72 pt 1 in 25.4 mm 54 pt 0.75 in 19.1 mm 54 pt 0.75 in 19.1 mm 54 pt 0.75 in 19.1 mm Margin requirements for first page
Paper size this page US Letter
Abstract—The purpose of this study is to investigate the cell attachment and viability on the micro-arc oxidization (MAO) microwave/hydrothermal treated titanium surfaces. The MAO samples were microwave irradiated in vessels containing 50mL of double-distilled water for 30 minutes (MWDD). The immersion solution consisted of 0.05mM calcium hydroxide and 0.03mM ammonium dihydrogen phosphate (MWCP), and same solution with the pH value adjusted to 9.6 (MWCP9.6) were compared. The hydrothermal (HT) samples were conducted under DD water at 250°C for 3hr. The cell viability (WST assay) results show that there was no significant different on the cell viability at 4hrs. After cultured for 1 day, the cell viability of MG63 cells on the samples from high to low is HT = MWDD = MWCP > MWCP9.6 > MAO. No significant different was found on the cell number between groups. However, the percentages of spreading cells to total adherent cells are higher on the MWDD, MWCP, and MWCP9.6 groups (75.7%, 69.9%, 69.4%, respectively), when compare to MAO and HT groups (48.4% and 41.5%). These results demonstrate that increased cell spreading on a MW groups with submicro-to- nano scale calcium phosphates on titania produced by microwave/ hydrothermal process.
I. INTRODUCTION
Titanium and its alloys have been widely used in medical applications such as artificial joints and dental implants because of their high strength-to-weight ratio and good biocompatibility. Nevertheless, they do not chemically bond to living bone. Various surface treatment techniques were developed to coat Ti metal and its alloys with bioactive calcium phosphate ceramics such as hydroxyapatite (HA) or tricalcium phosphate (TCP) to achieve a bone-bonding ability to Ti implants. Previous studies indicate that biomaterials surface with a roughness in the sub-micron or nano scale ranges can enhance osteoblasts adhesion, as well as the proliferation and differentiation expression when compared with nonmodified titanium surfaces.[1-3]
Micro-arc-oxidization (MAO) followed by hydrothermal treatment for titanium is a simple method to produce a biocompatible surface with micro scale needle-shape HA crystalline precipitates on the micro porous anatase titania surface.[4,5] However the bonding between Ti substrate and HA-MAO coating layer become weaker after the hydrothermal process.[6]
*Resrach supported by National Science Council Taiwan (NSC101-2320-B-039-004-MY3).
Dan-Jae Lin and Lih-Jyh Fuh are with China Medical University, Taichung, 40402 Taiwan, R.O.C. (Corresponding author: Dan-Jae Lin, Tel. +886-4-22053366 ext 7706, djlin@mail.cmu.edu.tw).
Chiing-Chang Chen is with Department of Science Application and Dissemination, National Taichung University of Education, Taichung, Taiwan, R.O.C.
Recently, we use MAO process following by microwave/hydrothermal treatment to produce calcium phosphates on the titania layer.[7] The outer shell of the MAO porous titania being rapidly covered with submicron-to-nano scale amorphous calcium phosphate (ACP) and TCP by 20 minutes of microwave irradiation. The study also demonstrated that crystalline HA and anatase titania were found in the coating. The process involving microwave irradiation is a time-saving and controllable method that could be suitable for synthesizing a calcium phosphates layer on a titanium surface. This study is to investigate the cell attachment and viability on the MAO and microwave/ hydrothermal treated titanium surfaces.
II. MATERIAL & METHODS A. Ti discs and Micro-Arc-Oxidation (MAO)
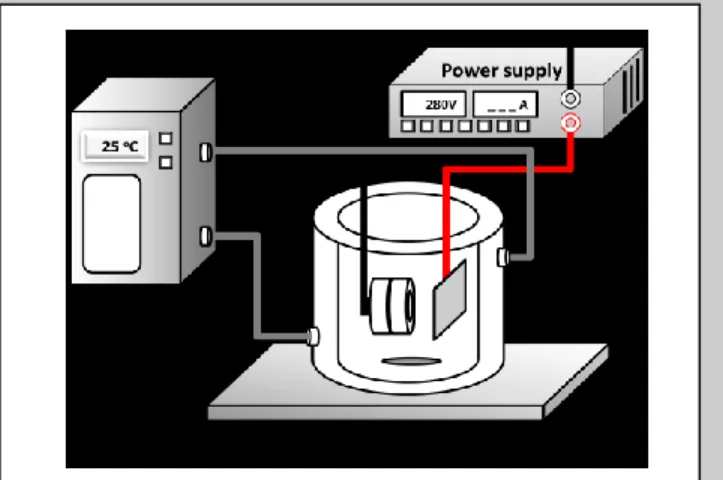
Commercially grade 2 pure titanium discs (12mm in a diameter and 1 mm in thickness) were polished using the grid SiC sand papers (#600), and rinsed with hydrofluoric acid and nitrate (1:3, v/v) in 15 seconds. Then, the Ti discs were cleaned 10 minutes ultrasonically in acetone, ethanol, and de-ionized water. Finally, the discs were vacuum-dried overnight at 50°C. The MAO process was performed at a galvanostatic mode under a constant current density of 10mA/cm2 to achieve a final voltage of 280V by a DC power supply for 3 minutes, the apparatus is shown in Figure 1.[8] Samples were cleaned with de-ionized water twice and were dried in a 55°C oven overnight.
Figure 1. Schematic view of the MAO process setup.
B. Hydrothermal and Microwave/Hydrothermal
The MAO-processed samples were immersed in vessels containing 50mL of double-distilled (DD) water and were irradiated by microwaves (2.45GHz, at 400W) for 30 minutes (MWDD). The solution consisted of 0.05mM calcium hydroxide and 0.03mM ammonium dihydrogen phosphate
Cell attachment and viability on micro-arc-oxidation (MAO)
microwave/hydrothermal treated titanium surface*
Dan-Jae Lin, Lih-Jyh Fuh, and Chiing-Chang Chen
54 pt 0.75 in 19.1 mm 54 pt 0.75 in 19.1 mm 54 pt 0.75 in 19.1 mm 54 pt 0.75 in 19.1 mm Margin requirements for the other pages
Paper size this page US Letter
(MWCP), and this solution with the pH value adjusted to 9.6 (MWCP9.6) for 30 minutes was compared (Figure 2). The MAO hydrothermal (HT) samples were prepared by immersing MAO-processed samples in a custom-made hydrothermal device containing 50mL of DD water at 250°C for 2.5hr. The specimens were cleaned ultrasonically with DD water twice and then vacuum dried before the analysis. It is noticeable that the micro scale HA needle precipitates on the MAO titania surface is very weak and will lose after ultrasonic, thus the HT samples was only with 3 dips in DD water.
Figure 2. Schematic view of the microwave/hydrothermal process setup. C. MG 63 Cell viability
MG63 human osteosarcoma cells were purchased from Bioresource Collection and Research Center (BCRC, Taiwan). The cells were grown in a minimum essential medium (α-MEM, Gibco, USA) containing nonessential amino acids and supplemented with 10% fetal bovine serum (FBS), 100mg/mL of streptomycin, 100U/mL of penicillin. Cells were maintained in a humidified atmosphere with 5% CO2 at 37°C. The culture medium was renewed twice per week. The cells were seeded at a density of 5×104 cells per well onto the autoclaved titanium discs and incubated in a 24-well plate for 4 and 24hours. Cell viability was evaluated using an enzymatic assay for mitochondria1 dehydrogenase activity (WST assay). The optical density (OD) value of the dissolved solute was then measured by an ELISA reader (Versa Max) at a 450nm wavelength. Measurements were performed in triplicate. Cell viability was calculated by the mean OD of three individual wells in the same group and expressed as normalized of control (MAO group).
D. Cell morphology and cell counting
To quantify the cell adherent/ spreading results, the Ti disc with cells on it was observed by scanning electronic microscopy (SEM, HITACHI S-4100 FEG-SEM). Samples were cleaned with PBS followed by 4% formaldehyde fixation, alcohol dehydration and critical point dried. Finally, the samples were fixed on a holder and coated with platinum.
The percentage of cells revealing spread morphology was quantified by dividing the number of spread cells by the total number of adherent cells. The spread cell is defined as the cell expends the lamellipodia to a double length of its original cell radius. Average percentages and standard deviations were calculated in five areas in 100 × pictures from two independent experiments.
III. RESULT
A. MG 63 Cell viability
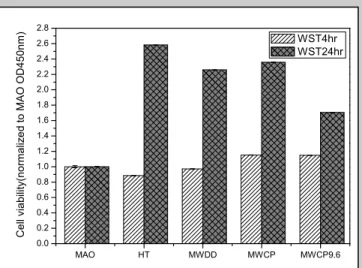
The cell viability (WST assay) results show that there was no significant different on the cell viability at 4hrs. After cultured for 1 day, the cell viability of MG63 cells on the samples were higher on HT, MWDD, and MWCP than that on MWCP9.6 and MAO groups (Figure 3).
Figure 3. Cell viability of MG63 cells cultured on samples for 4 hours and 24 hours.
B. Cell number and adherent morphology
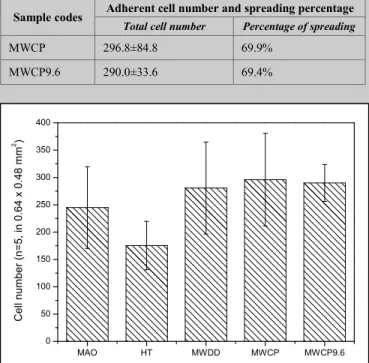
The adherent cell number of MG63 cells on the MAO, HT, MWDD, MWCP, and MWCP9.6 surface after culture for 4 hours are shown in Figure 4. The total number of adherent cells on MAO, MWDD, MWCP, and MWCP9.6 are similar, and the amount of adherent cell number on MWCP9.6 is significant higher than that on HT group. Most of the adherent cells on the MW groups (MWDD, MWCP, and MWCP9.6) are well spread in the 100× SEM pictures. The total cell numbers and calculated percentage of spreading cells are summarized in Table I. These results demonstrate that increased cell spreading on MW groups with submicro-to-nano scale calcium phosphates on the MAO titania surface.
TABLE I. ADHERENT CELL NUMBER AND SPREADING PERCENTAGE OF THE SAMPLES AFTER CULTUREED CELLS FOR 4 HOURS
Sample codes Adherent cell number and spreading percentage
Total cell number Percentage of spreading
MAO 245.0±74.7 48.4% HT 175.4±44.2 41.5% MWDD 280.6±84.1 75.7% MAO HT MWDD MWCP MWCP9.6 0.0 0.2 0.4 0.6 0.8 1.0 1.2 1.4 1.6 1.8 2.0 2.2 2.4 2.6 2.8 C e ll vi a b ili ty(n o rma lize d to MAO O D 4 5 0 n m) WST4hr WST24hr Infrared temperature sensor Microwave irradiation MAO Ti disc
54 pt 0.75 in 19.1 mm 54 pt 0.75 in 19.1 mm 54 pt 0.75 in 19.1 mm 54 pt 0.75 in 19.1 mm Margin requirements for the other pages
Paper size this page US Letter
Sample codes Adherent cell number and spreading percentage
Total cell number Percentage of spreading
MWCP 296.8±84.8 69.9%
MWCP9.6 290.0±33.6 69.4%
Figure 4. The adherent cell number of MG63 cells on the MAO, HT, MWDD, MWCP, and MWCP9.6 surface after culture for 4 hours.
IV. CONCLUSION
This study demonstrated that the 4hr viability and cell number of the MG63 cells in each group shows no more difference. The microwave/hydrothermal treated MAO titanium surface possess a better affinity to the MG63 cells than that of the MAO does, distinct morphological consequences in that more filopodia and lamellipodia at 4hr comparison. In the further, long-term in vitro cell assays should be performed to assess the effect on cell differentiation. Furthermore, in vivo experiments should allow more completed evaluation to give more evidence to optimize the surface characteristics with regard to the MAO processed and microwave/ hydrothermal treated titanium.
REFERENCES
[1] Webster TJ, Ergun C, Doremus RH, Siegel RW, Bizios R. Specific proteins mediate enhanced osteoblast adhesion on nanophase ceramics. Biomaterials 2001;22:1327–33.
[2] Yang WE, Hsu ML, Lin MC, Chen ZH, Chen LK, Huang HH. Nano/submicron-scale TiO2 network on titanium surface for dental implant application. Journal of Alloys and Compounds 2009;479 :642-647.
[3] Jimbo R, Sotres J, Johansson C, Breding K, Currie F, Wennerberg A. The biological response to three different nanostructures applied on smooth implant surfaces. Clin Oral Implants Res 2012;23:706-12. [4] Ishizawa H, Ogino M. Characterization of thin hydroxyapatite layers
formed on anodic titanium oxide films containing Ca and P by hydrothermal treatment. J Biomed Mater Res. 1995;29:1071-9. [5] Zhu X, Son DW, Ong JL, Kim K. Characterization of hydrothermally
treated anodic oxides containing Ca and P on titanium. J Mater Sci Mater Med. 2003;14:629-34.
[6] Lin DJ, Chen CY, Fuh LJ. Nano-sized calcium phosphates (CaPs) coatings on the anodic oxidized titania porous surface via a microwave irradiation process,9th World Biomaterials Congress, Chengdu, China, 2012.6.1 ~ 2012.6.5.
[7] Fuh LJ, Chen CY, Chen CC, Chen WC, Lin DJ. Microwave/ hydrothermal process to produce a sub-micron to nano-scale calcium phosphates on porous titania layer of the MAO titanium surface. Article
in preparation.
[8] Lin DJ, Tsai MT, Shieh TM, Huang HL, Hsu JT, Ko YC, Fuh LJ. In vitro Antibacterial Activity and Cytocompatibility of Bismuth Doped Micro-Arc Oxidized Titanium. Journal of biomaterials applications. 2013;27(5):553-563. MAO HT MWDD MWCP MWCP9.6 0 50 100 150 200 250 300 350 400 C e ll n u mb e r (n =5 , i n 0 .6 4 x 0 .4 8 mm 2 )