in
understanding the mechanism involved in the antioxidant nature of Hp.
Using ruminant Hp, we found that deer Hp mimics human Hp 2 containing a tandem
repeat over the α-chain based on our cloned cDNA sequence. Interestingly, the isolated deer Hp is homogeneous and tetrameric as a (α-β)4, although the location of –SH groups
(responsible for the formation of polymers) is exactly identical to that of human. Most
interestingly, an α-chain monoclonal antibody (W1) known to recognize both dissociated human and deer α-chains, only binds to the intact human Hp polymers, but not to deer Hp tetramers. It implicates that the epitope of deer α-chain is no longer exposed on the surface when forming tetramers. We propose that steric hindrance plays a major role in determining
the polymeric formation in human and deer polymers. Phylogenetic and immunochemical
analyses revealed that the Hp 2 allele of deer might have arisen at least 25 million years ago.
is raised that the evolved tetrameric structure of deer Hp might confer a physiological
advantage.
Collectively, this thesis presents evidence that neutrophils were associated with the
biosynthesis and release of Hp in milk. The major antioxidant domain of Hp was located in
the β chain. Phylogenetic analysis revealed that the Hp 2 allele of deer might have arisen at least 25 million years ago. The evolved tetrameric structure of deer Hp might be of a
致 謝
五年的博士研究生涯,轉眼間就要告一段落了,回想修業期間藉由許
多人的幫忙才得以完成論文。特別感謝指導教授 毛仁淡講座教授,不論在
學業或生活上都給予悉心指導與愛護,並且提供一個良好的研究環境,讓
我能無慮的完成學問。感謝中興大學毛嘉洪院長、台灣大學陳明汝教授及
交通大學林志生教授,對於論文的賜教及指正,使學生論文更完善。感謝
屏東科技大學廖明輝教授、李嘉偉教授及屏東縣家畜疾病防治所魯懿萍小
姐,對我在實驗過程中無私的教導與協助,不僅提供許多實驗材料,而且
毫無保留提供我寶貴的技術與經驗,讓我的研究得以順利完成,在此致上
最誠摯的謝意。
感謝嘉義永榮牧場曹榮旭先生全家人的支持,提供我許多寶貴的材料
並且在實驗過程中全力的配合與協助,在此表達我由衷的感激。除此以外,
特別感謝已畢業的博士班學長鄭財木博士、曾繼鋒博士及陳文亮博士,在
研究過程中提供我許多寶貴意見與協助,還有感謝實驗室的夥伴們:明誌、
宏輝、俊瑩、佩真、韶禪、文昭、立品、冠佑、中曦、乃齊、詩璇、筑婷、
威延、怡安、佩宇、佶穎、巾涵、俐穎、柏如、宛伶、沁紜、伊玟等學弟
妹及靜怡、善琦小秘書,大家給予我的鼓勵與幫忙,陪我共同經歷了實驗
過程中的起起伏伏,不僅讓我學習到很多,也充實了我的生活,在此一併
致上我由衷的謝意。
最後感謝我最親愛的家人,父親賴茂雄先生、母親張碧雲女士、哥哥
賴以恆,大嫂方宜綸,姪兒致成與姪女姿穎,還有非常關心我的大伯父賴
幹雄先生、四叔賴甘霖先生,四嬸陳慧楨女士、堂弟宇凡、宇建與毅桓,
因為有你們的支持與鼓勵,讓我能無後顧之憂的順利完成博士學位。
Dissertation index
Abstract in Chinese---I Abstract in English---III Acknowledgements---VI Index---VIII Chapter 1: Overview---1Work objectives of present investigation---1
References---7
Chapter 2: Neutrophils as one of the major haptoglobin sources in mastitis affected milk (Paper 1) ---13
Abstract---13
Introduction---14
Materials and Methods---16
Results---21
Discussion---24
Acknowledgments ---28
References---28
Figures---34
Chapter 3: Cloning and expression of human haptoglobin subunits in Escherichia coli: delineation of a major antioxidant domain (Paper 2) ---41
Abstract---41
Materials and Methods---44
Results and Discussion---50
Acknowledgments---56
References---56
Tables---62
Figures---64
Chapter 4: A unique tetrameric structure of deer plasma haptoglobin: an evolutionary advantage in the Hp 2-2 phenotype with homogeneous structure (Paper 3) ---69
Abstract---69
Introduction---70
Materials and Methods---73
Results---78 Discussion---84 Acknowledgments---91 References---91 Figures---98 Chapter 5: Discussion---108 Appendix---109 Curriculum vitae---110 Publications---116
Chapter 1
Overview
Haptoglobin (Hp) is an acute phase protein, responsive to infection and inflammation,
ubiquitously present in the plasma of all mammals [1-4]. A recent study has found that Hp
also exists in lower vertebrates, bony fish but not in frog and chicken [5]. The most
frequently reported biological functions of the protein are to capture released hemoglobin (Hb)
during excessive hemolysis and to transport Hb to the liver [6]. The captured hemoglobin is
internalized by a macrophage/monocyte receptor, CD163, via endocytosis. Interestingly, the
CD163 receptor only recognizes Hp and hemoglobin in complex, which indicates the
exposure of a receptor-binding neoepitope [6]. Thus, CD163 is identified as a hemoglobin
scavenger receptor. Besides the recycling of Hb-iron, the formation of the Hp-Hb complex
possesses two additional benefits. First, Hp has a bacteriostatic effect by hampering the iron
requiring process of bacterial replication as shown in rats inoculated with pathogenic
Escherichia coli (E. coli) [7]. Second, Hp was assigned an antioxidant role by inhibiting
Hb-driven free radical oxidative tissue damage [8-9]. Recently, we have shown that Hp is an
extremely potent antioxidant, which directly protects low-density-lipoprotein (LDL) from
Cu2+-induced oxidation. The potency is markedly superior to probucol: one of the most
potent antioxidants used in antioxidant therapy [10-12]. Transfection of Hp cDNA into
Human Hp is originally synthesized as a single αβ polypeptide; following posttranslational cleavage by a protease, α and β chains are formed and then linked by disulfide bridges producing the mature Hp [13]. The gene is characterized by two common
alleles Hp 1 and Hp 2 corresponding to α1β and α2β polypeptide chains, respectively, resulting in three main phenotypes: Hp 1-1, 2-1 and 2-2. DNA encoding for these two
chains are linearly oriented in chromosome 16q22.1 [14] with 5 and 7 introns in Hp 1 and Hp
2, respectively. These introns, however, are exclusively located at the region corresponding
to the α chain [9]. The α2 contains a unique nonhomologous crossing-over in a part of the α1 [15]. Hp 2-1 and 2-2 form polymers due to an additional –SH group at the α2 chain, which determines its phenotype specificity [16]. All the phenotypes share the same β chains,
each containing 245 amino-acid residues. The α1 chain (each with 83 amino acids) is “monovalent” forming one disulfide bridge with α and β subunit resulting in Hp dimer (α1β)2.
In contrast, the α2 chain (each with 142 amino acids) is “divalent” containing one extra free –SH that is able to interact with an additional α2. As such, α2 chains can bind to either α1 or α2 chains to form large polymers [(α1β)2-(α2β)n in Hp2-1 and (α2β)n in Hp2-2].
Because of its weaker binding affinity to hemoglobin and retarded mobility (or
penetration) between the cells, the polymeric structure of Hp 2-2 is dramatically more
prevalent in some groups of patients with certain diseases, such as diabetes and
originated from Hp 1 about 2 million years ago and then gradually displaced Hp 1 as a
consequence of a nonhomologous crossing-over between the structural alleles (Hp 1) during
meiosis [20-22], and is the first example of partial gene duplication of human plasma proteins
[20,23-24]. Thus, only humans possess additional Hp 2-1 and 2-2 phenotypes.
All mammalian species studied to date have been shown to possess Hp. Analysis of the
electrophoretic patterns of Hp-Hb complexes has suggested that most of mammals Hps are
similar to human Hp 1-1 [25]. Only the Hp found in the plasma of ruminants resemble
polymeric forms of human Hp 2-2 [26]. Structurally, bovine Hp is unique possessing only
Hp 2-2 phenotype [27-28] with molecular weight ranged from 660 to 730 kDa [28]. Their
size and hemoglobin binding complex are significantly larger than that of human Hp 2-2.
The amino-acid sequence deducted from the nucleotide sequence is similar to human Hp 2
containing a tandem repeat over the α chain. Thus, the Hp 2 allele is not unique in human. Recently, we have found that there is one additional -SH group (Cys-97) in bovine α chain
with a total of 8 -SH groups, which may be responsible for the overall polymeric structure
that is markedly different from human Hp 2-2 [28].
1.1 Work objectives of present investigation
There are three objectives of this present dissertation:
definition of the source of milk Hp from dairy cows with mastitis.
2. Production of the recombinant Hp subunits in Escherichia coli and determination of a
major antioxidant domain of human Hp.
3. Definition of the molecular evolution of mammals Hp and study of the polymeric
structure of deer Hp.
1.2 Neutrophils as one of the major haptoglobin sources in mastitis affected milk (Paper 1)
Bovine Hp, while not abundantly expressed in normal bovine plasma, is considered to be one
of the sensitive acute phase proteins during bacterial infections [28-29]. Several studies
indicate that its concentration increases dramatically in both plasma and milk during clinical
mastitis of dairy cows [30-32]. In experimentally induced mastitis by intramammary
bacterial-challenge with Streptococcus uberis, the observed increase in milk Hp has been
suggested to originate from the circulation [33]. Hiss et al., 2004 also found elevated Hp in
bovine milk after intramammary endotoxin challenge. It was further observed that Hp could
be locally synthesized within the mammary gland [34]. However, the source of milk Hp
found in naturally occurring mastitis is poorly understood
The purpose of the present study was to define the source of milk Hp from dairy cows
presence of Hp protein using Western blot, Hp mRNA expression using reverse
transcriptase-polymerase chain reaction (RT-PCR), partial cell typing using double confocal
immunofluorescence, and endogenously released Hp using ELISA. Finally, we localized Hp
in normal and mastitic mammary gland tissue and in cultured mammary epithelial cell line
(MAC-T). Our findings shows that the increased Hp was associated with neutrophils, in
which Hp was released into the medium in an ex vivo experiment. In mastitis affected
mammary glands, Hp was mainly localized within the epithelial cells. We also observed for
the first time that cultured epithelial MAC-T cells could synthesize Hp in vitro. In addition
to locally synthesized Hp within the mammary gland and Hp transferred from the circulation,
we propose that milk neutrophils and mammary epithelium also contribute to milk Hp
concentrations.
1.3 Cloning and expression of human haptoglobin subunits in Escherichia coli: delineation of a major antioxidant domain (Paper 2)
Since the purification for human native Hp and its corresponding α or β chain has been difficult, the purpose of the present study was to effectively express each recombinant Hp
subunit and to localize its antioxidant domain(s). Using E. coli, we show that α1, α2, β, and α2β chain was abundantly expressed and primarily present in the inclusion bodies consisting of about 30% of the cell-lysate proteins. In human liver cells, however, the α2β is
postranscritionally cleaved into α and β chains between the residues Arg and Ile [35-36]. A more recent study suggests that a complement C1r-like protease is responsible for the
cleavage [37]. Our data indicate that this protease is not present in the E. coli. Each cloned
subunit retained its immunoreactivity as confirmed using antibodies specific to α or β chain. By circular dichroism, the structure of each expressed subunit was disordered as compared to
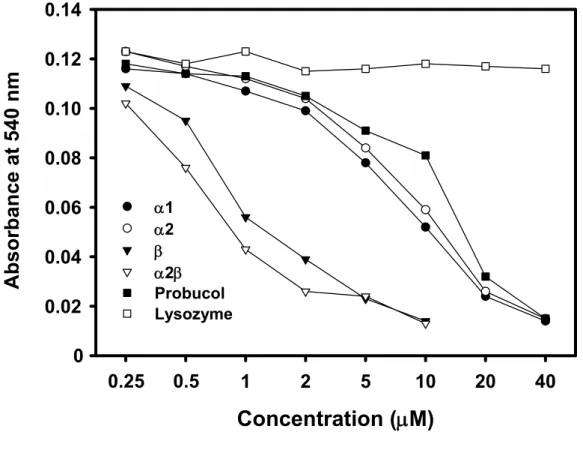
the native Hp. The antioxidant activity was found to be associated with both α and β chains when assessed by Cu2+-induced oxidation of low density lipoprotein (LDL). Of remarkable
interest, the antioxidant activity of β chain was extremely potent and markedly greater than that of native Hp (3.5 x), α chain (10 x) and probucol (15 x). The present study is a clinically proved potent compound used for antioxidant therapy. The “unrestricted”
structure of β subunit may therefore render its availability for free-radical scavenge, which provides a utility for the future design of a “mini-Hp” in antioxidant therapy. It may also
provide a new insight in understanding the mechanism or specific amino acids involved in the
antioxidant nature of Hp.
1.4 A unique tetrameric structure of deer plasma haptoglobin: an evolutionary advantage in the Hp 2-2 phenotype with homogeneous structure (Paper 3)
Similar to blood types, human Hp is classified as three phenotypes: Hp 1-1, 2-1, and 2-2.
phenotype or Hp 1 in almost all the animal species [25]. The Hp 2-2 protein consists of
complicated large polymers cross-linked by α2-β subunits or (α2-β)n (where n>3, up to 12 or
more) and is associated with the risk of the development of diabetic, cardiovascular, and
inflammatory diseases [17-19]. In the present study, we found that deer plasma Hp mimics
human Hp 2 containing a tandem repeat over the α-chain based on our cloned cDNA sequence. Remarkably interesting, the isolated deer Hp is homogeneous and tetrameric as a
(α-β)4, although the location of –SH groups (responsible for the formation of polymers) is
exactly identical to that of human. Denaturation of deer Hp using 6M urea in reducing
condition (143 mM β-mercaptoethanol) followed by renaturation still sustained the formation of (α-β)4 suggesting that the Hp tetramers are not randomly assembled. Interestingly, an
α-chain monoclonal antibody (W1) known to recognize both human and deer α-chains, only binds to the intact human Hp polymers, but not to deer Hp tetramers. It implicates that the
epitope of deer α-chain is no longer exposed on the surface when Hp tetramers are formed. We propose that steric hindrance plays a major role in determining the polymeric formation in
human and deer polymers. Phylogenetic and immunochemical analyses revealed that the Hp
2 allele of deer might have arisen at least 25 million years ago. A mechanism involved in
forming Hp tetramers is proposed and discussed, and the possibility is raised that the evolved
References
[1] González-Ramón N., Hoebe K., Alava M.A., Van Leengoed L., Piñeiro M., Carmona S.,
Iturralde M., Lampreave F., Piñeiro A., Pig MAP/ITIH4 and haptoglobin are
interleukin-6-dependent acute-phase plasma proteins in porcine primary cultured
hepatocytes, Eur. J. Biochem. (2000) 267:1878-1885.
[2] Wang Y., Kinzie E., Berger F.G, Lim S.K., Baumann H., Haptoglobin, an
inflammation-inducible plasma protein, Redox Rep. (2001) 6:379-385.
[3] Raijmakers M.T., Roes E.M., te Morsche R.H., Steegers E.A., Peters W.H., Haptoglobin and
its association with the HELLP syndrome, J. Med. Genet. (2003) 40:214-216.
[4] Gervois P., Kleemann R., Pilon A., Percevault F., Koenig W., Staels B., Kooistra T., Global
suppression of IL-6-induced acute phase response gene expression after chronic in vivo
treatment with the peroxisome proliferator-activated receptor-{alpha} activator fenofibrate, J.
Biol. Chem. (2004) 279:16154-16160.
[5] Wicher K.B., Fries E., Haptoglobin, a hemoglobin-binding plasma protein, is present in bony
fish and mammals but not in frog and chicken, Proc. Natl. Acad. Sci. U. S. A. (2006)
103:4168-4173.
[6] Kristiansen M., Graversen J.H., Jacobsen C., Sonne O., Hoffman H., Law S.K.A., Moestrup
S.K., Identification of the haemoglobin scavenger receptor, Nature (2001) 409:198-201.
(1982) 215:691-693.
[8] Miller Y.I., Altamentova S.M., Shaklai N., Oxidation of low-density-lipoprotein by
hemoglobin stems from a heme-initiated globin radical: antioxidant role of haptoglobin,
Biochemistry (1997) 36:12189-12198.
[9] Langlois M.R., Delanghe J.R., Biological and clinical significance of haptoglobin
polymorphism in humans, Clin. Chem. (1996) 42:1589-1600.
[10] Mao S.J.T., Yates M.T., Jackson R.L., Antioxidant activity and serum levels of probucol and
probucal metabolites, Methods Enzymol. (1994) 234:505-513.
[11] Tseng C.F., Lin C.C., Huang H.Y., Liu H.C., Mao S.J.T., Antioxidant role of human
haptoglobin, Proteomics (2004) 4:2221-2228.
[12] Lai I.H., Tsai T.I., Lin H.H., Lai W.Y., Mao S.J.T., Cloning and expression of human
haptoglobin subunits in Escherichia coli: Delineation of a major antioxidant domain, Protein
Expr. Purif. (2007) 52:356-362.
[13] Kurosky A., Barnett D.R., Lee T.H., Touchstone B., Hay R.E., Arnott M.S., Bowman B. H.,
Fitch W.M., Covalent structure of human haptoglobin: a serine protease homolog, Proc. Natl.
Acad. Sci. U. S. A. (1980) 77:3388-3392.
[14] McGill J.R., Yang F., Baldwin W.D., Brune J.L., Barnett D.R., Bowman B.H., Moore C.M.,
Localization of the haptoglobin alpha and beta genes (HPA and HPB) to human chromosome
[15] Bowman B.H., Kurosky A., Haptoglobin: the evolutionary product of duplication, unequal
crossing over, and point mutation, Adv. Hum. Genet. (1982) 12:189-261.
[16] Wejman J.C., Hovsepian D., Wall J.S., Hainfeld J.F., Greer J., Structure and assembly of
haptoglobin polymers by electron microscopy, J. Mol. Biol. (1984) 174:343-368.
[17] Miyoshi H., Ohshiba S., Matsumoto A., Takada K., Umegaki E., Hirata I., Haptoglobin
prevents renal dysfunction associated with intravariceal infusion of ethanolamine oleate, Am.
J. Gastroenterol. (1991) 86:1638-1641.
[18] Engstrom G., Lind P., Hedblad B., Wollmer P., Stavenow L., Janzon L., Lindgarde F., Lung
function and cardiovascular risk: relationship with inflammation-sensitive plasma proteins,
Circulation (2002) 106:2555-2560.
[19] Hochberg I., Roguin A., Nikolsky E., Chanderashekhar P.V., Cohen S., Levy A.P.,
Haptoglobin phenotype and coronary artery collaterals in diabetic patients, Atherosclerosis
(2002) 161:441-446.
[20] Maeda N., Yang F., Barnett D.R., Bowman B.H., Smithies O., Duplication within the
haptoglobin Hp2 gene, Nature (1984) 309:131-135.
[21] Maeda N., Nucleotide sequence of the haptoglobin and haptoglobin-related gene pair. The
haptoglobin-related gene contains a retrovirus-like element, J. Biol. Chem. (1985)
260:6698-6709.
primates, J. Biol. Chem. (1988) 263:15740-15747.
[23] Smithies O., Walker N.F., Genetic control of some serum proteins in normal humans, Nature
(1955) 176:1265-1266.
[24] Black J.A., Dixon G.H. Amino-acid sequence of alpha chains of human haptoglobins,
Nature (1968) 218:736-741
[25] Javid J., Human haptoglobins, Curr. Top Hematol. (1978) 1:151-192.
[26] Travis J., Sanders B., Haptoglobin evolution: polymeric forms of Hp in the bovidae and
cervidae families, J. Exp. Zool. (1972) 180: 141-148.
[27] Lai I.H., Lin K.Y., Larsson M., Yang M.C., Shiau C.H., Liao M.H., Mao S.J.T., A unique
tetrameric structure of deer plasma haptoglobin: an evolutionary advantage in the Hp 2-2
phenotype with homogeneous structure, FEBS J. (2008) 275:981-993.
[28] Lai Y.A., Lai I.H., Tseng C.F., Lee J., Mao S.J.T., Evidence of tandem repeat and extra
thiol-groups in the polymeric formation of bovine haptoglobin: a unique structure of Hp 2-2
phenotype, J. Biochem. Mol. Biol. (2007) 40:1028-1038.
[29] Petersen H.H., Nielsen J.P., Heegaard P.M., Application of acute phase protein
measurements in veterinary clinical chemistry, Vet. Res. (2004) 35:163-187.
[30] Eckersall P.D., Young F.J., Nolan A.M., Knight C.H., McComb C., Waterston M.M., Hogarth
C.J., Scott E.M., Fitzpatrick J.L., Acute phase proteins in bovine milk in an experimental
[31] Grönlund U., Hallén Sandgren C., Persson Waller K., Haptoglobin and serum amyloid A in
milk from dairy cows with chronic sub-clinical mastitis, Vet. Res. (2005) 36:191-198.
[32] Nielsen B.H., Jacobsen S., Andersen P.H., Niewold T.A., Heegaard P.M., Acute phase
protein concentrations in serum and milk from healthy cows, cows with clinical mastitis and
cows with extramammary inflammatory conditions, Vet. Rec. (2004) 154:361-365.
[33] Pedersen L.H., Aalbaek B., Røntved C.M., Ingvartsen K.L., Sorensen N.S., Heegaard P.M.,
Jensen H.E., Early pathogenesis and inflammatory response in experimental bovine mastitis
due to Streptococcus uberis, J. Comp. Pathol. (2003) 128:156-164.
[34] Hiss S., Mielenz M., Bruckmaier R.M., Sauerwein H., Haptoglobin concentrations in blood
and milk after endotoxin challenge and quantification of mammary Hp mRNA expression, J.
Dairy Sci. (2004) 87:3778-3784.
[35] Goldstein L.A., Heath E.C., Nucleotide sequence of rat haptoglobin cDNA. Characterization
of the alpha beta-subunit junction region of prohaptoglobin, J. Biol. Chem. (1984)
259:9212-9217.
[36] Hanley J.M., Heath E.C., A novel proteolytic activity in serum processes rat prohaptoglobin,
Arch. Biochem. Biophys. (1985) 239:404-419.
[37] Wicher K.B., Fries E., Prohaptoglobin is proteolytically cleaved in the endoplasmic
reticulum by the complement C1r-like protein, Proc. Natl. Acad. Sci. U. S. A. (2004)
Chapter 2
Neutrophils as one of the major haptoglobin sources in mastitis affected
milk (Paper 1)
Abstract
The antioxidant haptoglobin (Hp) is an acute-phase protein responsive to infectious and
inflammatory diseases. Hp and somatic cell counts (SCC) are sharply elevated in bovine
milk following intramammary administration of endotoxin or bacteria. However, the
sources of milk Hp responsible for such increase are not fully understood. The purpose of
this study was to define the source of milk Hp from dairy cows with naturally occurring
mastitis. Quarter milk samples selected from 50 dairy cows were separated into four groups
according to SCC as group A: <100 (n=19); B: 100-200 (n=10); C: 201-500 (n=10); and D:
>500 × 103 (n=11) cells/mL. Our results reveal that milk Hp concentrations were correlated
with SCC (r=0.742; P<0.01), and concentrations in group D were ~10 fold higher than in
group A. Reverse transcriptase-polymerase chain reaction (RT-PCR) analysis indicates that
the milk somatic cell cells from group D were not only capable of synthesizing Hp but could
also markedly increase Hp mRNA expression. Western blot, immunocytochemistry, double
confocal immunofluorescence, and Hp releasing experiment demonstrate that neutrophils
was significantly elevated in the epithelium of mammary gland tissue with mastitis and was
also expressed in the cultured mammary epithelial cells. We propose neutrophils and
epithelial cells may play an essential role in elevating milk Hp in addition to previous
suggestions that Hp may be derived from mammary tissues and circulation.
Key words: haptoglobin / neutrophil / mastitis / MAC-T cells / immunocytochemistry
2.1 Introduction
Haptoglobin (Hp) is an acute phase protein responsive to inflammation and infection [1, 2, 3].
One of the major functions of Hp is to capture released hemoglobin during excessive
hemolysis [4] and to scavenge the hemoglobin-induced free radicals during oxidative stress
[5]. We have recently shown that Hp is an extremely potent antioxidant, which directly
prevents low-density-lipoproteins (LDL) from Cu2+- and radical compound-induced oxidation
[6, 7]. Transfection of Hp cDNA into Chinese hamster ovary (CHO) cells protects them
against oxidative stress [7]. Structurally, bovine Hp is unique possessing only Hp 2-2
phenotype [8, 9] with molecular weight ranged from 660 to 730 kDa [9] which is different
from that of human expressed as Hp 1-1, 2-1, or 2-2.
Bovine Hp, while not abundantly expressed in normal bovine plasma, is considered to be
one of the sensitive acute phase proteins during bacterial infections [3, 9]. Several studies
mastitis of dairy cows [1, 10, 11]. In experimentally induced mastitis by intramammary
bacterial-challenge with Streptococcus uberis, the observed increase in milk Hp has been
suggested to originate from the circulation [12]. Hiss et al., 2004 also found elevated Hp in
bovine milk after intramammary endotoxin challenge [13]. It was further observed that Hp
could be locally synthesized within the mammary gland [13]. The source of milk Hp found
in naturally occurring mastitis is poorly understood.
The purpose of the present study was to define the source of milk Hp from dairy cows
with high somatic cell counts (SCC) (or mastitis). We investigated milk somatic cells for the
presence of Hp protein using Western blot, Hp mRNA expression using reverse
transcriptase-polymerase chain reaction (RT-PCR), partial cell typing using double confocal
immunofluorescence, and endogenously released Hp using ELISA. Finally, we localized Hp
in normal and mastitic mammary gland tissue and in cultured mammary epithelial cell line
(MAC-T). Our findings shows that the increased Hp was associated with neutrophils, in
which Hp was released into the medium in an ex vivo experiment. In mastitis affected
mammary glands, Hp was mainly localized within the epithelial cells. We also observed for
the first time that cultured epithelial MAC-T cells could synthesize Hp in vitro. In addition
to locally synthesized Hp within the mammary gland and Hp transferred from the circulation,
we propose that milk neutrophils and mammary epithelium also contribute to milk Hp
2.2 Materials and methods 2.2.1 Sample collection and SCC
Quarter milk samples from individual cows (Bos taurus) were collected at the Yong Rong
Dairy Farm (Chyayi, Taiwan). SCC in each sample was measured using a Fossomatic 4000
cell counter (Foss Electric, Hillerød, Denmark) at the Livestock Research Institute, Council of
Agriculture, Executive Yuan (Hsinchu, Taiwan). Quarter milk samples were then selected
and assigned to four groups according to SCC (A: < 100; B: 100-200; C: 201-500; and D: >
500 × 103 cells/mL with n = 19, 10, 10, and 11, respectively).
2.2.2 Purification of bovine Hp and preparation of anti-Hp antibodies
Native bovine Hp was isolated from the plasma using an anti-bovine Hp immunoaffinity
column, followed by gel-permeation chromatography (Superose-12) on a HPLC system as
previously described [9]. Mouse and rabbit polyclonal antibodies against bovine
recombinant Hp or mouse monoclonal antibody (mAb; 2H12) against bovine native Hp were
prepared in our laboratory according to previously established procedures [9, 14].
2.2.3 Determination of milk Hp levels using ELISA
(Immunology Consultants Laboratory, Newberg, OR) according to the manufacture’s
instructions. One hundred µL of skimmed milk (1:10 dilution) were used for the determination of Hp, while skimmed milk was prepared as previously described [15].
2.2.4 Isolation of milk somatic cells and protein extraction
Milk somatic cells were isolated from 200 mL freshly collected milk. Briefly, milk samples
maintained at 4 ºC were centrifuged at 1,000 g for 20 min at 4 ºC. After removing the fat
layer and supernatant, the somatic cells were briefly washed twice with PBS at 4 °C and
maintained on ice. Total cell proteins were extracted using a lysis buffer containing 0.3%
Triton X-100 in PBS and sonicated at 4 ˚C for 5 min, followed by centrifugation at 10,000 g
for 10 min at 4 ˚C. The remaining supernatant was stored at -20 ˚C until analyses by
SDS-PAGE and Western blot.
2.2.5 Gel electrophoresis and Western blot
SDS-PAGE was carried out on 1.5-mm-thick slab gel, using a discontinuous system as
previously described [8]. Unless otherwise specified, the gel containing 15% (for reducing)
or 4% (for non-reducing samples) polyacrylamide was used with a top stacking gel of 5%
polyacrylamide. Approximately 20 µg of the protein was loaded onto the gels and each tested sample was preheated at 100 ºC for 10 min in a loading buffer (12 mM Tris-HCl, pH
6.8, 0.4% SDS, 5% glycerol, 0.02% bromphenol blue) with or without 140 mM
2-mercaptoethanol. The samples were then run for about 1.5 h at 100 V and stained by
Coomassie brilliant blue. Molecular-mass standard containing 12 prestained proteins
(3.5-260 kDa) was purchased from Invitrogen (Carlsbad, CA). Western blot analysis was
performed similar to that described previously [6].
2.2.6 Analysis of Hp mRNA expression of milk somatic cells
Total RNA was extracted from milk somatic cells using RNeasy mini kit (Qiagen, Hilden,
Germany) according to the manufacture’s instructions. The first strand cDNA was
synthesized using moloney murine leukemia virus (MMLV) reverse transcriptase (Invitrogen).
Briefly, equal amount of total RNA (1 µg) was added to a reaction mixture containing 50 mM
Tris-HCl (pH 8.3), 10 mM dithiothreitol, 0.5 µg oligo (dT)18, 75 mM KCl, 3 mM MgCl2, 0.5
mM dNTP mix, 40 U of RNase inhibitor, and 200 U of MMLV reverse transcriptase, and
proceeded at 37 °C for 50 min, followed by 70 °C for 15 min. Equal amount of total cDNA
(100 ng) was amplified by PCR using Hp specific primers, while using glyceraldehyde
3-phosphate dehydrogenase (GAPDH) as a house-keeping control. The primer design was
based on the published nucleotide sequence of bovine Hp [15] with
5’-TGCTGCAGGGATCATCGGTGGCTCATTGGA-3’ and 5’-CGGAAAACCATCGCTAA
primers prepared were 5’-CCTGGAGAAACCTGCCAAGT-3’ (forward) and 5’-GCCAA
ATTCATTGTCGTACCA-3’ (reverse). The PCR cycling profile was 95 ºC for 5 min
followed by 30 cycles at 94 ºC for 30, 55 ºC for 30 s, 72 ºC for 50 s (or 30 s in GAPDH) with
a final extension at 72 ºC for 10 min. The RT-PCR products (214 bp for GAPDH and 755
bp for Hp) were resolved on 1.5% agarose gel [14], followed by an ethidium bromide staining.
The band intensity corresponding to Hp was determined using a Quantity One software of Gel
Doc 2000 Gel Documentation System (Bio-Rad Laboratories, Hercules, CA).
2.2.7 Mammary epithelial cell culture (MAC-T) and Hp mRNA expression
MAC-T, an established and immortalized epithelial cell line isolated from bovine mammary
tissue was cultured as previously described [16, 17]. In general, the cells (1 × 104 cells per
well) were grown at 37 °C (5% CO2) in a 24-well culture plate in complete Dulbecco’s
modified Eagle medium (DMEM) (Invitrogen) supplemented with 10% fetal bovine serum
(containing no immunoreactive bovine Hp), 50 µg/mL of streptomycin, and 50 IU/mL of
penicillin (Invitrogen). Hp mRNA expression were analyzed using RT-PCR similar to the
procedures described above.
2.2.8 Immunocytochemical staining of somatic and MAC-T cells and mammary tissues
for immunostaining according to the manufacturer’s instructions. In brief, cytospins
containing freshly isolated somatic or MAC-T cells were treated with ice-chilled methanol
(100%) for 15 min and then rehydrated by PBS. Cells were permeabilized in PBS
containing 0.3% Triton X-100 for 10 min, while the endogenous peroxidase was blocked by
incubating with 3% H2O2. After blocking with 2% gelatin, cells were incubated with
unlabeled mouse anti-CD5 mAb (lymphocyte marker), mouse anti-CD11b mAb (neutrophil
marker) (Serotec, Oxford, UK), or mouse anti-Hp polyclonal antibody for 1 h. The slides
were then incubated with biotinylated anti-mouse IgG for 30 min, followed by washes and
incubation with HRP-conjugated streptavidin for 30 min. After washes, the slides were
developed with 3-amino-9-ethylcarbazole (AEC) or 3,3’-diaminobenzidine (DAB) substrate
and counterstained with hematoxylin. For normal and mastitic mammary tissues (n = 5
each), each section was deparaffinized in a 50 ºC oven for 10 min and then placed for two
5-min washes in xylene at 24 ºC. The sections were rehydrated through sequential soakings
in 100, 95, 90, 80, and 50% alcohol for 2 min each, followed by final washes in deionized
water. The sections were then stained using procedures described above.
2.2.9 Double immunofluorescence using confocal laser scanning microscopy
Cytospin prepared cells used for localization of CD11b and Hp were permeabilized as
mAb and rabbit anti-Hp polyclonal antibody for 1 h. The slides were then incubated with a
mixture of fluorescent isothiocyanate (FITC) conjugated anti-mouse IgG and rhodamine
conjugated anti-rabbit IgG (Jackson Immunoresearch Laboratories, West Grove, PA) for 1 h
in dark. Cells were washed with PBS before examination using a Fluoview FV500 confocal
laser scanning microscopy (Olympus, Tokyo, Japan).
2.2.10 Time course of Hp protein released from somatic cells in vitro
Two ml of isolated somatic cells (adjusted to 1 × 104 cells/mL at 4 ºC) from Groups A and D
(n = 3 for each) were suspended in Roswell Park Memorial Institute medium (RPMI) 1640
medium (JRH Biosciences, Lenexa, KS) containing 100 µg/mL ampicillin with or without
protease inhibitor of 1 mM phenylmethylsulfonyl fluoride (PMSF) (Sigma, St.Louis, MO) in
a test tube and were incubated at 37 ºC for 0, 2, 4, 8 and 16 h, respectively, followed by
centrifugation at 1,000 g at 4 ˚C for 5 min. The supernatant was then subjected to ELISA for
the determination of Hp concentrations.
2.3 Results
2.3.1 Correlation between Hp levels and SCC in bovine milk
Selected bovine milk samples (n=50) were assigned into four groups according to their SCC
respectively). The mean ± SEM of milk Hp concentrations determined by an ELISA in groups A, B, C, and D were 0.23 ± 0.08, 0.74 ± 0.23, 1.11 ± 0.28, and 2.22 ± 0.53µg/mL, respectively. The mean Hp concentration of group D was ~10-fold higher than that of group
A (P < 0.001). The overall Hp concentrations were correlated with SCC (r = 0.742; P <
0.01).
2.3.2 Hp in milk somatic cells
Because Hp levels in the milk samples were positively correlated with SCC, we hypothesized
that somatic cells might contribute to the presence of Hp in milk. We extracted protein from
the somatic cells from the low (group A) and high SCC (group D) to examine the presence of
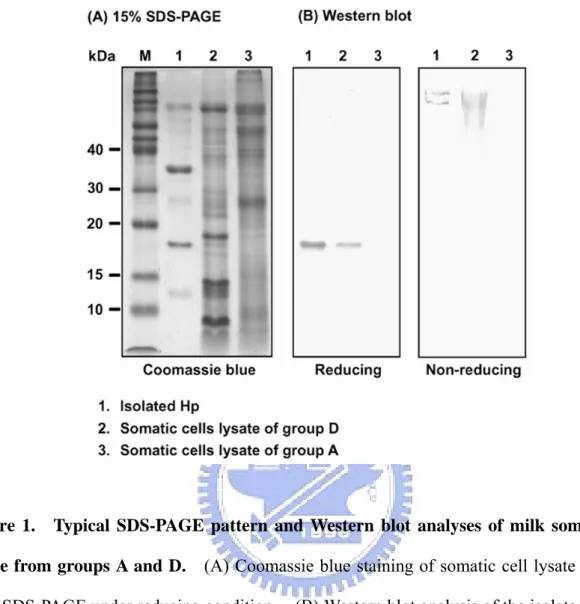
Hp using a Western blot. Fig. 1 depicts the presence of Hp in group D, but not in group A.
It implicates the endogenous synthesis and expression of Hp between the analyzed cells of
groups A and D were different or/and altered. The electrophoretic pattern of bovine Hp
(non-reduced) is similar to that previously reported by our laboratory [9].
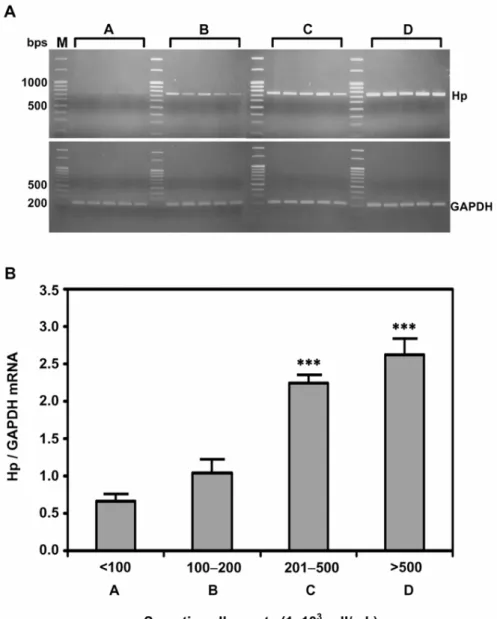
2.3.3 Endogenous expression of Hp mRNA in milk somatic cells
To determine whether somatic cells could endogenously synthesize Hp, RT-PCR for the
expression of Hp mRNA was conducted, while using GAPDH as a house-keeping control.
0.001). The mean Hp mRNA expression in groups C and D were evidently higher than
those in groups A and B (P < 0.001).
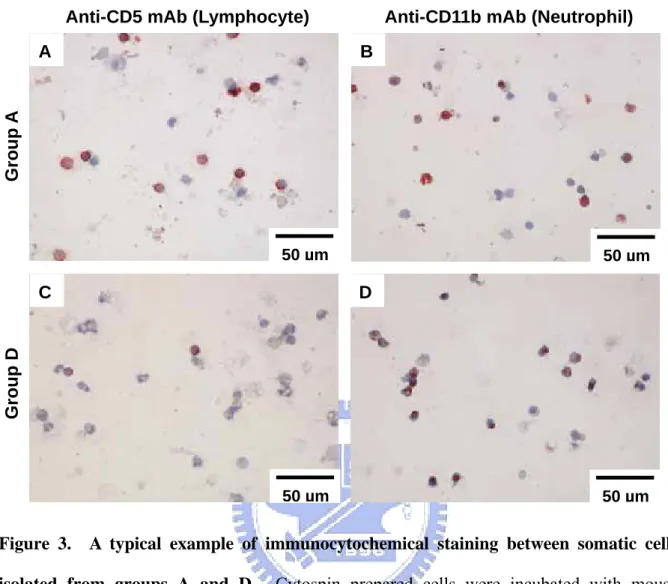
2.3.4 Somatic cell typing
To identify the cell types in group D, we used anti-CD5 (lymphocyte marker) and anti-CD11b
(neutrophil marker) mAb as a probe in immunostaining, while comparing group A as a
non-mastitis control. We found lymphocytes to be a major cell type in group A and
neutrophils the predominant cell type in group D (Fig. 3). The immunostaining was specific
because the background control using non-immuned sera or non-related mAb was negative.
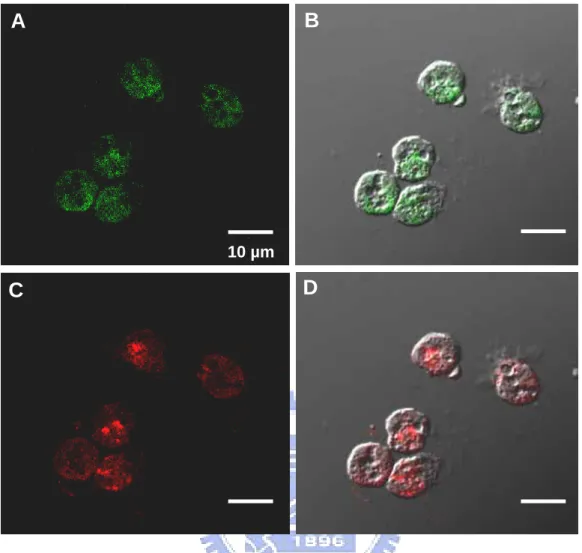
2.3.5 Colocalization of Hp and CD11b in neutrophils using confocal microscopy
In addition to RT-PCR, we tested whether neutrophils could express Hp in situ. Somatic cells
isolated from group D enriched with neutrophils were examined for localization of Hp using a
double confocal laser scanning microscopy. Fig. 4 shows that all the neutrophils identified
by CD11b (in green FITC) exhibited expression of Hp (in red rhodamine), while no staining
was observed for cells treated with non-immuned sera or unrelated mAb.
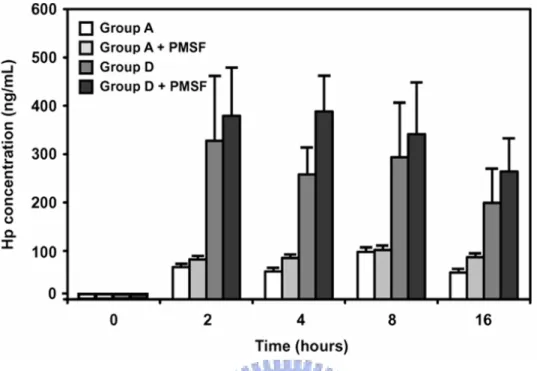
2.3.6 Release of Hp from the somatic cell ex vivo
somatic cells from the groups A and D (n=3 quarters for each) and investigated their Hp
secretion into the RPMI 1640 medium over time. Fig. 5 shows that both groups were able to
secrete Hp into the medium with a markedly greater secretion in group D than in group A.
Essentially, the released Hp reached maximal levels within the first 2 h tested. Thus, the
milk neutrophils not only express Hp but also release it into the milk.
2.3.7 Localization of Hp in mammary tissue and MAC-T cell
We used mouse anti-bovine Hp polyclonal antibody to localize the bovine Hp near the area of
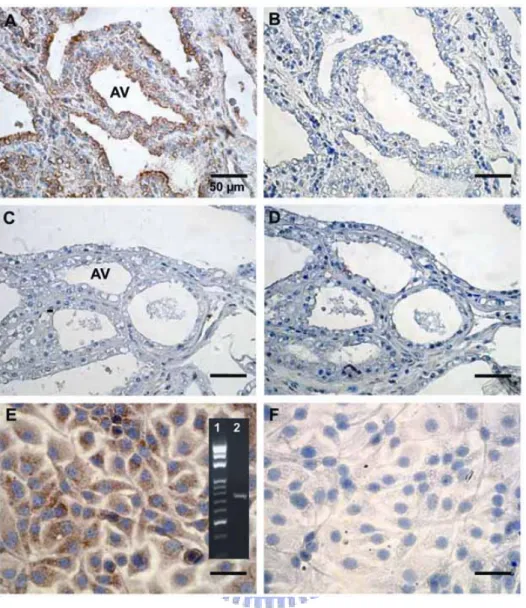
alveoli in mammary tissues (n = 5). Results indicate that only mastitic mammary tissue
expressed Hp relative to normal tissue. Figs. 6A and 6B illustrate a typical example
showing that Hp was primarily localized at alveoli of mastitic mammary tissue, but not in the
normal tissues (Figs 6C and 6D). To provide additional evidence that mammary alveolar
epithelial cells can express Hp, we showed that cultured- MAC-T cells were able to express
Hp mRNA (panel of Fig. 6E). We further demonstrated the presence of Hp protein in
MAC-T cells using immunocytochemical staining (Figs. 6E and 6F).
2.4 Discussion
2.4.1 Correlation between Hp levels and SCC in bovine milk
procedures. In general, SCC of a quarter greater than 500 × 103 cells/mL is considered to be
associated with mastitis [18, 19]. A limit of ≤ 100 × 103 cells/mL has been suggested for a
healthy quarter [20]. If the SCC exceeds 200 × 103 cells/mL, the quarter is likely to be
infected [21]. Results from the present study showed a 10-fold increase in Hp
concentrations in milk containing SCC > 500 × 103 (Group D) relative to SCC < 100 × 103
(Group A). It suggests that the somatic cells were responsible for the elevation of Hp in milk,
at least in part.
2.4.2 Bovine Hp in milk somatic cells
Previous studies have proposed that milk Hp can be derived from mammary tissues and
circulating blood following intramammary infection [22, 13]. Although the factors
attributed to the elevated milk Hp in naturally occurring mastitis are not fully understood, the
present study suggests that neutrophils in milk are a major source of milk Hp of cow with
mastitis. There are several lines of evidence to support this notion. First, Hp levels
determined by ELISA were positively correlated with the SCC in our group analyses in which
the increase in SCC was mostly attributed to neutrophils. Second, using Western blot
analysis Hp was found abundantly in the cell lysate of group D, but not in that of the normal
group A (Fig. 1). A recent study using matrix assisted laser desorption ionization-time of
granules of bovine granulocytes isolated from peripheral blood of healthy cattle, although the
cell types (such as neutrophil) have not been specified [23]. Third, the Hp mRNA levels in
cells of the high SCC groups B-D were significantly higher than in the cells of the normal
group A, The underlying mechanism involved in high Hp expression in the higher SCC
groups appears to be related to the different cell types. We also observed that neutrophils
were the predominate cell type in group D, but not in group A when examined by
immunocytochemistry (data not shown). This suggests that neutrophils in high SCC milk
account for the major synthesis of Hp. Furthermore, using laser confocal scanning
microscopy, we observed that all the neutrophils isolated from the somatic cells were capable
of expressing Hp as determined by double immunofluorescence (Fig. 4). Fourth, these cells
not only synthesized Hp but also released it into milk (Fig. 5).
This is the first study demonstrating that Hp is synthesized by cattle neutrophils using
RT-PCR, inconsistent with an early report indicating that human neutrophils do not produce
Hp [24]. In that study, the authors hypothesized that Hp of neutrophils was derived from the
liver and stored in specific granules via endocytosis [24]. While conducting our
investigation, a recent study reported that Hp can be released from human neutrophils [25]
indicating that the presence of Hp mRNA in human neutrophils, although the level of
expression was minimal. Taken together, it is conceivable that neutrophils may contribute to
2.4.3 Milk Hp from the other sources
It has been shown that Hp is expressed in the mammary tissue using ELISA and RT-PCR,
although the cell types involved were not specified [13]. Using an epithelial cell line
(MAC-T) and mammary gland tissue sections we further demonstrated that epithelial cells are
able to endogenously synthesize and express Hp (Fig. 6), consistent with results by Thielen et
al. [26] using an in situ hybridization on the epithelium of mastitic mammary tissue.
Another study has also suspected the presence of Hp in epithelium of alveolus [6].
Nonetheless, the localization of Hp in bovine alveolar epithelium of mastitic tissue or cultured
MAC-T cells is now demonstrated in the present study (Fig. 6).
2.4.4 Remarks on Hp secretion into the milk and its physiologic function
This study shows that neutrophils and mammary epithelial cells represent an additional
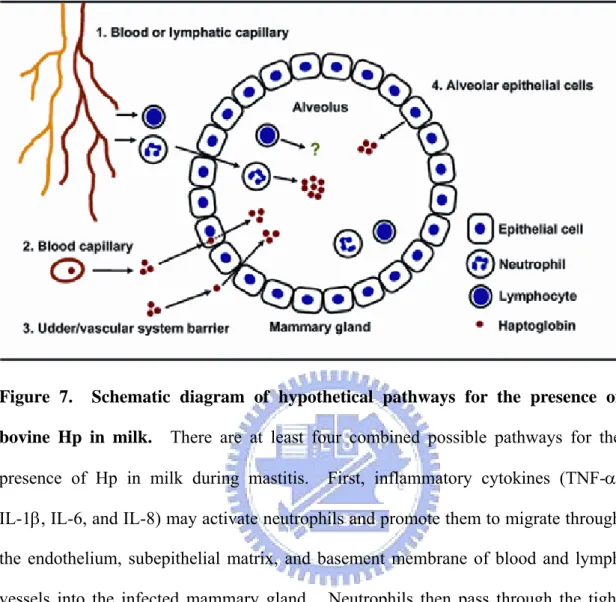
extra-hepatic source of Hp in milk. The possible mechanism by which neutrophils migrate
into the alveolus of mammary gland and how Hp is recruited in milk are explained and
summarized in Fig. 7. The physiologic benefits of local Hp expression in mammary tissue
during infection remain illusive. During mastitis, activated neutrophils produce significant
amount of reactive oxygen species (in order to kill the bacteria) which may cause tissue
free radicals, it may there effectively utilize Hp to attenuate such intracellular damage [7].
Further understanding of the role played by neutrophils as a source of Hp may provide insight
in to understanding an additional function of neutrophils in milk.
Acknowledgements
This work was supported by Grants NSC (94-95)-2313-B-009-001,
95-2313-B-009-003-MY2 and 97-2313-B-009-001-MY2 (SJTM) and NSC
94-2313-B-020-023 (JWL) from the National Science Council, Taiwan, R.O.C. We also
thank James Lee of the National Chiao Tung University for critical review of this manuscript.
References
[1] Eckersall P.D., Young F.J., Nolan A.M., Knight C.H., McComb C., Waterston M.M.,
Hogarth C.J., Scott E.M., Fitzpatrick J.L., Acute phase proteins in bovine milk in an
experimental model of Staphylococcus aureus subclinical mastitis, J. Dairy Sci. (2006)
89:1488-1501.
[2] Gervois P., Kleemann R., Pilon A., Percevault F., Koenig W., Staels B., Kooistra T.,
Global suppression of IL-6-induced acute phase response gene expression after chronic
in vivo treatment with the peroxisome proliferator-activated receptor-α activator
[3] Petersen H.H., Nielsen J.P., Heegaard P.M., Application of acute phase protein
measurements in veterinary clinical chemistry, Vet. Res. (2004) 35:163-187.
[4] Kristiansen M., Graversen J.H., Jacobsen C., Sonne O., Hoffman H., Law S. K.A.,
Moestrup S.K., Identification of the haemoglobin scavenger receptor, Nature (2001)
409:198-201.
[5] Langlois M.R., Delanghe J.R., Biological and clinical significance of haptoglobin
polymorphism in humans, Clin. Chem. (1996) 42:1589-1600.
[6] Lai I.H., Tsai T.I., Lin H.H., Lai W.Y., Mao S.J.T., Cloning and expression of human
haptoglobin subunits in Escherichia coli: delineation of a major antioxidant domain,
Protein Express. Purif. (2007) 52:356-362.
[7] Tseng C.F., Lin C.C., Huang H.Y., Liu H.C., Mao S.J.T., Antioxidant role of human
haptoglobin, Proteomics (2004) 4:2221-2228.
[8] Lai I.H., Lin K.Y., Larsson M., Yang M.C., Shiau C.H., Liao M.H., Mao S.J.T., A unique
tetrameric structure of deer plasma haptoglobin: an evolutionary advantage in the Hp 2-2
phenotype with homogeneous structure, FEBS J. (2008) 275:981-993.
[9] Lai Y.A., Lai I.H., Tseng C.F., Lee J., Mao S.J.T., Evidence of tandem repeat and extra
thiol-groups in the polymeric formation of bovine haptoglobin: a unique structure of Hp
2-2 phenotype, J. Biochem. Mol. Biol. (2007) 40:1028-1038.
in milk from dairy cows with chronic sub-clinical mastitis, Vet. Res. (2005) 36:191-198.
[11] Nielsen B.H., Jacobsen S., Andersen P.H., Niewold T.A., Heegaard P.M., Acute phase
protein concentrations in serum and milk from healthy cows, cows with clinical mastitis
and cows with extramammary inflammatory conditions, Vet. Rec. (2004) 154:361-365.
[12] Pedersen L.H., Aalbaek B., Røntved C.M., Ingvartsen K.L., Sorensen N.S., Heegaard
P.M., Jensen H.E., Early pathogenesis and inflammatory response in experimental bovine
mastitis due to Streptococcus uberis, J. Comp. Pathol. (2003) 128:156-164.
[13] Hiss S., Mielenz M., Bruckmaier R.M., Sauerwein H., Haptoglobin concentrations in
blood and milk after endotoxin challenge and quantification of mammary Hp mRNA
expression, J. Dairy Sci. (2004) 87:3778-3784.
[14] Chen W.L., Liu W.T., Yang M.C., Hwang M.T., Tsao J.H., Mao S.J.T., A novel
conformation-dependent monoclonal antibody specific to the native structure of
β-lactoglobulin and its application, J. Dairy Sci. (2006) 89:912-921.
[15] Chen W.L., Hwang M.T., Liau C.Y., Ho J.C., Hong K.C., Mao S.J.T., β-Lactoglobulin is
a thermal marker in processed milk as studied by electrophoresis and circular dichroic
spectra, J. Dairy Sci. (2005) 88:1618-1630.
[16] Huynh H.T., Robitaille G., Turner J.D., Establishment of bovine mammary epithelial
cells (MAC-T): an in vitro model for bovine lactation, Exp. Cell Res. (1991)
[17] Lee J.W., Zhao X., Recombinant human interleukin-8, but not human interleukin-1beta,
induces bovine neutrophil migration in an in vitro co-culture system, Cell Biol. Int.
(2000) 24:889-895.
[18] Olde Riekerink R.G., Barkema H.W., Veenstra W., Berg F.E., Stryhn H., Zadoks R.N.,
Somatic cell count during and between milkings, J. Dairy Sci. (2007) 90:3733-3741.
[19] Pyörälä S., Indicator of inflammation in the diagnosis of mastitis, Vet. Res. (2003)
34:565-578.
[20] Merle R., Schröder A., Hamann J., Cell function in the bovine mammary gland: a
preliminary study on interdependence of healthy and infected udder quarters, J. Dairy
Res. (2007) 74:174-179.
[21] Poutrel B., Stegemann M.R., Roy O., Pothier F., Tilt N., Payne-Johnson M., Evaluation
of the efficacy of systemic danofloxacin in the treatment of induced acute Escherichia
coli bovine mastitis, J. Dairy Res. (2008) 75:310-318.
[22] Eckersall P.D., Young F.J., McComb C., Hogarth C.J., Safi S., Weber A., McDonald T.,
Nolan A.M., Fitzpatrick J.L., Acute phase proteins in serum and milk from dairy cows
with clinical mastitis, Vet. Rec. (2001) 148:35-41.
[23] Cooray R., Waller K.P., Venge P., Haptoglobin comprises about 10% of granule protein
extracted from bovine granulocytes isolated from healthy cattle, Vet. Immunol.
[24] Wagner L., Gessl A., Parzer S.B., Base W., Waldhäusl W., Pasternack M.S., Haptoglobin
phenotyping by newly developed monoclonal antibodies. Demonstration of haptoglobin
uptake into peripheral blood neutrophils and monocytes, J. Immunol. (1996)
156:1989-1996.
[25] Theilgaard-Mönch K., Jacobsen L.C., Nielsen M.J., Rasmussen T., Udby L., Gharib M.,
Arkwright P.D., Gombart A.F., Calafat J., Moestrup S.K., Porse B.T., Borregaard. N.,
Haptoglobin is synthesized during granulocyte differentiation, stored in specific granules,
and released by neutrophils in response to activation, Blood (2006) 108:353-361.
[26] Thielen M.A., Mielenz M., Hiss S., Zerbe H., Petzl W., Schuberth H.J., Seyfert H.M.,
Sauerwein H., Short communication: Cellular localization of haptoglobin mRNA in the
experimenttally infected bovine mammary gland, J. Dairy Sci. (2007) 90:1215-1219.
[27] Boulanger V., Zhao X., Lacasse P., Protective effect of melatonin and catalase in bovine
neutrophil-induced model of mammary cell damage, J. Dairy Sci. (2002) 85:562-569.
[28] Lauzon K., Zhao X., Bouetard A., Delbecchi L., Paquette B., Lacasse P., Antioxidants to
prevent bovine neutrophil-induced mammary epithelial cell damage, J. Dairy Sci. (2005)
88:4295-4303.
[29] Tseng C.F., Mao S.J.T., Analysis of antioxidant as a therapeutic agent for atherosclerosis,
Curr. Pharm. Anal. (2006) 2:369-384.
37:369-400.
[31] Kolb A.F., Engineering immunity in the mammary gland, J. Mammary Gland Biol.
Neoplasia. (2002) 7:123-134.
[32] Monks J., Neville M.C., Albumin transcytosis across the epithelium of the lactating
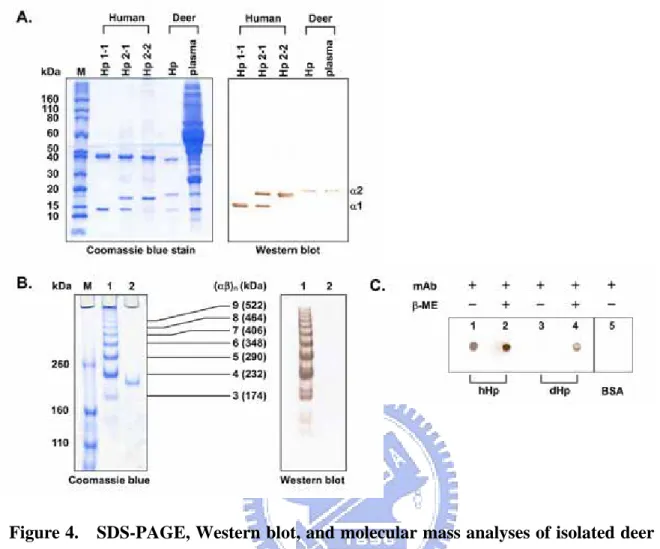
Figure 1. Typical SDS-PAGE pattern and Western blot analyses of milk somatic cell lysate from groups A and D. (A) Coomassie blue staining of somatic cell lysate run on a
15% SDS-PAGE under reducing condition. (B) Western blot analysis of the isolated Hp and somatic cell lysate under reducing (left panel with 15% SDS-PAGE) and non-reducing conditions (right panel with 4% SDS-PAGE) using a mouse polyclonal antibody prepared against bovine recombinant Hp. Lane M, molecular markers in kDa.
Figure 2. Expression levels of Hp mRNA in somatic cells. (A) Expression of Hp mRNA
in milk somatic cells according to SCC (A: < 100; B: 100-200; C: 201-500; and D: > 500 × 103 cells/mL with n = 5 in each group). Equal amount of total cDNA (100 ng) was amplified by PCR, using GAPDH as a house-keeping control. (B) Each bar represents the mean ± SEM. ∗∗∗ P< 0.001 as compared to group A.
Figure 3. A typical example of immunocytochemical staining between somatic cells isolated from groups A and D. Cytospin prepared cells were incubated with mouse
anti-CD5 (lymphocyte marker) (A and C) and anti-CD11b mAb (neutrophil marker) (B and D), followed by standard procedures using a LSAB method. AEC was used as the chromogenic substrate for CD5 or CD11b staining (in red), while hematoxylin was employed for nuclear staining
Anti-CD5 mAb (Lymphocyte) Anti-CD11b mAb (Neutrophil)
Group D Group A 50 µm 50 µm 50 µm 50 µm D A C B
Figure 4. Colocalization of CD11b and Hp in milk neutrophils using confocal laser scanning microscopy. CD11b as a marker for neutrophils was visualized with a
FITC-conjugated second antibody (A and B). Likewise, Hp was visualized with a rhodamine-conjugated second antibody (C and D).
10 µm
A
B
Figure 5. Time course of Hp protein released into RPMI 1640 medium by somatic cells from group A and D ex vivo. Freshly isolated somatic cells from milk samples of group A
and D (n = 3 of each group) at 4 ºC were immediately suspended in RPMI 1640 and kept at 37 ºC over time. Supernatant containing Hp was determined using an ELISA. Protease inhibitor (PMSF) was also added in parallel to each group in an attempt to minimize the Hp degradation. Each bar represents the mean ± SEM.
Figure 6. Immunocytochemical localization of Hp in mammary gland and MAC-T cell.
Mammary tissues with (A and B) and without mastitis (C and D) were incubated with a mouse anti-Hp polyclonal antibodies (A and C), while using normal non-immuned mouse serum as a negative control (B and D). MAC-T cells were incubated with mouse anti-Hp polyclonal antibodies (E), while using normal non-immuned mouse serum as a negative control (F). DAB was used as a chromogenic substrate for Hp staining (in brown) and hematoxylin was employed as a nuclear stain (in blue). Panel in E represents the Hp mRNA expression (Lane 2) and DNA molecular weight markers (Lane 1) using RT-PCR. AV = alveolar.
Figure 7. Schematic diagram of hypothetical pathways for the presence of bovine Hp in milk. There are at least four combined possible pathways for the
presence of Hp in milk during mastitis. First, inflammatory cytokines (TNF-α, IL-1β, IL-6, and IL-8) may activate neutrophils and promote them to migrate through the endothelium, subepithelial matrix, and basement membrane of blood and lymph vessels into the infected mammary gland. Neutrophils then pass through the tight junction between the alveolar epithelial cells and enter into the alveolus to secrete cellular Hp (please see review article for more detail [30]) (pathway 1). Second, plasma Hp of hepatic origin mimicking albumin and immunoglobulin [31, 32] is able to directly enter into the epithelial cells via a receptor-mediated process and secrete into alveolus (including endocytosis and exocytosis) (pathway 2). Third, plasma Hp is able to spill into the udder by passive diffusion through a compromised udder/vascular system barrier (pathway 3). Fourth, the mammary epithelial cells are able to endogenously synthesize and express Hp in the alveolus (pathway 4).
-41-Chapter 3
Cloning and expression of human haptoglobin subunits in Escherichia coli:
delineation of a major antioxidant domain (Paper 2)
Abstract
Human plasma haptoglobin (Hp) comprises α and β subunits. The α subunit is heterogeneous in size, therefore isolation of Hp and its subunits is particularly difficult. Using Escherichia coli, we show that α1, α2, β, and α2β chain was abundantly expressed and primarily present in the inclusion bodies consisting of about 30% of the cell-lysate proteins. Each cloned subunit retained its immunoreactivity as confirmed using antibodies specific to α or β chain. By circular dichroism, the structure of each expressed subunit was disordered as compared to the native Hp. The antioxidant activity was found to be associated with both α and β chains when assessed by Cu2+-induced oxidation of low density lipoprotein (LDL). Of remarkable interest,
the antioxidant activity of β chain was extremely potent and markedly greater than that of native Hp (3.5 x), α chain (10 x) and probucol (15 x). The latter is a clinically proved potent compound used for antioxidant therapy. The “unrestricted” structure of β subunit may therefore render its availability for free-radical scavenge, which provides a utility for the future design of a “mini-Hp” in antioxidant therapy. It may also provide a new insight in understanding the
-42-mechanism involved in the antioxidant nature of Hp.
Keywords: Human haptoglobin; α and β chains; cloning; mini-Hp fragment; antioxidant domain; monoclonal antibody; structure; circular dichroism
3.1 Introduction
Haptoglobin (Hp) is an acute-phase protein present in all mammals [1-3]. One of the major functions of Hp is to bind hemoglobin [4] and thereby prevent the oxidative tissue damage mediated by free hemoglobin [5]. We have recently demonstrated that chemically modified Hp losing its binding ability to hemoglobin, but is able to directly inhibit Cu2+ induced LDL
oxidation [6]. In humans, Hp is originally synthesized as a single polypeptide containing both α and β chains (Fig. 1A) [7]. Posttranslational cleavage between the Arg of α chain and Ile of β chain (followed by removal of Arg) results in the formation of a single α and β chain that is subsequently linked by disulfide bridges to form mature Hp [8].
The Hp gene is characterized by two common alleles Hp 1 and Hp 2 corresponding to α1β and α2β polypeptide chains, resulting in three main phenotypes: Hp 1-1, 2-1 and 2-2. DNA encoding for these two chains are linearly oriented in chromosome 16q22.1 [9] with 5 and 7 introns in Hp 1 and Hp 2, respectively. These introns, however, are exclusively located at the region corresponding to the α chain [5]. The α2 contains a unique nonhomologous
-43-crossing-over in a part of the α1 [10]. Hp 2-1 and 2-2 form polymers due to an additional –SH group at the α2 chain, which determines its phenotype specificity [11]. All the phenotypes share the same β chains (each with 245 amino acids). The α1 chain (each with 83 amino acids) is “monovalent” forming one disulfide bridge with α and β subunit resulting in Hp dimer (α1β)2.
In contrast, the α2 chain (each with 142 amino acids) is “divalent” containing one extra free –SH that is able to interact with an additional α2. As such, α2 chains can bind to either α1 or α2 chains to form large polymers [(α1β)2-(α2β)n in Hp2-1 and (α2β)n in Hp2-2]. The polymeric
phenotypes have been reported to be more prevalent in some groups of patients with certain diseases, such as diabetic and autoimmune diseases [5,12-14].
The purposes of this study were to produce recombinant Hp (rHp) α1, α2, β and α2β subunits in Escherichia coli expression system and to test whether E. coli contains a protease that may specifically cleave the linear αβ chain as that in mammalian cells [6,15], and to determine which subunit of Hp possesses antioxidant activity. We demonstrated that E. coli was devoid of a specific protease responsible for the cleavage of α and β chains. Each expressed α or β subunit not only retained the immunoreactivity, but also possessed the antioxidant activity. However, a major antioxidant domain was located in the β chain with a superior potency to probucol. Remarkably interesting, the unfolded structure of Hp, based on circular dichroic spectra, dramatically increased in antioxidant activity. The present study provides a potential
-44-utility for the future design of “mini-Hp” in developing a novel potent antioxidant. It may also provide a new insight in understanding the mechanism or specific amino-acids involved in the antioxidant nature of Hp.
3.2 Materials and methods 3.2.1 Materials
Escherichia coli JM109, M15 [pREP4], and the pQE30 expression vector were obtained from
Qiagen (Hilden, Germany). Plasmid preparation and gel-extraction kits were purchased from BD Biosciences (Palo Alto, CA). The proofreading DNA-polymerase and dNTP were purchased from Invitrogen (Carlsbad, CA). All restriction enzymes were purchased from New England Biolabs (Beverly, MA). T4-DNA-ligase and HiTrap chelating column were purchased from Fermentas (Burlington, Canada) and Amersham Biosciences (Uppsala, Sweden), respectively.
3.2.2 Preparation of mouse mAb and human Hp
Mouse monoclonal antibodies (mAb) 3H8 and G2D specific to human Hp α or β subunit were produced in our laboratory according to the standard procedures previously established [16]. Native Hp was isolated from human plasma by an immunoaffinity column using procedures
-45-previously established by us [17].
3.2.3 Plasmid construction of pQE30-Hp (α1, α2, β, α2β)
Gene fragments coding for human Hp α1, α2, β and α2β subunits were amplified by PCR using proofreading DNA-polymerase and oligonucleotide primers (Fig. 1A). The primer design was based on the published cDNA sequence of human Hp [18]. The primers were prepared for Hp α1 and α2 (forward 5'-GGGGTACCATGGTGGACTCAGGCAATGATGT-3' and reverse 5'-AACTGCAGTTACTGCACTGGGTTTGCCGGA-3'), Hp β (forward 5'-GGGGTACCATGA TCCTGGGTGGACACCTGG-3' and reverse 5'-AACTGCAGTTAGTTCTCAGCTATGGTCTT CT-3'), and Hp α2β (forward 5'-GGGGTACCATGGTGGACTCAGGCAATGATGT-3' and reverse 5'-AACTGCAGTTAGTTCTCAGCTATGGTCTTCT-3'). Both of Kpn I and Pst I restriction sites were incorporated into the 5' end of the forward sequence and reverse sequence primers, respectively. The cDNA of Hp α1, α2, β and α2β were ligated into the Kpn I / Pst I sites of an E. coli expression vector, pQE30. The plasmids were screened in JM109 and then expressed in M15 [pREP4]. Finally, the sequence of pQE30-Hp (α1, α2, β and α2β) was confirmed by DNA sequencing.
-46-E. coli [M15 (pREP4)] was transformed with the recombinant plasmid and cultured in l L of
Luria-Bertani (LB) medium containing ampicillin (100 µg/ml) at 37˚C on a rotary shaker. When the optical density reached 0.6 at 600 nm, the protein expression was induced by 1 mM IPTG at 37˚C for 2-4 h. The medium was centrifuged at 8,000 ×g for 5 min, and washed three times in a wash buffer containing 20 mM Tris-HCl, pH 8.0.
3.2.5 Disruption, wash, and isolation of inclusion bodies
The induced cells were then suspended in 40 ml of wash buffer and sonicated for 5 min at 4˚C, followed by centrifugation at 20,000 ×g for 20 min at 4˚C. The pellet containing the inclusion body was resuspended in 30 ml of 2 M urea containing 20 mM Tris-HCl, 0.5 M NaCl, 2% Triton X-100, pH 8.0 and sonicated as above, followed by centrifugation at 20,000 ×g for 20 min at 4˚C. Finally, the pellet was washed twice in wash buffer and stored frozen for late processing.
3.2.6 Solubilization of rHp subunits from inclusion bodies
The inclusion bodies were dissolved in a binding buffer containing 20 mM Tris-HCl, 0.5 M NaCl, 5 mM imidazole, 6 M guanidine-HCl, and 1 mM 2-mercaptoethanol, pH 8.0. The mixture was gently stirred at 4˚C for 12 h and the insoluble material was removed by centrifugation at 20,000 ×g for 20 min at 4˚C. Finally, the remaining soluble supernatant was then passed through a