Physiea C 235-240 (1994) 763-764
N o r t h - H o l l a n d
PHYSICA @
A New Synthetic Route to the Formation of Fullerene Superconductors*
M. K. Wu and H.Y. Tang
Materials Science Center, National Tsing Hua University, Hsinehu, Taiwan, ROC J. H. Hwang and C. Y. Mou
Department of Chemistry, National Taiwan University, Taipei, Taiwan ROC
We report a new route to the formation of less air sensitive fullerene superconductors using
the mixture of C60 and Alkaline-metal carbonates. The materials so prepared contain oxygen in addition to the alkaline metal dopants. The alkaline metal content determined from single crystal x-ray is found to be 2.4 instead of the stoichiometry 3.
I. Introduction
The discovery of fullerene supercon- ductors [ 1 ] has attractive great attention for the last three years. However, the highly reactive of the materials with atmosphere hindered the progress for better understanding o f this molecular superconducting system. On the other hand, there are propsoals suggesting that the superconducting temperature (Tc) of this molecular system, as that of the high T c cuprate superconductors, may exhibit a maximum if one can vary the carrier concentration [2]. In an attempt to test this suggestion, we discovered a new novel synthetic route to the formation of a less air sensitive fullerene superconductors by mixing oxygen-contain alkaline compounds
. . . OtO --- .
II. Samples Preparation
Bulk samples of alkaliae carbonates (e.g. K2CO3) doped C60 were prepared by directly mixing appropriate amount of
C60 and alkaline-metal carbonates. The powder or pressed pellets were sealed in a fused quarts. The reactant-containing tube was preheated at---150°C under vacuum to remove moisture before sealing. The tube was heated in a furnace at 200°C for one hour, then at 700°C for 12 hours.
I[I. Results and Discussions
SEM micrograph and the composition spectra using EDX analyzer of the as grown crystals indicate that the sample contains, in addition to the potassium, also high concentration of oxygen. Single crystal X-ray analyses [3] show that the reacted product of K2CO3+C60 has a fcc structure with 14.2.42(4)A, lattice constant which is almost identical to that of" the . . . I ~ 11[i i~'.;IIiK;I J.L t~3t:60 1,14.24(i) [4]). Detaded--~ . . . of the single crystal x-ray data shows that the actual chemical composition foand in this work is K2.4C60, instead of the stoi- chiometry K3C60 determined by Stephens et al. [4].
*support by ROC National Science Council
0921-4534/94/S07.00 © 1 9 9 4 - I - i s e v i c r S c i e n c e B.V. A l l r i g h t s r c s c r v c d .
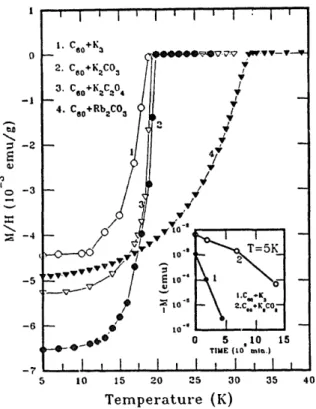
764 M.K. Wu et al./Physica C 235-240 (1994) 763-764 - ! .~ -4 1. C6o+Ka 2. C6o+KaCO~ O. Cso+K~C~04 4. Cso+ RbaCO3 ,y.tp.VV V VV V~ ¢~ --5 ~ ~ J / -Tt-.-~l t I 5 i0 15 , 'i i I , I : I ,'' I v / - 'V ,~ _ ¢
/
(
-
v v v~to ~ i I I ' IA' "ol --" -,I \ ,.%.K. I ' °F \
,o" I -I I I 0 5 .10 1,5 TIME {[0 rot,,.) i I ! ! z I i I , 20 25 30 35 40 T e m p e r a t u r e (K)Figure 1. Temperature dependence of ~.he magnetic susceptibility of K3C60 (1), C60+K2CO3 (2), C60+K2C204 ( 3 ) ~ d C60+Rb2CO3 (4); inset are the time dependence of the ZFC signals cf samples 1 and 2.
Figure I. shows the temperature de- pendence of the magnetic susceptibility of
several A2CO3-C60 samples. All meas-
urements were carried out with the sample exposed to air. We have also displayed in the inset of figure 1 the time evolution of the superconducting signals at 5 K, which are measured in zero field cooling, of a sample prepared by mixing C60 with K2CO 3 and the other with K-metal. The T c values, determined from the onset of superconducting transition, are found in general about two degrees higher thau their
corresponding relatives prepared from
Alkaline metals. The other novel result is the air stability of the materials prepared with the carbonates, as shown in the inset of figure 1. We found the stability of the materials prepared with this new process greatly improved. In fact, we believe the superconducting phase in this sample would actually be stabilized if the samples were not contaminated with the moisture.
The similarity in superconducting
transition of the samples prepared by using
alkaline-carbonates is surprising since
A2CO3 is not an electron donor. One
plausible mechanism is that the Alkaline- carbonate decomposes under the reacting conditions and the released K20 complex
reacted with the C60 molecules. The less
than 3 value of the potassium content may originate from the presence of oxygen which serves as an oxidative agent. The enhancement in the stability of the superconductivity can also be understood in terms of the inclusion of oxygen in these compounds. There were proposals [5] to generate hole doping under oxidative
condition. Whether the success in
incorporation oxygen to the Alkaline-metal
doped superconductors leads to the
realization of hole-doping in the fullerenes remains to be corroborated.
References
1. A. F. Hebard, et al., Nature, 350 (1991) 600.
2. R. Friedberg, et al., Phys. Rev. B46 (1992) 14150.
3. j. H. Hwang, et ai., unpublished. 4. P. W. Stephens., et al., Nature, 351
(1991)632.