Date 2015/December/5
Type of manuscript: Original article
New manuscript title: Atorvastatin use associated with acute pancreatitis: a
case-control study in Taiwan
Old manuscript title: Current use of atorvastatin correlates with increased
risk of acute pancreatitis: a case-control study in Taiwan
Running head: atorvastatin and acute pancreatitis Authors' full names:
Shih-Wei Lai MD 1,2, Cheng-Li Lin MS 1,3, Kuan-Fu Liao MD and MS 4,5,6 1College of Medicine, China Medical University and 2Department of Family
Medicine, China Medical University Hospital, Taichung, Taiwan
3Management Office for Health Data, China Medical University Hospital, Taichung,
Taiwan
4College of Medicine, Tzu Chi University, Hualien, Taiwan
5Department of Internal Medicine, Taichung Tzu Chi General Hospital, Taichung,
Taiwan
6Graduate Institute of Integrated Medicine, China Medical University
Corresponding author: Kuan-Fu Liao, Department of Internal Medicine, Taichung Tzu Chi General Hospital, No.66, Sec. 1, Fongsing Road, Tanzi District, Taichung City, 427, Taiwan
Phone: 886-4-2205-2121; Fax: 886-4-2203-3986 E-mail: kuanfuliaog@gmail.com
Word count: 295 in abstract, 2073 in text, 2 tables, and 28 references
list of abbreviations
˙1. ICD-9 code = International Classification of Diseases, 9th Revision, Clinical Modification
ABSTRACT
Background/Objective. Few data are present in literature on the relationship
between atorvastatin use and acute pancreatitis. The aim of this study was to explore this issue in Taiwan. Methods. Using representative claims data established from the Taiwan National Health Insurance Program, this case-control study consisted of 5810 cases aged 20-84 years with a first-time diagnosis of acute pancreatitis during the period 1998-2011and 5733 randomly selected controls without acute pancreatitis. Both cases and controls were matched by sex, age, comorbidities, and index year of diagnosing acute pancreatitis. Subjects who at least received 1 prescription for other statins or non-statin lipid-lowering drugs were excluded from the study. If subjects never had 1 prescription for atorvastatin, they were defined as never use of
atorvastatin. Current use of atorvastatin was defined as subjects whose last remaining one tablet of atorvastatin was noted ≤ 7 days before the date of diagnosing acute pancreatitis. Late use of atorvastatin was defined as subjects whose last remaining one tablet of atorvastatin was noted > 7 days before the date of diagnosing acute
pancreatitis. The odds ratio with 95% confidence interval of acute pancreatitis associated with atorvastatin use was calculated by using the logistic regression analysis. Results. The logistic regression analysis revealed that the odds ratio of acute pancreatitis was 1.67 for subjects with current use of atorvastatin (95% confidence interval 1.18, 2.38), when compared with subjects with never use of atorvastatin. The odds ratio decreased to 1.15 for those with late use of atorvastatin (95% confidence interval 0.87, 1.52), but without statistical significance. Conclusions. Current use of atorvastatin is associated with the diagnosis of acute pancreatitis. Clinically, clinicians should consider the possibility of atorvastatin-associated acute pancreatitis when patients present with a diagnosis of acute pancreatitis without a definite etiology but are taking atorvastatin.
Keywords: acute pancreatitis; atorvastatin; Taiwan National Health Insurance Program
INTRODUCTION
Atorvastatin is one of HMG-CoA reductase inhibitors, which are often known as statins and are commonly used to treat hypercholesterolemia with considerable effectiveness. Generally speaking, atorvastatin is a relatively safe and well tolerated statin with rare adverse effects including mild gastrointestinal disturbances, elevated serum transaminase levels and muscle-related disorders.1-3 However, few cases have
been reported that atorvastatin could be associated with acute pancreatitis as monotherapy or in combination with other offending drugs.4-9 The U.S. Food and
Drug Administration has also reported that since 1997 to 2013, 1183 persons (0.91%) had pancreatitis among 129845 persons reporting to have side effects when taking atorvastatin.10
Acute pancreatitis is known as an acute inflammatory condition of the pancreas with the potential for severe morbidity and mortality. The etiologies of acute pancreatitis have been extensively explored worldwide, such as alcohol consumption, biliary stones, hypertriglyceridemia and diabetes mellitus.11-13 In addition, many drugs have
been found to be associated with acute pancreatitis. Cases of drug-related acute pancreatitis are estimated to account for about 2% of acute pancreatitis cases.
Any adverse drug event needs further investigation based on a systematic analysis before a cause-effect relationship can be established. Given increased use of
atorvastatin and the aforementioned case reports and The U.S. Food and Drug Administration report, we make a plausible hypothesis that there could be a
relationship between atorvastatin use and acute pancreatitis. To our knowledge, the clinical relationship between atorvastatin use and acute pancreatitis using a
population-based study has not yet been explored. If the relationship really exists, atorvastatin-associated acute pancreatitis should be taken in consideration when
patients presenting with a diagnosis of acute pancreatitis without a definite etiology and concomitantly taking atorvastatin. Therefore, we used the claims data of the Taiwan National Health Insurance Program to explore the relationship between atorvastatin use only and acute pancreatitis.
METHODS Data source
The National Health Insurance Program was organized by the Taiwan government in 1995 as a nationwide, single-payer health insurance program. This program has enrolled about 99% of the entire population of 23 million people living in Taiwan.16
The National Health Insurance Research Database contained historical reimbursement claim data from the National Health Insurance Program. The details of the insurance program were well described in previous high-quality studies. Shortly speaking, The National Health Insurance Research Database was maintained and managed by National Health Research Institute. Before releasing claim data for research, the National Health Research Institute created a scrambled and anonymous identification number to link each insured individual reimbursement claim data, including sex, birth date and medical services record. The National Health Research Institute released the Longitudinal Health Insurance Database, which is a subset of the National Health Insurance Research Database. The Longitudinal Health Insurance Database contains all ambulatory and inpatient claims data on one million beneficiaries who were randomly sampled from the 2000 registry for beneficiaries of the National Health Insurance Research Database. For Personal Information Protection Act, all researchers must sign an agreement for no intention of attempting to obtain the privacy of
patients. The study was approved by the Institutional Review Board of China Medical University and Hospital in Taiwan (CMUH-104-REC2-115). Diseases were coded
according to the International Classification of Diseases 9th Revision Clinical
Modification (ICD-9 code), 2001 edition.
Study design and identification of cases and controls
This case-control study used data extracted from the Longitudinal Health Insurance Database. Subjects aged 20-84 years with a first-time diagnosis of acute pancreatitis during the period 1998-2011 were identified as cases (ICD-9 code 577.0). The index date for each case was defined as the date of diagnosing acute pancreatitis. Controls also aged 20-84 years were randomly selected from those without acute pancreatitis. Both cases and controls were matched by sex, age (every 5-year span), comorbidities, and index year of diagnosing acute pancreatitis. To increase unbiased results, subjects with chronic pancreatitis (ICD-9 code 577.1) or pancreatic cancer (ICD-9 code 157) before the date of diagnosing acute pancreatitis were excluded from the study. To focus on the relationship between atorvastatin use only and acute pancreatitis, subjects who at least received 1 prescription for other statins or non-statin lipid-lowering drugs were excluded from the study.
Comorbidities potentially related to acute pancreatitis
Comorbidities before the date of diagnosing acute pancreatitis potentially related to acute pancreatitis were included as follows: alcohol-related disease (ICD-9 codes 291, 303, 305.00, 305.01, 305.02, 305.03, 571.0-571.3, 790.3 and V11.3), biliary stone (ICD-9 code 574), cardiovascular disease including coronary artery disease, heart failure, cerebrovascular disease and peripheral atherosclerosis (ICD-9 codes 410–414, 428, 430–438 and 440–448), chronic kidney disease (ICD-9 codes 585–586 and 588.8–588.9), chronic obstructive pulmonary disease (ICD-9 codes 491, 492, 493 and 496), hepatitis B (ICD-9 codes V02.61, 070.20, 070.22, 070.30 and 070.32), hepatitis C (ICD-9 codes V02.62, 070.41, 070.44, 070.51 and 070.54), hyperparathyroidism (ICD-9 code 252.0), and hypertriglyceridemia (ICD-9 codes 272.1, 272.2 and 272.4).
Based on ICD-9 codes, the diagnosis accuracy of comorbidities included has been well assessed in previous studies.19-23
Definition of atorvastatin use
The mean elimination half-life of atorvastatin ranges from 11 to 24 hours in healthy people. Therefore, we used the period of 7 days as a cut-off point. If subjects never had 1 prescription for atorvastatin, they were defined as never use of atorvastatin. According to the prescription history, we can estimate the last remaining one tablet for atorvastatin. We adapted the definition of medication use from previous studies. Current use of atorvastatin was defined as subjects whose last remaining one tablet of atorvastatin was noted ≤ 7 days before the date of diagnosing acute pancreatitis or those still having atorvastatin tablets at the date of diagnosing acute pancreatitis. Late use of atorvastatin was defined as subjects whose last remaining one tablet of
atorvastatin was noted > 7 days before the date of diagnosing acute pancreatitis.
Statistical analysis
Cases and controls were compared for demographic status, atorvastatin use, and comorbidities using the Chi-square test and the Fisher-exact test for categorical variables and the t-test for continuous variables. Initially, all variables were included in the univariable unconditional logistic regression model to calculate the crude odds ratio (OR) and 95% confidence interval (CI) for the association between variables and acute pancreatitis. Then, significant variables found in the univariable analysis were further included in the multivariable unconditional logistic regression model. We used the SAS software for all data analysis (version 9.2 for Windows; SAS Institute Inc., Cary, North Carolina, USA). A P value < 0.05 was considered statistically significant.
RESULTS
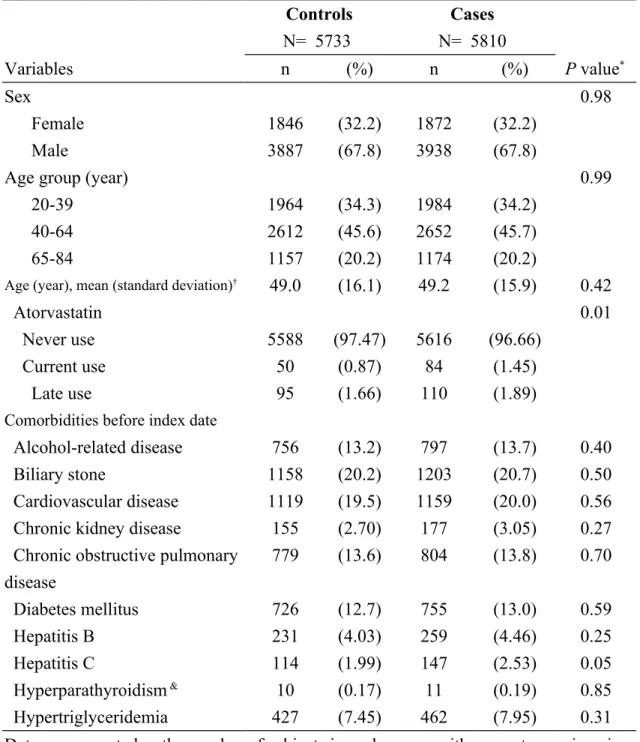
Distributions of demographic status, atorvastatin use, and comorbidities between cases of acute pancreatitis and controls during the period 1998-2011 are shown in Table 1. We identified 5810 cases of acute pancreatitis and 5733 controls with similar distributions of sex, age, and comorbidities. The cases were more likely to have higher proportions of current use of atorvastatin and late use of atorvastatin than the controls (1.45% vs. 0.87% for current use, and 1.89% vs. 1.66% for late use,
respectively, P = 0.01).
Atorvastatin use and risk of acute pancreatitis
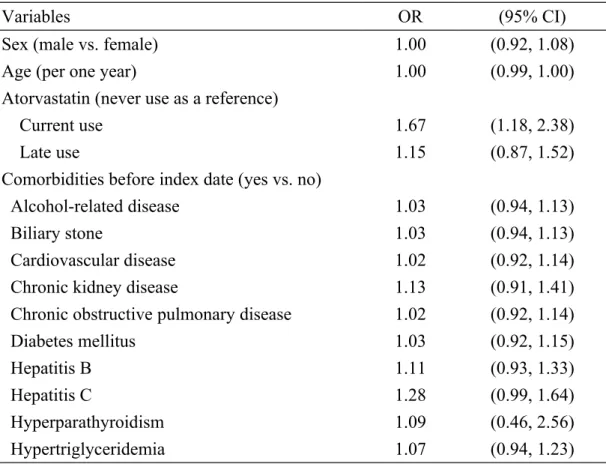
The univariable unconditional logistic regression model revealed that the odds ratio of acute pancreatitis was 1.67 for subjects with current use of atorvastatin (95% confidence interval 1.18, 2.38), when compared with subjects with never use of atorvastatin. The odds ratio decreased to 1.15 for those with late use of atorvastatin (95% confidence interval 0.87, 1.52), but without statistical significance (Table 2). Because no other variable was significantly related to acute pancreatitis in the univariable analysis, we did not perform the multivariable unconditional logistic regression model.
DISCUSSION
As well known, the etiologies of acute pancreatitis have been extensively explored
worldwide, such as alcohol consumption, biliary stones, hypertriglyceridemia and diabetes mellitus.11-13 In order to diminish the confounding effects from these
well-known risk factors, both cases and controls were well matched with comorbidities studied during the selection process of samples. Therefore, there was a similar distribution of comorbidities (Table 1). We could explore the relationship between atorvastatin use and acute pancreatitis without confounding. After matched with sex, age and comorbidities in cases and controls, we observed a modestly increased odds ratio of acute pancreatitis in patients with current use of atorvastatin as compared to
those with never use of atorvastatin (odds ratio 1.67, 95% confidence interval 1.18, 2.38), but patients with late use of atorvastatin did not have a significant association. This means that currently taking atorvastatin might be associated with acute
pancreatitis, independent of other comorbidities. Those who did not currently take atorvastatin might not have this association. Atorvastatin is a cholesterol-lowering agent, not treatment for gastro-intestinal diseases. Before conducting this study, no study revealed whether patients with acute pancreatitis might have higher proportion of taking atorvastatin. In this study, we observed that the cases of acute pancreatitis were more likely to have higher proportions of current use of atorvastatin and late use of atorvastatin than the controls (1.45% vs. 0.87% for current use, and 1.89% vs. 1.66% for late use, respectively, Table 1).
Although the exact mechanism of atorvastatin-associated acute pancreatitis cannot be fully clarified in this observational study, the direct toxic effect or idiosyncratic reaction of atorvastatin on the pancreas can partially explain this relationship because a dose-response effect was not found (data not shown). That is, patients with
atorvastatin-associated acute pancreatitis may be allergic or hypersensitive to this drug. Another possible mechanism can be partially explained by drug-drug interactions between atorvastatin and other concomitantly used drugs, which are mainly mediated by cytochrome P450 3A4 isoenzyme inhibition. That is, the
potentially toxic effect of atorvastatin on the pancreas is expanded by concomitantly used drugs which might inhibit cytochrome P450 3A4 isoenzyme.
To date, few cases have been reported that atorvastatin could be associated with acute pancreatitis as monotherapy or in combination with other offending drugs,4-9 but
no case received re-challenge test to confirm a cause-effect relationship. Similarly, the report of adverse drug reaction regarding atorvastatin-related pancreatitis published by
the U.S. Food and Drug Administration did not confirm a cause-effect relationship.10
In our study, in order to reduce the biased results, subjects who at least received 1 prescription for other statins or non-statin lipid-lowering drugs were excluded from the study. Therefore, this study was to focus on atorvastatin use only and association of acute pancreatitis. In spite of a case-control study, our results were less confounded by other lipid-lowering drugs or comorbidities. To date, no other
pharmacoepidemiological study focusing on atorvastatin use only and association of acute pancreatitis is available to be compared with. Further prospective trials are required to elucidate the precise relationship between atorvastatin use and acute pancreatitis.
Some important limitations should be considered in this study. First, theoretically, we should compare the group with atorvastatin use only and the group without using any drug. It is relatively difficult to design a case-control study using such an
inclusion criteria. Second, many drugs have occasional case reports of acute pancreatitis occurring in association. Theoretically, all prescribed medications
including atorvastatin should be corrected for analysis. In fact, it is relatively difficult to include all prescribed medications for analysis. Third, not knowing whether
patients did take it or not, we used atorvastatin prescriptions for instead. Fourth, the etiologies of acute pancreatitis were not recorded due to the inherent limitation of this database. It was very hard to estimate how many patients with idiopathic acute
pancreatitis could be substantially related to atorvastatin use but not detected clinically. Fifth, whether the association between atorvastatin use and acute pancreatitis could be a class effect with HMG-CoA reductase inhibitors cannot be determined in this study focusing only on atorvastatin, unless other statins and their possible association with acute pancreatitis are clarified. Sixth, a cause-effect
relationship between atorvastatin use and acute pancreatitis cannot be completely established by our observational study. More prospective studies are needed to establish whether atorvastatin use is a risk factor for acute pancreatitis.
Some strengths of this study should be considered. This topic is of great importance and intriguing, due to the worldwide use of this medication. We utilized the national healthcare database of Taiwan, which encompasses 99% of the whole population living in Taiwan, 16 to perform a well-matched case-control study to explore the
association of atorvastatin use with acute pancreatitis. The statistical methodology is excellent and the conclusions appear straightforward. It provides the update
information for further research.
We conclude that current use of atorvastatin is associated with the diagnosis of acute pancreatitis, independent of other comorbidities. Clinically, clinicians should consider the possibility of atorvastatin-associated acute pancreatitis when patients present with a diagnosis of acute pancreatitis without a definite etiology but are taking atorvastatin.
Acknowledgement
This study is supported in part by Taiwan Ministry of Health and Welfare Clinical Trial and Research Center of Excellence (MOHW105-TDU-B-212-133019). This funding agency did not influence the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Specific author contributions
Shih-Wei Lai planned and conducted this study. He substantially contributed to the conception of the article. He initiated the draft of the article and critically revised the article.
Cheng-Li Lin conducted the data analysis and critically revised the article. Kuan-Fu Liao planned and conducted this study. He participated in the data interpretation and also critically revised the article.
Conflict of Interest Statement
REFERENCES
1. Malinowski JM. Atorvastatin: a hydroxymethylglutaryl-coenzyme A reductase inhibitor. Am J Health Syst Pharm 1998;55:2253-67; quiz 302-3.
2. Bernini F, Poli A, Paoletti R. Safety of HMG-CoA reductase inhibitors: focus on atorvastatin. Cardiovasc Drugs Ther 2001;15:211-8.
3. Athyros VG, Tziomalos K, Karagiannis A, Mikhailidis DP. Atorvastatin: safety and tolerability. Expert Opin Drug Saf 2010;9:667-74.
4. Belaiche G, Ley G, Slama JL. [Acute pancreatitis associated with atorvastatine therapy]. Gastroenterol Clin Biol 2000;24:471-2.
5. Miltiadous G, Anthopoulou A, Elisaf M. Acute pancreatitis possibly associated with combined salicylate and atorvastatin therapy. JOP 2003;4:20-1.
6. Singh S, Nautiyal A, Dolan JG. Recurrent acute pancreatitis possibly induced by atorvastatin and rosuvastatin. Is statin induced pancreatitis a class effect? JOP 2004;5:502-4.
7. Kanbay M, Sekuk H, Yilmaz U, Gur G, Boyacioglu S. Acute pancreatitis associated with combined lisinopril and atorvastatin therapy. Dig Dis 2005;23:92-4.
8. Prajapati S, Shah S, Desai C, Desai M, Dikshit RK. Atorvastatin-induced pancreatitis. Indian J Pharmacol 2010;42:324-5.
9. Deshpande PR, Khera K, Thunga G, Hande M, Gouda ST, Nagappa AN. Atorvastatin-induced acute pancreatitis. J Pharmacol Pharmacother 2011;2:40-2. 10. eHealthMe study from FDA and social media reports. Review: could atorvastatin cause pancreatitis? http://www.ehealthme.com/print/ds15955104 [cited in 2015 January 1].
11. Gan SI, Edwards AL, Symonds CJ, Beck PL. Hypertriglyceridemia-induced pancreatitis: A case-based review. World J Gastroenterol 2006;12:7197-202.
12. Lai SW, Muo CH, Liao KF, Sung FC, Chen PC. Risk of acute pancreatitis in type 2 diabetes and risk reduction on anti-diabetic drugs: a population-based cohort study in Taiwan. Am J Gastroenterol 2011;106:1697-704.
13. Vidarsdottir H, Moller PH, Thorarinsdottir H, Bjornsson ES. Acute pancreatitis: a prospective study on incidence, etiology, and outcome. Eur J Gastroenterol Hepatol 2013;25:1068-75.
14. Wilmink T, Frick TW. Drug-induced pancreatitis. Drug Saf 1996;14:406-23. 15. Ksiadzyna D. Drug-induced acute pancreatitis related to medications commonly used in gastroenterology. Eur J Intern Med 2011;22:20-5.
16. National Health Insurance Research Database. Taiwan.
17. Lai SW, Liao KF, Liao CC, Muo CH, Liu CS, Sung FC. Polypharmacy correlates with increased risk for hip fracture in the elderly: a population-based study. Medicine (Baltimore) 2010;89:295-9.
18. Hung SC, Liao KF, Lai SW, Li CI, Chen WC. Risk factors associated with
symptomatic cholelithiasis in Taiwan: a population-based study. BMC Gastroenterol 2011;11:111.
19. Liao KF, Lai SW, Li CI, Chen WC. Diabetes mellitus correlates with increased risk of pancreatic cancer: a population-based cohort study in Taiwan. J Gastroenterol Hepatol 2012;27:709-13.
20. Lai SW, Liao KF, Muo CH, Hsieh DPH. No Association between Statin Use and Pancreatic Cancer Risk in Taiwan. Kuwait Medical Journal 2013;45:251-2.
21. Lai SW, Liao KF, Lin CL, Chen PC. Pyogenic liver abscess correlates with increased risk of acute pancreatitis: a population-based cohort study. J Epidemiol 2015;25:246-53.
22. Lai SW, Lai HC, Lin CL, Liao KF, Tseng CH. Chronic osteomyelitis correlates with increased risk of acute pancreatitis in a case-control study in Taiwan. Eur J Intern Med 2015;26:429-32.
23. Liao K-F, Lin C-L, Lai S-W, Chen W-C. Parkinson's disease and risk of pancreatic cancer: a population-based case-control study in Taiwan. Neurology Asia
2015;20:251-5.
24. Cilla DD, Jr., Whitfield LR, Gibson DM, Sedman AJ, Posvar EL. Multiple-dose pharmacokinetics, pharmacodynamics, and safety of atorvastatin, an inhibitor of HMG-CoA reductase, in healthy subjects. Clin Pharmacol Ther 1996;60:687-95. 25. Poli A. Atorvastatin: pharmacological characteristics and lipid-lowering effects. Drugs 2007;67 Suppl 1:3-15.
26. Liao KF, Lin CL, Lai SW, Chen WC. Zolpidem Use Associated With Increased Risk of Pyogenic Liver Abscess: A Case-Control Study in Taiwan. Medicine (Baltimore) 2015;94:e1302.
27. Liao KF, Lin CL, Lai SW, Chen WC. Sitagliptin use and risk of acute pancreatitis in type 2 diabetes mellitus: A population-based case-control study in Taiwan. Eur J Intern Med 2016;27:76-79.
28. Lennernas H. Clinical pharmacokinetics of atorvastatin. Clin Pharmacokinet 2003;42:1141-60.
Table 1. Descriptive characteristics of cases with acute pancreatitis and control subjects in Taiwan during the period 1998-2011
Controls N= 5733 Cases N= 5810 Variables n (%) n (%) P value* Sex 0.98 Female 1846 (32.2) 1872 (32.2) Male 3887 (67.8) 3938 (67.8)
Age group (year) 0.99
20-39 1964 (34.3) 1984 (34.2)
40-64 2612 (45.6) 2652 (45.7)
65-84 1157 (20.2) 1174 (20.2)
Age (year), mean (standard deviation)† 49.0 (16.1) 49.2 (15.9) 0.42
Atorvastatin 0.01
Never use 5588 (97.47) 5616 (96.66)
Current use 50 (0.87) 84 (1.45)
Late use 95 (1.66) 110 (1.89)
Comorbidities before index date
Alcohol-related disease 756 (13.2) 797 (13.7) 0.40
Biliary stone 1158 (20.2) 1203 (20.7) 0.50
Cardiovascular disease 1119 (19.5) 1159 (20.0) 0.56
Chronic kidney disease 155 (2.70) 177 (3.05) 0.27
Chronic obstructive pulmonary disease 779 (13.6) 804 (13.8) 0.70 Diabetes mellitus 726 (12.7) 755 (13.0) 0.59 Hepatitis B 231 (4.03) 259 (4.46) 0.25 Hepatitis C 114 (1.99) 147 (2.53) 0.05 Hyperparathyroidism & 10 (0.17) 11 (0.19) 0.85 Hypertriglyceridemia 427 (7.45) 462 (7.95) 0.31
Data are presented as the number of subjects in each group, with percentages given in parentheses, or mean with standard deviation given in parentheses.
*Chi-square test, &Fisher-exact test, and †t-test comparing subjects with and without
Table 2. Odds ratio and 95% confidence interval of acute pancreatitis
associated with atorvastatin use and other comorbidities in Taiwan during the period 1998-2011
Variables OR (95% CI)
Sex (male vs. female) 1.00 (0.92, 1.08)
Age (per one year) 1.00 (0.99, 1.00)
Atorvastatin (never use as a reference)
Current use 1.67 (1.18, 2.38)
Late use 1.15 (0.87, 1.52)
Comorbidities before index date (yes vs. no)
Alcohol-related disease 1.03 (0.94, 1.13)
Biliary stone 1.03 (0.94, 1.13)
Cardiovascular disease 1.02 (0.92, 1.14)
Chronic kidney disease 1.13 (0.91, 1.41)
Chronic obstructive pulmonary disease 1.02 (0.92, 1.14)
Diabetes mellitus 1.03 (0.92, 1.15)
Hepatitis B 1.11 (0.93, 1.33)
Hepatitis C 1.28 (0.99, 1.64)
Hyperparathyroidism 1.09 (0.46, 2.56)
Hypertriglyceridemia 1.07 (0.94, 1.23)