Solanaceous Vegetable Breeding at AVRDC–The
World Vegetable Center to Meet the Challenges of
Climate Change in the Tropics
Peter Hanson1, Paul Gniffke2, Jin Shieh3, and Chee-wee Tan4
1Plant Breeder, AVRDC - The World Vegetable Center, PO Box 42, Shanhua, Taiwan 74199, E-mail: peter.hanson@worldveg.org
2 Plant Breeder, AVRDC - The World Vegetable Center, PO Box 42, Shanhua, Taiwan 74199, E-mail: paul.gniffke@worldveg.org
3 Assistant Specialist, AVRDC - The World Vegetable Center, PO Box 42, Shanhua, Taiwan 74199, E-mail: jin.shieh@worldveg.org
4 Principal Research Assistant, AVRDC - The World Vegetable Center, PO Box 42, Shanhua, Taiwan 74199, E-mail: chee-wee.tan@worldveg.org
Abstract
Abiotic and biotic stresses are major constraints of vegetable production in the tropics and climate change is expected to aggravate these problems. Depending upon the crop, the combined effects of multiple stresses may reduce total yield, reduce product quality, increase postharvest losses, and alter nutrient content. Consequently, vegetable varieties for the tropics should possess tolerance to heat, salinity, and other abiotic stresses, carry an array of disease and insect resistances, and still produce high yields of good quality produce that meet market requirements. Meeting this challenge requires robust protocols to screen and identify useful alleles from exotic germplasm and efficient selection methods to facilitate incorporation of multiple stress-tolerance genes into new varieties. Development of tropically adapted varieties of tomato (Solanum lycopersicum L.) and pepper (Capsicum
annuum) is a high priority at AVRDC – The World Vegetable Center. Tomato production in
the tropics has been devastated by increased incidence and severity of tomato yellow leaf curl diseases (TYLCD) caused by whitefly-vectored begomoviruses. Development of resistant varieties is complicated because the pathogen is highly diverse and almost all TYLCD resistance genes originated from wild tomato species. Application of marker-assisted selection (MAS) by AVRDC has facilitated development of tomato varieties homozygous for multiple begomovirus resistance genes that are expected to offer higher levels of resistance to a wider range of begomoviruses. Most modern tomato varieties are
sensitive to heat, drought, and salinity, but sources of stress tolerance have been found mainly in wild tomato accessions. AVRDC uses a multidisciplinary approach to map genes conditioning heat, drought, and salinity tolerance in tomato and to identify markers linked to targeted genes to facilitate gene introgression. Sweet pepper is a high value crop but sensitive to heat. AVRDC’s strategy to develop a tropical sweet pepper emphasizes the evaluation of sweet pepper lines under Taiwan summer stress (high temperatures and humidity) for yield components (fruit number, fruit weight) and traits linked to heat stress adaptation (pollen viability, root mass, vegetative biomass). Lines performing well for different components or traits have been identified and our hypothesis is that crossing lines with complementary traits and selection will lead to new lines with improved levels of heat tolerance. Breeding varieties adapted to climate change will shift more attention toward breeding for tolerance to abiotic stresses, which are often genetically and physiologically complex. Manipulation of multiple genes and traits will complicate vegetable breeding and will require effective use of conventional breeding techniques and molecular markers involving multidisciplinary teams.
Introduction
The solanaceous vegetables include globally important crops such as tomato, chili pepper and sweet pepper as well as regionally important crops such as eggplant (Solanum melongena), African eggplant (S. aethiopicum, S. anguivi, S. macrocarpon), and African nightshade (S. scabrum, S. villosum, others). Solanaceous crops are major significant sources of pro-vitamin A carotenoids, vitamin C, and other micronutrients. Production of high value horticultural crops offers smallholders in developing countries opportunities to substantially boost income and alleviate poverty. Because horticulture is labor- and capital-intensive, jobs are generated on-farm; transport and processing of horticultural products also creates off-farm employment for semi-skilled or unskilled workers (Weinberger and Lumpkin, 2005). Unfortunately, insect pests, diseases, high temperatures, salinity, drought and other factors limit solanaceous crop production for smallholder farmers in Africa, Central/South America, and Asia. For example, tomato yields are 10 to 12 t/ha in sub-Saharan Africa and tropical Asia, compared with 28 t/ha worldwide (FAO, 2006). It is expected that the direct and indirect effects of climate change will only exacerbate problems for small-scale tropical vegetable farmers.
Depending on the crop and region, the combined effects of multiple abiotic and biotic stresses can reduce total yield, increase unmarketable yield, lower produce quality, increase postharvest losses, or alter nutrient content of the produce. The possible effects of climate change should be assessed by crop, and appropriate breeding objectives and strategies designed. For example, fruit set and yield of most tomato and sweet pepper will be adversely affected as mean temperatures exceed 25 °C (de la Peña et al., 2011), while eggplant may not suffer significant declines in fruit set until mean temperatures exceed 30 °C. Climate change will impose multiple stresses that should be addressed by a combination of improved varieties and management practices. For most crops, tolerance to heat, drought, salinity and other stresses are both physiologically and genetically complex, which will complicate breeding programs. Even if stress tolerant varieties are developed, farmers may not adopt them unless these varieties meet the requirements of all stakeholders in the value chain, including farmers, transporters, wholesalers, retailers and consumers.
AVRDC’s breeding programs aim to develop varieties to expand opportunities in tropical vegetable production. Vegetable varieties for the tropics should possess multiple disease and insect resistance and tolerance to stress—and still produce high yields of high quality, nutritious produce. This paper will focus on two examples of approaches AVRDC uses to breed improved varieties better adapted to climate change: (1) pyramiding virus resistance genes via marker-assisted selection to manage tomato-infecting begomoviruses and (2) improving heat tolerance in sweet pepper.
Tomato varieties resistant to tomato yellow leaf curl disease for the
tropics
A direct relationship between climate change and the rapid spread of tomato yellow leaf curl disease (TYLCD) in Taiwan, the subtropics and tropics, is not certain; however, periods of warm and dry conditions favor rapid increase of whiteflies (Bemisia tabaci), the vector of the begomoviruses that cause TYLCD. Early begomovirus infection of susceptible tomato plants results in severe stunting, reduced leaf size, foliar chlorosis, and yield losses up to 100%. Early efforts by the worldwide tomato breeding community to identify TYLCD resistance in cultivated tomato were unsuccessful but high levels of resistance were found in some wild tomato species, especially some accessions of S. chilense, S. peruvianum, and S.
yellow leaf curl virus (TYLCV) resistance genes Ty-1, Ty-2, Ty-3, Ty-4, Ty-5 (Ji et al., 2007;
Anbinder et al., 2009). Early TYLCD resistance breeding at AVRDC focused on Ty-2, which offered excellent resistance to begomoviruses in Taiwan, south India, and parts of the Middle East. However, the failure of Ty-2 to provide resistance to Tomato yellow leaf curl
Thailand virus, first detected in Taiwan in 2005 (Tsai et al., 2011), and other major
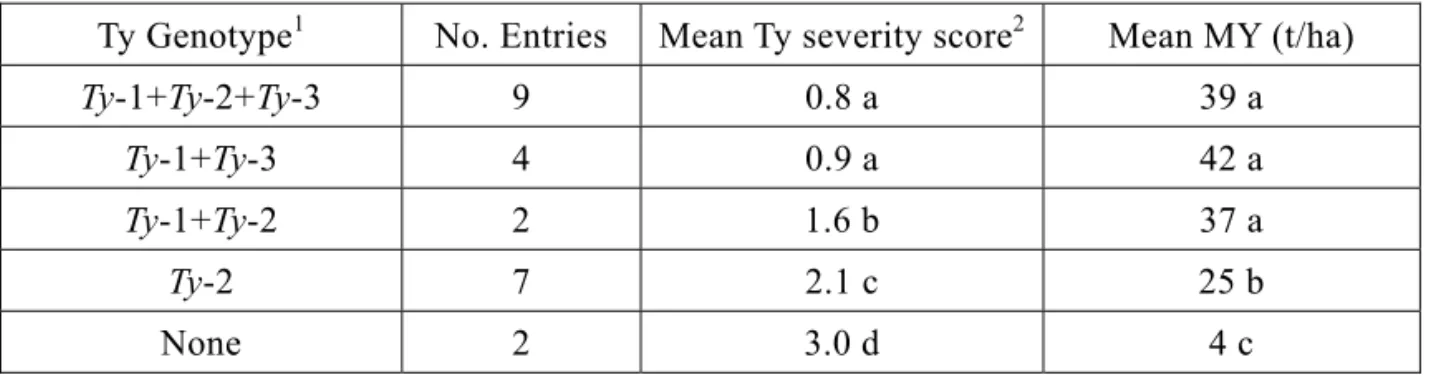
begomoviruses in the tropics prompted AVRDC to adopt a strategy of pyramiding multiple Ty genes into its lines to increase the chances of broad-based resistance to multiple begomoviruses. Screening for TYLCD resistance by exposing seedlings to viruliferous whiteflies and scoring plants for symptom severity was limited by several factors such as seasonal fluctuations of whitefly populations affecting TYLCD incidence; limitations imposed by screening with only the begomoviruses present in Taiwan; inability in some cases to determine by appearance whether targeted Ty genes were present in a particular line and whether genes were in homozygous or heterozygous condition; and wasted resources spent growing susceptible plants in the field. Fortunately, marker-assisted selection for TYLCD resistance genes offers many advantages, including the possibility of eliminating susceptible plants in a segregating population before transplanting, and the identification of plants homozygous for Ty-2 and Ty-3 or other combinations in early generations so that selection in later generations could focus on horticultural, fruit quality or nutritional traits. Routine application of marker-assisted selection (MAS) in tomato breeding began at AVRDC in 2007 with the availability of fast, relatively low cost polymerase chain reaction (PCR) protocols for Ty genes. Because Ty markers are not perfect, conventional exposure of populations and breeding lines to viruliferous whiteflies coupled with MAS reliably facilitated development of AVRDC lines with known combinations of resistance genes from 2009-2011 (Table 1). A 2009 AVRDC field trial of tomato lines in Taiwan under high TYLCD pressure indicated that the group mean of lines with combinations of
Ty-1+Ty-2+Ty-3, Ty-1+Ty-3, or Ty-2+Ty-3 yielded about nine-fold more than the susceptible
check (Table 2). Initial results from multilocation trials conducted by AVRDC cooperators in Jordan and Honduras indicate that AVRDC lines with the same two or three Ty gene combinations above display mild or no symptoms to TYLCD infection, although plants usually are begomovirus-infected.
Even though multiple Ty lines demonstrate effective resistance to many begomoviruses, the diversity of tomato-infecting begomoviruses increases the likelihood of virus forms that will overcome resistance. Use of cultural practices for TYLCD control such as pre- or
post-transplant nets to exclude whiteflies, application of imidacloprids, and crop-free periods coupled with resistance can augment TYLCD resistance. Varieties resistant to both begomoviruses and their vector offer great potential for durable TYLCD resistance. AVRDC is initiating work in collaboration with Cornell University to incorporate the acyl sugar insect resistance trait via MAS into AVRDC multiple Ty lines.
Heat tolerant sweet pepper
Among the persistent constraints to production of sweet pepper in tropical climates is the crop’s poor tolerance to high temperatures. Optimal temperatures for sweet peppers average 2-3 °C less than for hot chili peppers, and exposure to temperatures greater than 40 °C can cause dramatic reductions in net photosynthesis (Wu et al., 2001). When grown at high temperatures rather than at optimal cool temperatures, average fruit set and average fruit size are reduced, resulting in substantially reduced yield. AVRDC searches for genetic variability for these and other physiological parameters, and through recombination and selection, aims to improve the heat tolerance of sweet pepper. In addition to comparative yield component measurements gathered on varieties grown under contrasting temperature conditions (cool fall or winter vs. warm summer), we evaluate temperature sensitivity in root development and pollen viability.
An array of sweet pepper varieties, including heat-tolerant and susceptible checks, are grown in the field under spring and summer conditions, and flowers are harvested the day prior to expected anthesis. The flowers are dried under a heat lamp, and pollen is screened from them. The pollen is placed on glass microscope slides and a drop of 10% sucrose solution with 10 PPM boric acid is added. Samples are incubated at a given temperature for four hours, after which the germination process is stopped with a fixing solution (25% acetic acid, 75% ethanol). Slides are examined under magnification, and the percentage of germinated pollen grain and average pollen tube elongation are estimated. We have found that pollen viability declined progressively with increasing temperature beyond 36 °C, with most lines failing to germinate at temperatures greater than 39 °C; a very few lines, including heat-tolerant checks, may display low rates of pollen germination at temperatures as high as 42 °C. Pollen tube elongation mirrored these germination results. Heat tolerant lines incubated at 36 °C, produced tubes averaging 0.8 mm, while susceptible lines produced pollen tubes 0.2 mm or less in length. Heat tolerant lines could not be
distinguished from non-tolerant lines at incubation temperatures of 27 °C.
Roots of plant lines with contrasting heat tolerance were carefully excavated at monthly intervals, separated into three categories (primary [tap] root, secondary roots, and fibrous roots), cleaned, and dried. The dry weight of the primary root was best correlated with summer fruit yield potential (r=0.707). This study was compromised by the practice of establishing field crops via plug transplants, which often truncate the primary root.
Summary characterization of numerous genotypes for the noted field performance traits (fruit count, fruit weight, vegetative biomass, as well as relative fruit counts and weights under contrasting growing temperatures, root fraction weights, pollen viability, and pollen tube elongation at elevated temperatures) were compiled, and correlations were conducted with yield results under stressful conditions. Correlations of the several yield components and contributing developmental traits suggested that the most effective way to identify heat tolerant varieties is to grow them under stressful conditions, and select those that perform best. However, genotypes were frequently identified that excelled in one or another of these parameters, but which were not regarded as heat-tolerant. We hope that recombinations among selected parent lines and improvements in our methodologies will allow us to combine those genetic contributors to heat tolerance that are currently obscured by countervailing influences. Greater clarity in understanding the processes of response to elevated temperatures and their genetic control will allow us to make progress in developing superior sweet pepper with improved tolerance to high temperatures.
Outlook
For some solanaceous crops, especially tomato, sources of genes to improve abiotic traits include wild relatives, landraces, or other exotic germplasm. The problems of working with unadapted germplasm with poor horticultural traits and linkage drag, and traits conditioned by many small quantitative trait loci (QTLs) present tremendous challenges to plant breeders. Success will require strong interdisciplinary teams working to design effective protocols that identify trait sources, efficient high throughput marker protocols that effectively screen large populations for many markers, and basic research to understand the mechanisms of crop adaptation to stress.
Literature Cited
Anbinder, I., Reuveni, M., Azari, R., Paran, I., Nahon, S., Shlomo, H., Chen, L., Lapidot, M., and Levin, I. 2009. Molecular dissection of Tomato leaf curl virus resistance in tomato
line TY172 from Solanum peruvianum. Theor. Appl. Genet. 119: 519-530.
de la Peña, R.C., Ebert, E.W., Gniffke, P., Hanson, P., and Symonds, R.C. 2011. pp 396-410 in: Crop Adaptation to Climate Change. S.S. Yadav, R., Redden, J.L. Hatfield, H. Lotze-Campen, and A. Hall, A., eds. John Wiley and Sons, New Delhi.
Food and Agriculture Organization of the United Nations. 2006. Agricultural data FAOSTAT. April 2006.
http://faostat.fao.org/faostat/collections?version=ext&hasbulk=0&subset=agriculture Ji, Y., Scott, J.W., Hanson, P. Graham, E., and Maxwell, D. 2007. Sources of resistance,
inheritance, and location of genetic loci conferring resistance to members of the tomato-infecting begomoviruses. Pp. 343–266 in: Tomato Yellow Leaf Curl Virus Disease. H. Czosnek, ed. Springer, Dordrecht, the Netherlands.
Tsai, W.S., Shih, S.L., Kenyon, L., Green, S.K., and Jan, F.-J. 2011. Temporal distribution and pathogenicity of the predominant tomato-infecting begomoviruses in Taiwan. Plant Pathology 60: 787-799.
Weinberger, K. and Lumpkin, T.A. 2007. Diversification into horticulture and poverty reduction: A research agenda. World Development 35: 1464-1480.
Wu, H., Shou, S.Y., Zhu, Z.J., and Yang, X.T. 2001. Effects of high temperature stress on photosynthesis and chlorophyll fluorescence in sweet pepper (Capsicum frutescens L.) Acta Horticulturae Sinica, 28(6): 517-521.
170
Table 1. Elite AVRDC Multiple Ty tomato lines and checks identified from AVRDC trials, 2009-2011.
1
R=homozygous for resistance allele, S=homozygous for susceptible allele as determined by PCR. Ty3a is allelic to Ty3. SLM4-34 is a marker associated with a resistance gene near or allelic to Ty5.
2Values are percent healthy plants after drench inoculation with Ralstonia solanacearum (strain Pss4 from Taiwan) in the greenhouse.
AVRDC Distribution
code
Internal code Pedigree Tomato yellow leaf curl virus (TYLCV)
resistance genes1 Bacterial wilt Disease Resist.3 Fruit Shape4 Fruit wt
Ty1 Ty2 Ty3 Ty3a Ty5 SLM
4-34 % Surv 2 g AVTO1007 CLN3078A CLN3078F1-12-34-29-7-8-7 S R R S S S 16 TMV SQR 80 AVTO1132 CLN3078I CLN3078F1-12-34-27-9-4-10-19-14 S R R S S S 78 TMV SQR 69 AVTO1139 CLN3078L CLN3078F1-12-34-26-8-23-7-6 S R R S S S 85 TMV OBL 77 AVTO1141 CLN3109F CLN3109F1-26-35-11-2-29-10-11 S R S R S S 23 TMV SQR 68
AVTO1130 CLN3126A-7 CLN3126A-10-23-8-11-1-13-7 S R S R S S 53 TMV Plum 65
AVTO1002 CLN3125E CLN3125F2-21-15-7-25-14 R R R S S S 9 TMV, F-2 SQR 144
AVTO1133 CLN3125O-19 CLN3125F2-21-15-13-29-25-19 R R R S S S 78 TMV, F-2 OBL-R 84
AVTO1005 CLN3125P CLN3125F2-21-4-13-1-0 R R R S S S 83 TMV, F-2 OBL-R 69 AVTO1122 CLN3150A-5 CLN3150F1-4-18-8-26-4-5 S R S S R S 30 TMV PL-SQR 61 AVTO1143 CLN2819B CLN2819F1-2-1-4-25-6-13-13-21-4-4 R R R S S R 8 TMV OBL 62 AVTO1137 CLN3167B CLN3167F1-13-18-10-29-6-6-8 S S S S R S 73 TMV, F-2 R 94 AVTO0922 CLN3024A CLN3024F2-104-48-1-18-0 R R S S S S 87 - SQR 59 VI045743 TY52 (ck) BL982 R S S S S S NT PL 70 AVTO0301 CLN2498D (ck) CLN2498F1-68-15-22-17-19-12-17-8-0 S R S S S S 83 TMV OBL 56
Table 2. Group mean comparisons of Ty genotypes with different Ty gene combinations Evaluated under:
Ty Genotype1 No. Entries Mean Ty severity score2 Mean MY (t/ha)
Ty-1+Ty-2+Ty-3 9 0.8 a 39 a
Ty-1+Ty-3 4 0.9 a 42 a
Ty-1+Ty-2 2 1.6 b 37 a
Ty-2 7 2.1 c 25 b
None 2 3.0 d 4 c
1Lines within the group are homozygous for indicated Ty genes.
2Severity scores range from 0 (no symptoms) to 3 (severe leaf size reduction, curling, yellowing and plant stunting)
Entry means within columns followed by the same letter not significantly different according to single degree-of-freedom linear contrasts