Title Page:
Type of manuscript:Brief communications
Manuscript title: Gout and Parkinson's disease in older people: an
observation in Taiwan
Running head: gout and Parkinson's disease
Authors' full names:
Shih-Wei Lai, MD1,2; Chih-Hsueh Lin, MD and MS1,2; Cheng-Li Lin, MS6,7; Kuan-Fu Liao, MD and MS 3,4,5
1School of Medicine, 3Graduate Institute of Integrated Medicine, and
6Department of Public Health, China Medical University, Taichung, Taiwan
2Department of Family Medicine, and 7Management Office for Health Data, China Medical University Hospital, Taichung, Taiwan
4Department of Internal Medicine, Taichung Tzu Chi General Hospital, Taichung, Taiwan
5Department of Health Care Administration, Central Taiwan University of Science and Technology, Taichung, Taiwan
Corresponding author: Kuan-Fu Liao, MD and MS
Department of Internal Medicine, Taichung Tzu Chi General Hospital, No.66, Sec. 1, Fongsing Road, Tanzi District, Taichung City, 427, Taiwan
Phone: 886-4-2205-2121 Fax: 886-4-2203-3986
Disclosure of Conflicts of Interest
I certify that all my affiliations with or financial involvement in, within the past 5 years and foreseeable future, any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript are completely disclosed (e.g., employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, royalties).
The authors disclose no conflicts of interest.
Shih-Wei Lai,
Chih-Hsueh Lin
Cheng-Li Lin, Kuan-Fu Liao,
ABSTRACT
The goal of this study was to investigate the association between gout and Parkinson's disease in older people in Taiwan. Utilizing the Taiwan National Health Insurance database, this case-control study included 3854 subjects aged 65 years or older with newly diagnosed Parkinson's disease as the case group (mean age 75.0 years and standard deviation 5.0 years), and 15416 subjects without Parkinson's disease as the control group (mean age 74.0 years and standard deviation 5.3 years). Multivariable logistic regression analysis showed that no association was detected between gout and Parkinson's disease in both gender (odds ratio = 0.98, 95% CI = 0.86-1.12, in men and odds ratio = 1.03, 95% CI = 0.88-1.21, in women, respectively). We conclude that no association can be detected between gout and Parkinson's disease in older people in Taiwan.
In order to investigate the association between gout and Parkinson's disease in older people in Taiwan, we conducted this case-control study utilizing the Taiwan National Health Insurance database. The details of insurance program can be found in previous studies. This present study included 3854 subjects aged 65 years or older with newly diagnosed Parkinson's disease as the case group (1994 men and 1860 women, mean age 75.0 years and standard deviation 5.0 years) (based on International Classification of Diseases 9th Revision-Clinical Modification, ICD-9 332 and A-code A221), and 15416 subjects without Parkinson's disease as the control group (7976 men and 7440 women, mean age 74.0 years and standard deviation 5.3 years). Both groups were matched by age, sex, and index date in the database from 2000 to 2010. The index date was defined as the date of cases diagnosed with Parkinson's disease. Gout was diagnosed before the diagnosis of Parkinson's disease based on ICD-9 274. We analyzed the data separately by gender. Subjects with other major psychiatric diseases (ICD-9 291-293, 294.0, 294.8, 294.9, 295, 296.0, 296.1, 296.4–296.9, 297 and 298), mental retardation (ICD-9 317–319), and only diagnosis of hyperuricemia (ICD-9 790.61) but without gouty attack (ICD-9 274) were excluded from this study.
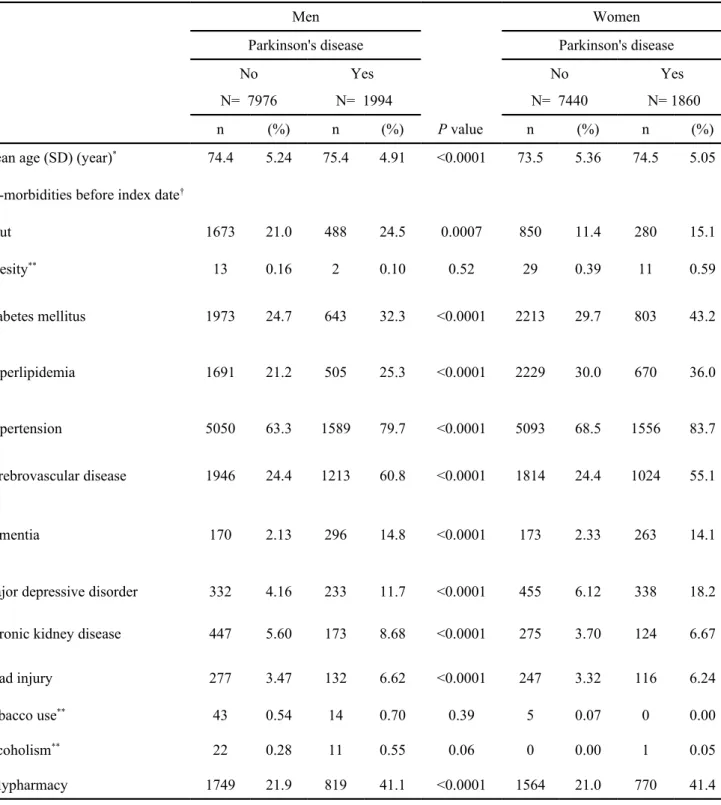
Table 1 shows the basic characteristics between Parkinson's disease cases and controls. In men, there were 488 subjects with gout among Parkinson disease cases (24.5%) and 1673 subjects with gout among control subjects (21.0%) (P = 0.0007).
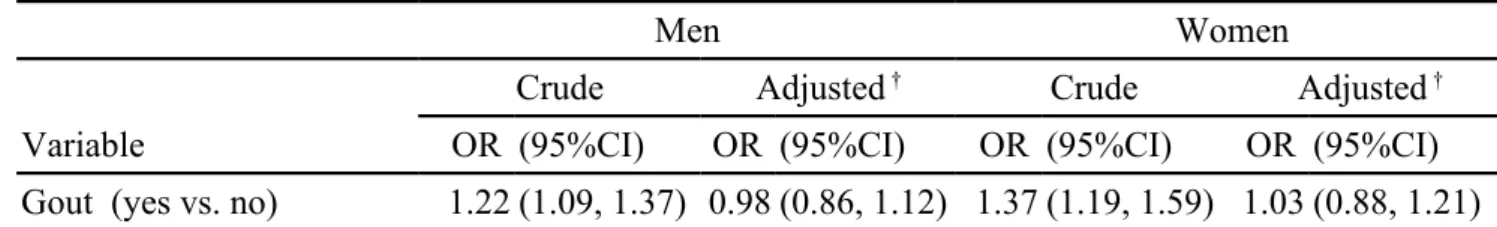
In women, there were 280 subjects with gout among Parkinson disease cases (15.1%) and 850 subjects with gout among control subjects (11.4%) (P < 0.0001). After controlling for covariates that were significantly related to Parkinson disease in the crude analysis, multivariable logistic regression analysis showed that no association could be detected between gout and Parkinson's disease in both gender (odds ratio = 0.98, 95% CI = 0.86-1.12, in men and odds ratio = 1.03, 95% CI = 0.88-1.21, in women, respectively) (Table 2). As a reference of non-gout group, the further analysis stratified by anti-gout therapy or not also showed that no association could be
detected between gout and Parkinson's disease, no matter whether the patients received anti-gout therapy or not (data not shown).
Recent studies have shown strong evidence that there is an inverse association of serum uric acid or gout with Parkinson's disease risk. 3-5 That is, individuals with hyperuricemia or gout may have lower risk of Parkinson's disease. Moreover, the evidence has shown that oxidative stress might be involved in Parkinson’s disease.6 The inverse association between Parkinson's disease and uric acid or gout can be partially explained by the mechanism that uric acid has an antioxidant effect which could reduce oxidative stress and it might further delay the progression of Parkinson's disease.7-9 A cohort study by De Vera M et al in Canada has shown that patients with gout had lower risk of Parkinson's disease compared with subjects without gout
(relative risk 0.70, 95% CI 0.59-0.83). 4 However, in this present study, we observed that no association could be detected between gout and Parkinson's disease in both gender. In further analysis, no association could be detected between anti-gout therapy and Parkinson's disease. Because this is only an observational study, we can not make a plausible explanation about the above different results. Moreover,
population selected and methodological characteristics may partially account for the conflicting results between the others studies and this present study. Therefore, more prospective studies are needed to elucidate this issue.
Some potential bias should be mentioned. First, since this database had no record of serum uric acid level. It would be possible to underestimate the prevalence of hyperuricemia. Second, the ICD-9 332 includes Parkinson's disease and secondary Parkinsonism. It would be possible to overestimate the prevalence of Parkinson's disease. Third, in addition to anti-gout drugs, some anti-parkinsonian drugs may also influence serum uric acid level. Forth, because of the time lag between index date of diagnosing Parkinson's disease and onset of Parkinson's disease, we could not make sure that gout attack was before or after onset of Parkinson's disease.
We conclude that no association can be detected between gout and Parkinson's disease in older people in Taiwan.
Funding
This study was supported in part by Taiwan Ministry of Health and Welfare Clinical Trial and Research Center of Excellence (MOHW103-TDU-B-212-113002). The funding agency did not infuence the study design, data collection and analysis, decision to publish, or
preparation of the manuscript.
The authors thank the National Health Research Institute in Taiwan for providing the insurance claims data.
Conflict of Interest Statement
REFERENCES
[1] Liao KF, Lai SW, Li CI, Chen WC. Diabetes mellitus correlates with increased risk of pancreatic cancer: a population-based cohort study in Taiwan. J Gastroenterol
Hepatol 2012; 27: 709-13.
[2] Lai SW, Lin CH, Liao KF, Su LT, Sung FC, Lin CC. Association between polypharmacy and dementia in older people: A population-based case-control study in Taiwan. Geriatr Gerontol Int 2012; 12: 491-98.
[3] Annanmaki T, Muuronen A, Murros K. Low plasma uric acid level in Parkinson's disease. Mov Disord 2007; 22: 1133-7.
[4] De Vera M, Rahman MM, Rankin J, Kopec J, Gao X, Choi H. Gout and the risk of Parkinson's disease: a cohort study. Arthritis Rheum 2008; 59: 1549-54.
[5] Winquist A, Steenland K, Shankar A. Higher serum uric acid associated with decreased Parkinson's disease prevalence in a large community-based survey. Mov
Disord 2010; 25: 932-6.
[6] Surendran S, Rajasankar S. Parkinson's disease: oxidative stress and therapeutic approaches. Neurol Sci 2010; 31: 531-40.
[7] Schlesinger I, Schlesinger N. Uric acid in Parkinson's disease. Mov Disord 2008;
23: 1653-7.
[8] Bowman GL, Shannon J, Frei B, Kaye JA, Quinn JF. Uric acid as a CNS antioxidant. J Alzheimers Dis 2010; 19: 1331-6.
[9] Alonso A, Sovell KA. Gout, hyperuricemia, and Parkinson's disease: a protective effect? Curr Rheumatol Rep 2010; 12: 149-55.
Table 1. Basic characteristics between Parkinson's disease cases and controls
Men Women
Parkinson's disease Parkinson's disease
No Yes No Yes
N= 7976 N= 1994 N= 7440 N= 1860
n (%) n (%) P value n (%) n (%)
Mean age (SD) (year)* 74.4 5.24 75.4 4.91 <0.0001 73.5 5.36 74.5 5.05
Co-morbidities before index date†
Gout 1673 21.0 488 24.5 0.0007 850 11.4 280 15.1 Obesity** 13 0.16 2 0.10 0.52 29 0.39 11 0.59 Diabetes mellitus 1973 24.7 643 32.3 <0.0001 2213 29.7 803 43.2 Hyperlipidemia 1691 21.2 505 25.3 <0.0001 2229 30.0 670 36.0 Hypertension 5050 63.3 1589 79.7 <0.0001 5093 68.5 1556 83.7 Cerebrovascular disease 1946 24.4 1213 60.8 <0.0001 1814 24.4 1024 55.1 Dementia 170 2.13 296 14.8 <0.0001 173 2.33 263 14.1
Major depressive disorder 332 4.16 233 11.7 <0.0001 455 6.12 338 18.2
Chronic kidney disease 447 5.60 173 8.68 <0.0001 275 3.70 124 6.67
Head injury 277 3.47 132 6.62 <0.0001 247 3.32 116 6.24
Tobacco use** 43 0.54 14 0.70 0.39 5 0.07 0 0.00
Alcoholism** 22 0.28 11 0.55 0.06 0 0.00 1 0.05
Polypharmacy 1749 21.9 819 41.1 <0.0001 1564 21.0 770 41.4 <0.0001
Data are presented as the number of subjects in each group, with percentages given in parentheses. Chi-square test, **Fisher’s exact test and * t-test comparing subjects with and without Parkinson's disease
†The co-morbidities included before index date were as follows: gout (ICD-9 274), obesity (ICD-9 278.00 and 278.01, and A-code A183), diabetes mellitus (ICD-9 250, and A-code A-181
hyperlipidemia (ICD-9 272.0, 272.1, 272.2, 272.3 and 272.4), hypertension (ICD-9 401-405, and A-code A260 and A269), cerebrovascular disease (ICD-9 430–438, and A-code A290-A299 dementia (ICD-9 290.0, 290.1, 290.2, 290.3, 290.4, 294.1 and 331.0), major depressive disorder (ICD-9 296.2, 296.3, 300.4 and 311), chronic kidney disease (ICD-9 585, 586, 588.8 and 588.9
Table 2. Odds ratios and 95% confidence intervals of Parkinson's disease associated with gout
Men Women
Crude Adjusted † Crude Adjusted †
Variable OR (95%CI) OR (95%CI) OR (95%CI) OR (95%CI)
Gout (yes vs. no) 1.22 (1.09, 1.37) 0.98 (0.86, 1.12) 1.37 (1.19, 1.59) 1.03 (0.88, 1.21)
†Adjusted OR in both gender: adjusted for age, diabetes mellitus, hyperlipidemia, hypertension, cerebrovascular disease,