©2010 Taipei Medical University
O R I G I N A L A R T I C L E
Background: Health care systems have recognized the importance of clinical effectiveness as demonstrated by systematic reviews (SRs). However, related efforts for developing SRs and its subsequent outcomes vary among countries, particularly in the Asia-Pacific region.
Purpose: This study examines the development of SRs and compares the performance among 11 countries and regions in the Asia-Pacific in order to identify feasible promotion strategies for alliances in this part of the world.
Methods: We retrieved data on published SRs from PubMed by employing previously developed search strategies to examine the developing situation, not only in general but also in each country and region. We then compared the performance of each country with regard to SRs in terms of several predefined aspects. In addition to comparing the raw number of publications, this study also took into account other factors such as the total number of physicians and gross domestic product.
Results: Among the 11 countries and regions included in the study, Australia set an out-standing example in SR activities. New Zealand, Singapore, Hong Kong, China, and India also contributed significantly to this body of knowledge. Japan, South Korea, and Taiwan could improve by producing more Cochrane or non-Cochrane style SRs.
Conclusion: The findings reveal the importance of governmental support for the develop-ment of SRs. This includes providing the required resources such as research infrastructure, funding, and manpower. The principles and methods of SRs also need further promotion. In addition, it is crucial to bring together all research partners in the region, particularly those with already established Cochrane entities, to reduce unnecessary barriers to communication and to accelerate progress in SR research.
Received: Sep 14, 2009 Revised: Jan 28, 2010 Accepted: Feb 6, 2010 KEY WORDS: Cochrane Library; evidence-based medicine; health policy; international comparison; systematic review
Comparison of the Increasing Number of Published
Systematic Reviews in Selected Countries in the
Asia-Pacific Region
Chiehfeng Chen
1,2,3,4, Heng-Lien Lo
5, Pei-Chuan Tzeng
4, Ya-Wen Chiu
5*,
Wen-Ta Chiu
6, Ken N. Kuo
5*
1Department of Public Health, School of Medicine, College of Medicine, Taipei Medical University, Taipei, Taiwan 2Graduate Institute of Biomedical Informatics, College of Medicine, Taipei Medical University, Taipei, Taiwan 3Department of Surgery, Taipei Medical University–Wan Fang Hospital, Taipei, Taiwan
4Evidence-Based Medicine Center, Taipei Medical University–Wan Fang Hospital, Taipei, Taiwan 5Division of Health Policy Research and Development, Institute of Population Health Sciences,
National Health Research Institutes, Zhunan, Miaoli, Taiwan
6Taipei Medical University, Taipei, Taiwan
*Corresponding authors. Both from the Division of Health Policy Research and Development, Institute of Population Health Sciences, National Health Research Institutes, 35 Keyan Road, Zhunan, Miaoli 35053, Taiwan.
Ya-Wen Chiu’s e-mail: bettychiu@nhri.org.tw Ken N. Kuo’s e-mail: kennank@nhri.org.tw
1. Introduction
In 1972, the British epidemiologist Archie Cochrane wrote Effectiveness and Efficiency: Random Reflections on
Health Services,1 which provided a succinct description of his ideas for improving the National Health Service in Britain. He suggested that all existing and new in-terventions should undergo properly designed evalua-tions, in particular, randomized controlled trials (RCTs), to demonstrate reliable clinical effectiveness. RCT is a scientific method that employs an epidemiological ex-perimental design to gain unbiased treatment results by using objectively collected data and statistical analysis. The intervention can be considered effective only if the benefits outweigh the cost.1 RCTs became the “gold standard” for verification of the effectiveness of clinical care and the basis for the subsequent development of evidence-based medicine (EBM).
The number of RCTs has grown rapidly, with the Cochrane Central Register of Controlled Trials holding more than 600,000 entries.2 Properly applying such a mas-sive amount of medical knowledge to clinical practice, however, has proven to be a great challenge. Moreover, the trials vary in quality. A systematic review (SR) can offer critical exploration, evaluation, and synthesis of the unmanageable amount of information and separates that which is insignificant, unsound, or redundant dead-wood in the medical literature from the salient and critical studies that are worthy of reflection.3
Cook and colleagues described SRs as scientific in-vestigations in themselves, with preplanned methods and an assembly of original studies as their “subjects”. By performing rigorous strategies to limit bias and ran-dom errors, SRs synthesize the results from multiple primary investigations.4
In response to Cochrane’s call for systematic, up-to-date reviews of RCTs and in order to leverage the power of the international community in promoting Cochrane’s ideals, 77 representatives and organizations from 11 countries came together and founded the Cochrane Collaboration in 1993. Since then, the core task of the Cochrane Collaboration has been to prepare, maintain, and disseminate SRs.5 Full Cochrane SR reports are col-lected in the Cochrane Database of Systematic Reviews (CDSR) and, with other Cochrane databases, form the Cochrane Library.
Now that the science of SR has undergone decades of evolution, it is broadly agreed that SRs are the least biased and most reliable way to summarize research evidence and provide support for decision makers. Moreover, the development of SRs has significantly driven the global EBM movement.6,7 The results of SRs are further presented in different evidence reports, to meet different demands and to different health care players. Decision-makers nowadays realize the impor-tance of using the best evidence available to make better-informed decisions about health care. Thus, it is
not surprising that EBM is now implemented in most developed countries. For example, the Roundtable on
Evidence-based Medicine, convened by the Institute of
Medicine in the United States, targeted that by 2020, over 90% of clinical decisions in the United States will be supported by accurate, timely, and up-to-date clinical information, and reflect the best available scientific evidence.8 The European Union and the World Health Organization (WHO) have also emphasized EBM, and worked closely together to promote EBM research and its applications in policy- and decision-making.9
Although SRs synthesize many studies and serve as an important part of EBM implementation, SRs have not yet gained much attention from the health care professions in Asia. For instance, it was not until 1999 that the first Cochrane entity was established in China (Table 1).10 And in Taiwan in the last decade, relatively few publications were conducted with an SR-related approach.11,12 In addition, health care professionals did not make frequent use of the Cochrane Library. A 2001 study based in a medical center in Taiwan showed that only 51.4% of physicians had experience using the Cochrane Library.13 A survey conducted in 2007 by the National Health Research Institutes among regional hospitals showed similar results.14 On the other hand, other countries in the Asia-Pacific region, such as Australia and New Zealand, have performed well in SRs. It is worth knowing the SR performance among countries in the Asia-Pacific region and identifying related facilitating strategies. The most direct way of comparing the SR per-formance of various countries would be to compare the number of published SRs from each country. Previous studies, however, have noted that this approach does not take into account other contributing components such as the number of researchers and economic situa-tion in each country. In addisitua-tion, the relative develop-ment between SR publications and total biomedical publications among different countries is another impor-tant aspect.15
The purposes of this study were to evaluate national SR research performance by comparing the SR publica-tions among selected countries in the Asia-Pacific region using objective measures, and to identify facilitating factors for promoting SR research accordingly.
2. Methods
2.1. Data sourcesThis study analyzed the SR literature indexed in PubMed (http://www.ncbi.nlm.nih.gov/sites/entrez) before the end of 2008. One of the challenges to identifying SRs in a medical database is the absence of a generally applied definition of SR. Although most SRs are generated by synthesizing many RCTs, RCT is a primary research re-sult, which means that the term RCT is not, by itself,
a sensitive key word for identifying SR literature. Mallett and Clarke’s study reported that each SR included an average of 6.6 trials, but also that a considerable number of trials were not included in any SR study.16 Since the
article is on SRs, we decided to employ SR and related words as the search key words. Moher and colleagues estimated an annual frequency of 2500 published articles, with about one fifth of these being Cochrane reviews.17
Montori et al suggested a more specific strategy to re-trieve SRs from MEDLINE. Among the various terms, “Cochrane database of systematic review.jn” was the most precise single term.18 Our previous experience
showed that only a small fraction of CDSRs could be found by searching for “systematic review” OR “system-atic literature review” in PubMed. In the present study, we took Montori et al’s suggestion in relation to PubMed searching strategy, employing the following: MEDLINE [Title/Abstract] OR (systematic[Title/Abstract] AND review [Title/Abstract]) OR meta-analysis[Publication Type] OR Cochrane Database Syst Rev [TA]. All searches were limited to dates from January 1, 1989 to December 31, 2008, and only to articles published in English.
The publications from each country were extracted by restricting the “affiliation” field in PubMed searches to individual country names and matching these with
prior search results for SRs by Boolean operators. The selected countries comprised Australia, China, India, Japan, South Korea, Malaysia, New Zealand, Singapore, Taiwan, and Thailand. Because of comparable patterns of social and health service development in advanced Asian economies, Hong Kong was added for comparison.19,20
Duplicate literature from China, Hong Kong and Taiwan were eliminated.
2.2. Comparison indicators
The overall trend of development in the field of SR lit-erature was first examined by applying Price’s Law.21,22
Price argued that the trend in a particular growing sci-ence production would be exponential. Countries were also compared with regard to their total number of SR publications.23 Previous studies have suggested that,
to some degree, the number of research publications is correlated with other social and health indices, such as the number of physicians and the per capita gross do-mestic product (GDP).15 In the present study, except for
Hong Kong and Taiwan, the number of physicians and GDP (adjusted by purchasing power parity) were re-trieved from WHO’s World Health Statistics.24–27 Time
spans and data resources are listed for each data set in
Table 1 Cochrane Centres and relevant branches in the Asia-Pacific region* Cochrane Centres
Organization location/established year Funding resources and branches
Australasian Based in Monash Institute of Health Services Funding from the Australian Government Department Cochrane Centre Research/Monash University (1994) of Health and Ageing, as well as from not-for-profit
health research funding agencies
New Zealand Located at the Department of Obstetrics Supported by the New Zealand Ministry of Health, Branch and Gynaecology, The University of The University of Auckland, and the New Zealand
Auckland (February 2004) Guidelines Group
Singapore Hosted by the Singapore Clinical Research Funded by the National Research Foundation (NRF) Branch Institute (SCRI) (July 2005)
Thai Cochrane Based in Khon Kaen University (2001) Supported by the WHO, UK and Australasian
Network Cochrane Centres, etc.
Chinese Cochrane Based in West China Hospital, Supported by the Ministry of Health, National Natural Centre Sichuan University (March 1999) Science Foundation of China, Sichuan University Hong Kong Based in the Chinese University of Supported by the Chinese University of
Branch Hong Kong (2002) Hong Kong
South Asian Located at the Christian Medical College, The Indian Council of Medical Research (ICMR), Cochrane Centre Vellore, India (South Asian Cochrane Effective Health Care Research Programme Consortium
Network since January 2005; officially UK (via DFID and the International Health Group, registered as a center in July 2008) Liverpool School of Tropical Medicine), the Cochrane
Schizophrenia Group (via the Department of Health, UK, and the University of Nottingham), Christian Medical College, Vellore, India, etc.
*The Cochrane Collaboration. Cochrane Centres. Available at: http://www.cochrane.org/contact/entities.htm#CENTRES [Date accessed: August 8, 2008]. WHO = World Health Organization; DFID = UK Department For International Development.
the results section. In addition, López-Muñoz et al pro-posed the participation index (PI) to further examine performance in a predefined subject.15 PI is the quo-tient calculated by the ratio of the number of publica-tions generated in a specific discipline or country to the total number of publications generated in that entity. We also examined the correlation between PI for SRs and PI for overall biomedical sciences in each country to determine the development of SR relative to all health sciences.15
3. Results
The publication of SR articles grew gradually until the end of the 20th century and climbed rapidly thereafter. In 1989, there were only 126 SR publications. The number had increased to 8501 in 2008. This indicates a total growth of 66.47 times over the 20-year period. Figure 1 gives the linear and exponential growth models for estimating the output of SR literature. The exponential model provides a correlation coefficient r2 of 0.9554, which indicates that only 4.5% of the variability is un-explained by the model. As shown in the graph, the exponential curve is more suitable than the linear model for illustrating the trend in SR publications over the last 20 years.
Despite the great interest in producing SRs around the world, outputs differed among regions and coun-tries (Table 2). In terms of the PI, nearly half of the SRs were produced by Australia (2554, PI = 0.51), followed by China (506, PI = 0.10), Japan (492, PI = 0.10), and
New Zealand (429, PI = 0.09). Taiwan had 129 SRs (PI = 0.03), slightly more than South Korea (124, PI = 0.02), Thailand (117, PI = 0.02), and Malaysia (15, PI = 0.00). Ranking by the total number of biomedical publications presen ted a different picture, with Japan taking the lead (PI = 0.46), followed by Australia (PI = 0.14) and China (PI = 0.10).
Table 2 also provides the country profiles in relation to total number of physicians and GDP. The data were ob-tained primarily from WHO’s World Health Statistics.24–27
In considering the different PI contributions in the field of SR and in overall health sciences production, Figure 2 shows the relationship between SR produc-tion and total producproduc-tion in biomedicine and health sciences among the subject countries. Australia, New Zealand, Hong Kong, Singapore, and Thailand had rela-tively higher productivity of SRs when compared to overall biomedicine and health science, while India, Malaysia, Taiwan, South Korea, and Japan had relatively low productivity.
Taking into account the social-health context of the different countries, the association between SRs and GDP per capita converted by purchasing power parity is drawn in the two-dimensional graph shown in Figure 3. There is no doubt that Australia ranks highest on both. Those countries located in the upper-left cor-ner of the graph (such as Singapore, Hong Kong, Japan, and Taiwan) have adequate economic capabilities but show less interest in SRs.
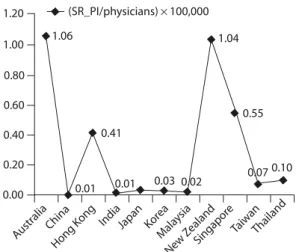
Figure 4 further outlines the correlation between SR production and research manpower in terms of the total number of physicians. As we carried out the anal-ysis by PI relative to the total number of physicians in
–2000 –1000 1000 2000 3000 4000 5000 6000 7000 8000 9000 10,000 0 1989 1990 1991 1992 1993 1994 1995 1996 1997 1998 1999 2000 2001 2002 2003 2004 2005 2006 2007 2008 Year Number o f pub lications y=8E – 162e0.1894x R2=0.9554 y=418.78x – 833,998 R2=0.8979
each country, the rankings echoed the results in Figure 2 and confirmed that the best performers were Australia, New Zealand, Singapore, Hong Kong, and Thailand. Figure 5 shows the proportion of CDSR and non-CDSR literature originating from the studied countries. Over half of the SRs were published in CDSR in Malaysia, fol-lowed by Thailand (40%), New Zealand (34%), Australia (26%), and India (15%). Fewer than 5% of SRs were published in CDSR in Japan, South Korea and Taiwan.
4. Discussion
After steady growth in the 1990s, SR production surged at the beginning of the 21st century. The increasing
publication of SRs, as Price’s Law suggests, highlights
Table 2 Country profiles in relation to systematic reviews, total biomedical publications and other socioeconomic health indices
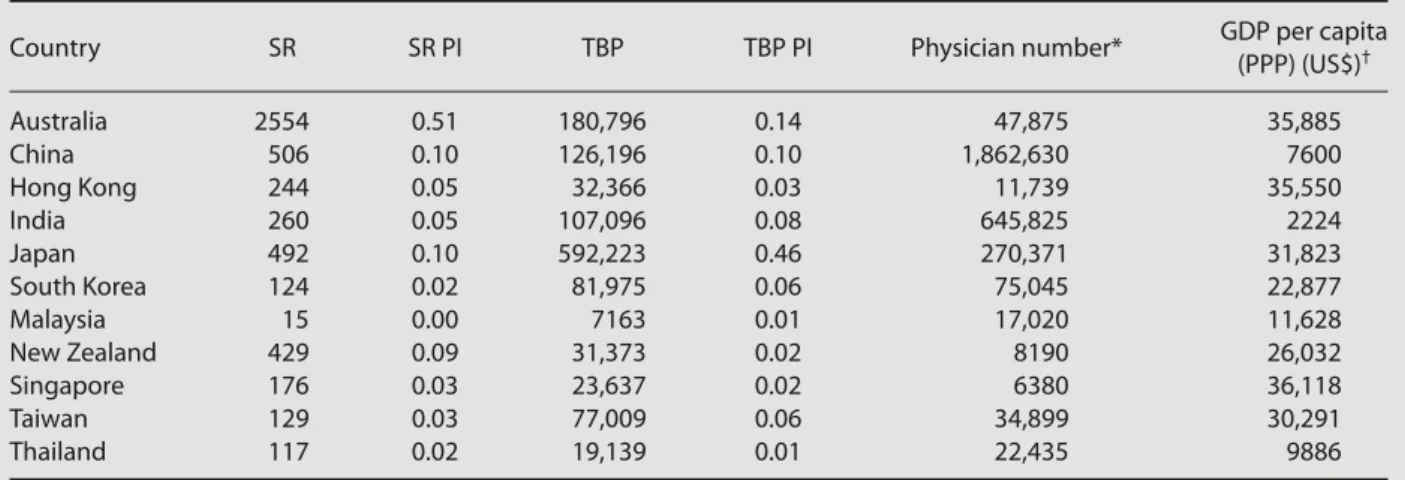
Country SR SR PI TBP TBP PI Physician number* GDP per capita
(PPP) (US$)† Australia 2554 0.51 180,796 0.14 47,875 35,885 China 506 0.10 126,196 0.10 1,862,630 7600 Hong Kong 244 0.05 32,366 0.03 11,739 35,550 India 260 0.05 107,096 0.08 645,825 2224 Japan 492 0.10 592,223 0.46 270,371 31,823 South Korea 124 0.02 81,975 0.06 75,045 22,877 Malaysia 15 0.00 7163 0.01 17,020 11,628 New Zealand 429 0.09 31,373 0.02 8190 26,032 Singapore 176 0.03 23,637 0.02 6380 36,118 Taiwan 129 0.03 77,009 0.06 34,899 30,291 Thailand 117 0.02 19,139 0.01 22,435 9886
*Except for Taiwan and Hong Kong, figures were obtained from Reference 24, while Taiwan’s physician number was obtained from Reference 25; †except for Taiwan and Hong Kong, figures were obtained from Reference 24, while Hong Kong’s GDP per capita (PPP) was obtained from
Reference 26, and Taiwan’s GDP per capita (PPP) was obtained from Reference 27. SR = systematic review; PI = participation index; TBP = total biomedical publications; GDP = gross domestic product; PPP = purchasing power parity.
Figure 3 Relationship between systematic review (SR) participation index (PI) and per capita gross domestic product (GDP) adjusted by purchasing power parity (PPP) among the studied Asia-Pacific countries.
0.00 0 5000 10,000 15,000 20,000 25,000 30,000 35,000 40,000 0.10 India China Australia Taiwan Japan Hong Kong Singapore Thailand Malaysia Korea New Zealand SR_PI GDP per capita (PPP) (US$) 0.20 0.30 0.40 0.50 0.60 2.58 2.47 0.91 Australia New Zealand Hong Kong Singapore Thailand China India Taiwan Korea Japan Malaysia 0.89 0.55 0.02 –0.38 –0.47 –0.58 –0.62 –0.79 –1.00 –0.50 0.00 0.50 1.00 1.50 2.00 2.50 3.00 (SR PI/TBP PI)-1
Figure 2 Country performance in systematic reviews (SR) compared to total biomedical publications (TBP). PI = partici-pation index.
the expansion of a developing area of science. In 2008, a total of 8501 new SR articles were found in PubMed. In other words, around 23.3 SR articles were released daily. In 1998, the equivalent figure was only 4.7, and in 1989 just 0.3.
Some factors have fostered the popularity of SRs in recent years. Each year, MEDLINE indexes over 560,000 new articles and Cochrane Central adds about 20,000 new randomized trials, approximately 1500 new arti-cles and 55 new trials everyday.28 It is therefore
neces-sary to develop an effective way to organize medical literature. The best medical knowledge management structure has been deemed the “5S” form: studies, syn-theses, synopses, summaries, and systems.29 SRs,
cate-gorized in the syntheses level, play an important role in synthesizing primary studies and converting them to a form that can support clinical decision-making. The process is also meaningful to decision-makers at the policy and country levels.30 The most influential
driv-ing force in this synthesis was the 1993 launch of the
Cochrane Collaboration, which aimed to produce and disseminate SRs of health care interventions.5 Mallett and Clarke’s study estimated that at least 10,000 SRs would be needed to digest all the clinical trials in the Cochrane Central Register of Controlled Trials, not to mention the demand of regularly maintaining published SRs to keep them up-to-date.16 More recently, the CDSR has been listed in the Science Citation Index (SCI) with an impact factor of 5.182 in 2008, the 12th highest in the “Medicine, General & Internal” category.31 This rec-ognition should attract more interest in the field of SRs within the academic society.
A peak in SR output appeared in 2000. This sudden rise was caused by a PubMed decision to list CDSR be-ginning in that year. Since then, PubMed has indexed around 400–500 CDSR articles annually, which account for one in 10 of the total SR articles retrieved from a PubMed search. The proportion of SRs in our study is relatively low compared with other studies. Using dif-ferent inclusion criteria in November 2004, Moher et al identified an estimated annual frequency of 2500 pub-lications of SRs indexed in MEDLINE, of which about 20% were Cochrane reviews.17 Montori and colleagues, however, employed rigorous criteria and concluded that CDSR published 56% of all SRs during their study period in 2000.32 Although we applied Moher et al’s cri-teria, the variation is unavoidably larger than previous studies because of the long study period and the tre-mendous number of publications, which make it un-feasible to apply rigorous inclusion criteria such as an independent screening of all eligible articles.
Compared to the overall development of biomedi-cal sciences in different countries, SR did not develop equally among the Asia-Pacific countries we studied. On the one hand, countries such as Australia and New Zealand made excellent progress, while on the other hand, countries in East Asia such as Taiwan, South Korea, and Japan lagged far behind their counterparts. It is noteworthy that the well-performing countries re-ceived strong support from the local Cochrane entity. According to the structure of the Cochrane Collaboration, several Cochrane Centres and relevant branches were founded to help coordinate and support members of the Collaboration and promote the objectives of the Collaboration at the national level. Within the Asia-Pacific region, related Cochrane branches were estab-lished in New Zealand, Singapore, and Thailand and supervised by the Australasian Cochrane Centre. Other centers include the South Asian Cochrane Centre, which is located in India, and the Chinese Cochrane Centre, which is situated in Sichuan, China, and which has a branch in Hong Kong. Most of the Cochrane entities mentioned above are funded by governments and con-sequently facilitate the output of SRs. The contribution of Cochrane entities is significant when analyzing the proportional distribution of CDSR and non-CDSR SR papers. Obviously, there is room for countries such as Austra
lia China
Hong
Kong India Japan Kor ea Mala ysia New Zea land Singapor e TaiwanThai land 0.00 0.20 0.40 0.60 0.80 1.00 1.20 1.06 0.41 0.01 0.01 0.03 0.02 1.04 0.55 0.07 0.10 (SR_PI/physicians)×100,000
Figure 4 Relationship between systematic review (SR) partici-pation index (PI) and physician numbers in the studied Asia-Pacific countries. 0 Malaysia Thailand New Zealand Australia India China Singapore Japan Korea Taiwan 129 122 481 229 160 451 220 1892 283 70 7 8 47 146 662 40 55 16 15 11 2 0 Hong Kong 10 20 30 40 50 Percentage 60 70 80 90 100 CDSR publication Non-CDSR publication Figure 5 Cochrane Database of Systematic Reviews (CDSR) and non-CDSR publications among the studied Asia-Pacific countries.
Japan, South Korea, and Taiwan to improve their partici-pation in generating and promoting SRs. One feasible way to address this is to call for cross-national coopera-tion across the East Asian region and to learn from the experiences of countries with well-established Cochrane entities such as Australia and New Zealand.
The correlation between scientific production of SRs and domestic economic circumstances in each country provides a way to examine governmental priorities. Generally speaking, the higher the socioeconomic sta-tus, the greater the research productivity. However, we did not observe this pattern with regard to SR publica-tions. Advanced Asian health systems, such as those of Japan, Singapore, Hong Kong, and Taiwan, seemed to be underproductive in this regard. This finding points out that the scientific achievement of a country in a particular field might inevitably be influenced by its policies on scientific research and development.
When we considered the total number of physicians, as representative of the professional manpower directly related to clinical research, the leading places were Australia, New Zealand, Singapore, and Hong Kong. This interesting shift in order, particularly in the cases of Hong Kong and Singapore, may be because both have the advantage of English as their official language. For those countries in which English is not their official language, the strategies of paving the way for the gen-eration of SRs may need to take into account language barriers and provide related support. This is important not only for those countries seeking to enhance their research capabilities, but also for the whole Cochrane society which is concerned with fostering greater diver-sity in the new century.
This study has some limitations. First, the search was limited to English-language literature and thus did not take into account what related literature there may be in other languages. We further examined the pro-portion of non-English literature based on our previous searching strategy. Among the selected countries and regions, China had 25% non-English SR publications, while the rest had non-English SR publications making up less than 6% of their totals. We believe that the most important and highest-quality studies have been pub-lished in English-language journals. Second, instead of comprehensively searching all databases, our search focused only on PubMed. The coverage of literature among different databases varies by topic.33–36 Although common practice is to combine MEDLINE and EMBASE to get a thorough set of reviews, we did not search EMBASE as it includes more European literature which did not seem as useful in this study that focused on the Asia-Pacific region.37 We believe that the trends we found would be the same even if we had included other databases.
In summary, over the past 20 years, health care pro-fessionals around the world have endeavored to de-velop, conduct, and publish SRs. The importance of SRs
will continue into the foreseeable future. Among the Asia-Pacific countries and regions, Australia has set an outstanding example in the development of SRs as well as EBM activities. To some extent, New Zealand, Singapore, Hong Kong, China, and India have also con-tributed significantly to this body of knowledge. Japan, South Korea, and Taiwan can improve their participation by producing more Cochrane SRs. The findings from our research suggest that, as demonstrated by other neighboring Asia-Pacific counties, it is important to have governmental support for building the capacity of SR. The SR is inevitably resource-demanding, including re-search infrastructure, funding, and rere-search manpower. Bringing together all research partners around the region, particularly those with already established Cochrane entities, is also crucial to reducing unnecessary barriers to communication and to accelerating progress in SR research.
References
1. Cochrane AL. Effectiveness and Efficiency: Random Reflections on Health Services. London: Royal Society of Medicine Press, 1999: 1–44.
2. The Cochrane Library. Product Descriptions. Available at: http:// www3.interscience.wiley.com/cgi-bin/mrwhome/106568753/ ProductDescriptions.html#central [Date accessed: January 13, 2010]
3. Mulrow CD. Systematic reviews: rationale for systematic reviews. BMJ 1994;309:597–9.
4. Cook DJ, Mulrow CD, Haynes RB. Systematic reviews: synthesis of best evidence for clinical decisions. Ann Intern Med 1997;126: 376–80.
5. Chalmers I. The Cochrane collaboration: preparing, maintaining, and disseminating systematic reviews of the effects of health care. Ann N Y Acad Sci 1993;703:156–63.
6. Moynihan R. Evaluating Health Services: A Reporter Covers the Science of Research Synthesis. New York: Milbank Memorial Fund, 2004:6–21.
7. Hill GB. Archie Cochrane and his legacy: an internal challenge to physicians’ autonomy? J Clin Epidemiol 2000;53:1189–92. 8. Institute of Medicine. The Learning Healthcare System: Workshop
Summary. Washington, DC: The National Academies Press, 2007:ix.
9. Figueras J, McKee M, Lessof S, Duran A, Menabde N. Health Systems, Health and Wealth: Assessing the Case for Investing in Health Systems. WHO European Ministerial Conference on Health System, June 25–27, 2008, Tallin, Estonia. Copenhagen: WHO Regional Office for Europe, 2008.
10. The Cochrane Collaboration. Cochrane Entities. Available at: http://www.cochrane.org/contact/entities.htm [Date accessed: August 8, 2009]
11. Hsu WA, Chung KP. Assessing the quality of effectiveness studies for systematic review in evidence-based medicine. J Health Sci 2005;7:307–16. [In Chinese]
12. Shau WY. How to critically appraise medical literatures. Formos J Med 2003;7:535–42. [In Chinese]
13. Yu M, Huang C, Lai Y. A survey of physicians’ knowledge and usage of the medical database. Chin J Family Med 2001;11:128–38. [In Chinese]
14. Chiu YW, Weng YH, Lo HL, Ting HW, Hsu CC, Shih YH, Kuo KN. Physicians’ characteristics in the usage of online database: a
representative nationwide survey of regional hospitals in Taiwan. Inform Health Soc Care 2009;34:127–35.
15. López-Muñoz F, Alamo C, Quintero-Gutiérrez F, García-García P. A bibliometric study of international scientific productivity in attention-deficit hyperactivity disorder covering the period 1980– 2005. Eur Child Adolesc Psychiatry 2008;17:381–91.
16. Mallett S, Clarke M. How many Cochrane reviews are needed to cover existing evidence on the effects of healthcare interventions? Evid Based Med 2003;8:100–1.
17. Moher D, Tetzlaff J, Tricco AC, Sampson M, Altman DG. Epidemiol-ogy and reporting characteristics of systematic reviews. PLoS Med 2007;4:e78.
18. Montori VM, Wilczynski NL, Morgan D, Haynes RB, for the Hedges Team. Optimal search strategies for retrieving systematic reviews from Medline: analytical survey. BMJ 2005;330:68.
19. Gauld R, Ikegami N, Barr MD, Chiang TL, Gould D, Kwon S. Advanced Asia’s health systems in comparison. Health Policy 2006;79:325–36.
20. Wagstaff A. Health systems in East Asia: what can developing countries learn from Japan and the Asian tigers? Health Econ 2007;16:441–56.
21. Price DJ. Little Science, Big Science. New York: Columbia University Press, 1963:1–30.
22. Price DJ. Networks of scientific papers. Science 1968;149:510–5. 23. Groneberg-Kloft B, Scutaru C, Kreiter C, Kolzow S, Fischer A,
Quarcoo D. Institutional operating figures in basic and applied sciences: scientometric analysis of quantitative output bench-marking. Health Res Policy Syst 2008;6:6.
24. World Health Organization. World Health Statistics 2009. Geneva, Switzerland: World Health Organization, 2009:95–118.
25. Department of Health, R.O.C. Health Statistics in Taiwan. Taipei, Taiwan: Department of Health, 2008:73–82.
26. International Monetary Fund. World Economic Outlook Database 2009. Available at: http://www.imf.org/external/pubs/ft/weo/2009/ 01/weodata/index.aspx [Date accessed: May 14, 2009]
27. Directorate General of Budget, Accounting and Statistics, Executive Yuan, R.O.C. National Statistics, Republic of China (Taiwan), 2009. Available at: http://eng.stat.gov.tw/mp.asp?mp=5 [Date accessed: May 14, 2009]
28. Glasziou P, Haynes B. The paths from research to improved health outcomes. Evid Based Med 2005;10:4–7.
29. Haynes B. Of studies, syntheses, synopses, summaries, and systems: the “5S” evolution of information services for evidence-based healthcare decisions. Evid Based Med 2006;11:162–4.
30. Lavis JN, Lomas J, Hamid M, Sewankambo NK. Assessing country-level efforts to link research to action. Bull World Health Organ 2006;84:620–8.
31. The Cochrane Library. Issue 3 Highlights 2009. Available at: http://www3.interscience.wiley.com/homepages/106568753/ highlights_pm.pdf [Date accessed: August 8, 2009]
32. Montori V, Wilczynski N, Morgan D, Haynes RB, the Hedges Team. Systematic reviews: a cross-sectional study of location and citation counts. BMC Med 2003;1:2.
33. Royle P, Bain L, Waugh N. Systematic reviews of epidemiology in diabetes: finding the evidence. BMC Med Res Methodol 2005;5:2. 34. Wilkins T, Gillies R, Davies K. EMBASE versus MEDLINE for family
medicine searches: can MEDLINE searches find the forest or a tree? Can Fam Physician 2005;51:848–9.
35. Löhönen J, Isohanni M, Nieminen P, Miettunen J. Coverage of the bibliographic databases in mental health research. Nord J Psychiatry 2009 Nov 2. [Epub ahead of print]
36. Slobogean GP, Verma A, Giustini D, Slobogean BL, Mulpuri K. MEDLINE, EMBASE, and Cochrane index most primary studies but not abstracts included in orthopedic meta-analyses. J Clin Epidemiol 2009;62:1261–7.
37. Primo E, Vázquez M, García-Sicilia J. Comparative Study of Journal Selection Criteria Used by MEDLINE and EMBASE, and Their Application to Spanish Biomedical Journals. The 9th European
Conference of Medical and Health Libraries, September 20–25, 2004, Santander, Spain.