From the Department of Emergency Medicine, Department of Orthopedics, and the College of Public Health, National Taiwan University; National Taiwan University Hospital; and National Taiwan University College of Medicine, Taipei, Taiwan.
Supported by the Bureau of National Health Insurance, Taiwan (grant number DOH89-NH-058).
Z-S. Yen, MD, MPH, Department of Emergency Medicine; M-S. Lai, MD, PhD, College of Public Health, National Taiwan University; C-T. Wang, MD, Department of Orthopedics; L-S. Chen, BA, Department of Administration, National Taiwan University Hospital; S-C. Chen, MD; W-J. Chen, MD, PhD, Department of Emergency Medicine; S-M. Hou, MD, PhD, Department of Orthopedics.
Address reprint requests to Dr. S-M. Hou, Department of Orthopedics, National Taiwan University Hospital, No.7 Chung-Shan South Road, Taipei 100, Taiwan. E-mail: zuishen@ha.mc.ntu.edu.tw
Submitted July 11, 2003; revision accepted March 15, 2004.
Osteoarthritis (OA) is a common disease characterized by
progressive deterioration and loss of articular cartilage,
subchondral sclerosis, and osteophyte formation. In the
Framingham Osteoarthritis Study, one-third of the patients
aged > 60 years had radiographic evidence of OA of the
knee
1. The prevalence rate of symptomatic OA in Taiwan is
5.8%, and one-third of cases of OA involved the knee
2.
Current nonoperative treatment of OA is usually focused
on reduction of pain, for which nonsteroidal
antiinflamma-tory drugs (NSAID) are frequently used (e.g., naproxen).
NSAID exhibit both analgesic and antiinflammatory effects
but can also cause frequent and serious adverse effects in the
elderly
3,4. It is estimated that the annual rate of
gastroin-testinal (GI) complications resulting in hospitalization and
death among patients with OA is 0.73% and 0.11%,
respec-tively
5. Celecoxib, a cyclooxygenase-2 (COX-2) inhibitor,
has been shown to be effective in treating OA and to reduce
GI tract toxic effects compared with other nonselective
NSAID
6-8. However, the safety advantage of celecoxib
comes at the expense of higher drug costs.
Hyaluronan (HA) is a natural constituent of joint fluid and
all connective tissue. Recent studies using intraarticular
injections of HA for OA of the knee have shown a beneficial
effect on knee pain and function without serious adverse
effects
9-13. Questions remain about whether the effectiveness
of celecoxib and HA injections justifies their higher costs.
The goal of our study was to consider how factors such
as costs, improvement rates, side effects, and quality of life
might affect drug choices for the treatment of OA of the
knee in Taiwan. We therefore developed a decision model to
estimate the cost-effectiveness of alternative treatment
strategies for OA of the knee. The purpose of this study was
to determine the relative merits of each strategy in terms of
societal costs and of quality-adjusted life.
Cost-Effectiveness of Treatment Strategies for
Osteoarthritis of the Knee in Taiwan
ZUI-SHEN YEN, MEI-SHU LAI, CHEN-TI WANG, LI-SHU CHEN, SHYR-CHYR CHEN, WEN-JONE CHEN,
and SHENG-MOU HOU
ABSTRACT. Objective. To evaluate the cost-effectiveness of 3 treatment strategies for osteoarthritis (OA) of the knee: naproxen, celecoxib, and hyaluronan.
Methods. We developed a decision model to estimate the costs and effectiveness of 3 treatment strategies: 250 mg naproxen 3 times daily for 26 weeks, 100 mg celecoxib twice daily for 26 weeks, and 25 mg hyaluronan by intraarticular injection once per week for 5 weeks followed by conven-tional treatment for 21 weeks. The probabilities and utility data were obtained by surveying the liter-ature and consulting experts. Cost data were obtained from insurance reimbursement data of National Taiwan University Hospital and were converted to 2002 US dollars. The timeframe of the decision tree was 26 weeks. Outcomes were expressed in aggregated costs, quality-adjusted life-years (QALY), and the incremental cost-effectiveness ratio (ICER). Sensitivity analyses were performed on most variables.
Results. The expected total costs for the naproxen, celecoxib, and hyaluronan strategies were US$498.98, US$547.80, and US$678.00, respectively. The ICER of the celecoxib strategy compared with the naproxen strategy was US$21,226 per QALY gained. The ICER of the hyaluronan strategy versus the celecoxib strategy was US$42,000 per QALY gained. The ICER of the hyaluronan strategy decreased to about US$25,000 per QALY gained if the weekly treatment cost of hyaluronan was decreased to US$31.
Conclusion. Celecoxib treatment results in a reasonable cost-effectiveness ratio for patients with OA of the knee. Hyaluronan treatment, however, may not be an economically attractive choice under the current healthcare scenario in Taiwan. (J Rheumatol 2004;31:1797–803)
Key Indexing Terms:
COST-BENEFIT ANALYSIS OSTEOARTHRITIS
MATERIALS AND METHODS
Study design
This study was conducted utilizing common principles of cost-effective-ness analysis14. Three treatment strategies for the management of OA were
compared: (1) 250 mg naproxen 3 times daily for 26 weeks, (2) 100 mg celecoxib twice daily for 26 weeks, and (3) 25 mg HA (Artz®, Seikagaku
Corp., Tokyo, Japan) by intraarticular injection once per week for 5 weeks followed by conventional treatment for 21 weeks. A base-case scenario and sensitivity analyses were simulated in the study.
Study setting and population
Base case. The base case was a hypothetical 60-year-old woman who had
symptomatic and radiologically verified OA of the right knee. She was working fulltime. Her knee pain after a 50-ft walk was 50 mm on 100 mm visual analog scale (VAS). Global assessment of her right knee on a scale from 1 (very poor) to 5 (very good) was 2 (poor). She declined surgical intervention. Three treatment strategies (naproxen, celecoxib, and HA) were available and one of the therapeutic options had to be chosen. Based on the related literature, the probability of improvement of her knee OA was 0.31 with naproxen7,10,15, 0.35 with celecoxib6,7, and 0.36 with HA9-13.
The probability of serious GI complications in 26 weeks was 0.0037 with naproxen3-5,16and 0.0000075 with celecoxib6,7. The probability of local
injection pain from HA was 0.0219-13.
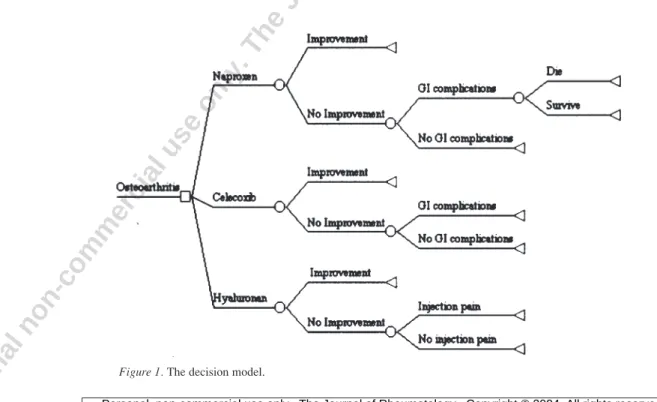
Decision model. We developed a decision model (Figure 1) to simulate
possible outcomes of OA of the knee using treatment with naproxen, cele-coxib, or HA. We identified the following clinical outcomes: OA with or without improvement, OA with GI complications, and OA with local injec-tion pain from HA treatment. OA with improvement meant that a patient improved clinically (improvement of ≥ 20 mm on the VAS or of ≥ 2 grades on global assessment). Conventional treatments of OA in the outpatient setting may include nonpharmacologic therapies (e.g., patient education and weight loss), calcium supplements, muscle relaxants, topical anal-gesics, and acetaminophen.
Over-the-counter medication is not covered by Taiwan’s compulsory National Health Insurance (NHI) and was not included in the nonpharma-cologic therapies. No NSAID, including naproxen, celecoxib, and HA, was considered conventional treatment. Serious GI complications from naproxen or celecoxib were defined as serious adverse events requiring hospitalization. Common serious GI complications are bleeding and
perfo-rated ulcers of the upper or lower GI tract. Patients who have GI complica-tions resulting from naproxen are at risk of mortality during their hospital-ization. Patients with minor GI complications such as dyspepsia were treated in an outpatient setting. Except during the period when receiving HA injections and hospitalization, patients were followed up at the outpa-tient department at 2-week intervals. A timeframe of 26 weeks was used in the model.
Probabilities. Estimates of probabilities (Table 1) used in the model were
obtained by surveying the literature and consulting experts. For each vari-able, a base-case estimate was determined if the study was considered authoritative by the authors or most similar to our clinical practice. For example, in Altman’s trial10, 59 of 164 patients who received HA injections
improved clinically. So the improvement probability of the HA strategy was 0.36. We estimated the effectiveness of naproxen, celecoxib, and HA by using the percentage improvement without subtracting the placebo effect. To represent the degree of uncertainty, a plausible range of each esti-mate was also determined using other studies of similar topics that were not considered authoritative. These ranges of estimates were further used in the sensitivity analyses.
Costs. From a societal perspective, cost data (Table 2) were collected,
including costs of outpatient treatments, inpatient treatments for serious GI complications, and time lost from work. Cost data for each treatment strategy represented the average reimbursement from the NHI received by National Taiwan University Hospital from July 2001 to February 2002. These costs (in New Taiwan dollars, NT$) were then converted to 2002 US dollars at the rate of NT$34.96 to US$117.
Three hundred OA patients were selected randomly from the database, and their reimbursements from the NHI were averaged to reflect real soci-etal costs. Weekly costs of each treatment included actual costs of medica-tions, physician fees, and administrative fees (e.g., fees to administer outpatient visits). For example, our estimate of the weekly cost of a single HA injection was US$41.84, which includes the cost of HA, physician fees, and administrative fees, and was the average reimbursement to a patient who received one HA injection in the outpatient department (OPD). Loss of productivity, i.e., time lost from work, was calculated using the average Taiwanese industrial wage of US$239.37/week in 200317. The cost
associ-ated with each clinical outcome was determined by aggregating the various elements of the strategy. For example, the total cost for a patient with clin-ical improvement from 5 HA injections was computed as follows: (5 × the
weekly cost of HA) + (21 × the weekly cost of conventional treatment of OA) + [15.5 (number of OPD visits) × 0.5 (0.5 days/OPD visit) × the weekly wage loss/7 (daily wage loss due to an OPD visit)]. Costs were not discounted in our analysis because of the short timeframe (26 weeks).
Quality of life. Estimates of quality-of-life adjustments for various clinical
outcomes in the model for patients in Taiwan were unknown prior to this study, but were required to perform a decision analysis that includes patient preferences. Since no existing data were available on the health utilities relating to the issue of OA, a panel of experts was used. This panel, composed of 2 orthopedic physicians, 2 emergency physicians, and one internist with extensive experience in the treatment of knee OA, agreed to
participate in a standard-gamble procedure. They assessed their own utili-ties for each health status by using the standard reference gamble tech-nique18(Table 1). The quality-adjusted life-year (QALY) associated with
each clinical outcome during the 26 weeks (timeframe) was determined by aggregating the various time periods of health status multiplied by their quality-of-life adjustments (utilities).
Assumptions in the model. We used various assumptions in this model.
First, only patients with serious GI complications were admitted. Patients with minor GI complications were treated as outpatients. Second, if patients failed to improve after receiving therapies, they could not change to other treatment strategies. For example, if patients received naproxen
Table 1. Estimates of variables used in the decision model.
Variable Base-Case Estimate Plausible Range References
Probability of improvement
Naproxen 0.31 0.2~0.6 7, 10, 15
Celecoxib 0.35 0.2~0.6 6, 7
Hyaluronan 0.36 0.2~0.6 9–13
Probability of serious GI complications
from naproxen treatment 0.0037 0.001~0.05 3–5, 16
Probability of mortality from serious GI
complications 0.0006 0.0001~0.006 4, 5, 16
Probability of serious GI complications
from celecoxib treatment 0.0000075 0~0.0001 8
Injection pain from hyaluronan treatment 0.036 0.01~0.10 9–13
Quality-of-life adjustments
OA with improvement from treatment
with naproxen, celecoxib, or hyaluronan 0.95 0.94~0.96 ¶
OA without improvement from treatment
with naproxen or celecoxib 0.84 0.83~0.85 ¶
OA with conventional treatment 0.85 0.84~0.86 ¶
OA with hyaluronan injections 0.84 0.83~0.85 ¶
OA with injection pain from hyaluronan
treatment 0.83 0.82~0.84 ¶
Serious GI complications 0.75 0.74~0.76 ¶
¶ Based on an assessment by an expert panel. GI: gastrointestinal; OA: osteoarthritis.
Table 2. Estimated OA-related costs. All values are in 2002 US dollars. Ranges for cost estimates represent 30%
of the baseline estimate.
Variable Base-Case Cost, US$ Range, US$ Source
Weekly OPD treatment cost
Naproxen 10.48 7.34~13.62 Reimbursement¶
Celecoxib 12.52 8.76~16.28 Reimbursement¶
Hyaluronan 41.84 29.29~54.39 Reimbursement¶
Conventional treatment 9.88 6.92~12.84 Reimbursement¶
GI complications 14.54 10.18~18.90 Reimbursement¶
Weekly inpatient treatment cost
Serious GI complications 423.31 296.32~550.30 Reimbursement¶
Weekly time lost from work 239.37 167.56~311.18 §
Unit cost of medication
Naproxen 250 mg 0.16 – Reimbursement¶
Celecoxib 100 mg 0.48 – Reimbursement¶
Hyaluronan 25 mg 34.58 – Reimbursement¶
Administrative fee per OPD visit 1.52 – Reimbursement¶
Physician fee per OPD visit 5.56 – Reimbursement¶
¶Average reimbursement of the National Health Insurance to National Taiwan University Hospital from July
2001 to February 2002. §Average industrial wage rate in 2002 in Taiwan. OA: osteoarthritis; OPD: outpatient
treatment but failed to improve, they could not change to celecoxib or HA therapies. If patients suffered from serious GI complications, they were assigned to the “OA without improvement” group and received conven-tional therapies for OA and outpatient treatments for GI complications after discharge. We also assumed that there were no mortality events, except death from GI complications. When performing the sensitivity analyses, changes in the weekly costs of the 3 treatment strategies were assumed to affect only the costs of naproxen, celecoxib, or HA, while the costs of physician fees, administrative fees, and other medications remained constant.
Measurements and outcome variables
The outcome measurements for this analysis were aggregate costs, QALY, and incremental cost per QALY. These outcome variables were calculated for all 3 treatment strategies. The expected total costs and the effectiveness of the QALY were calculated according to our decision model simulation. Data analysis
Cost-effectiveness was tabulated. Because of variations in published data, costs, and varying responses to treatments, we also performed sensitivity analyses on most variables used in the decision model to see how changing these estimates over a wide range affected selection of the optimal strategy. A computer program (Data 3.5 of TreeAge) was used for all calculations.
RESULTS
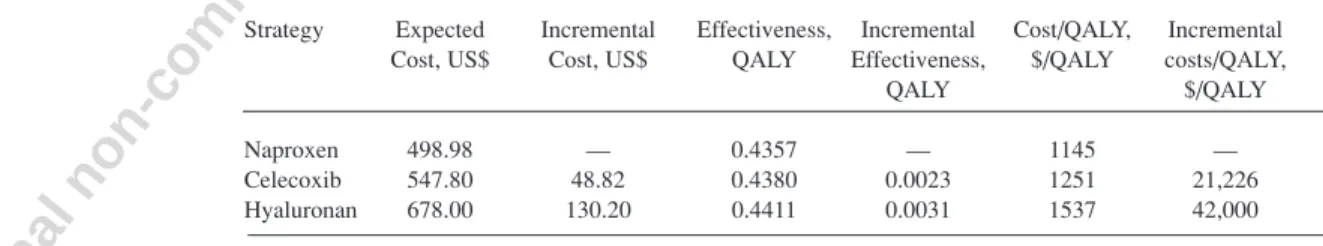
Base-case analysis. According to the simulation results, on
average, when a 60-year-old woman with OA of the right
knee was treated with naproxen, the expected total cost was
US$498.98 and the expected effectiveness was 0.4357
QALY. On the other hand, if treated with celecoxib or HA,
the expected total cost increased to US$547.80 or
US$678.00, respectively. Values for the expected
effective-ness of the celecoxib and HA strategies were 0.4380 and
0.4411 QALY, respectively (Table 3). The incremental
cost-effectiveness ratio (ICER) of the celecoxib strategy
compared with naproxen was US$21,226 per QALY gained.
The ICER of the HA strategy versus celecoxib was
US$42,000 per QALY gained.
Sensitivity analysis. One-way sensitivity analyses identified
several influential variables (Figures 2 and 3). Although the
probability of serious GI complications from naproxen
treat-ment was estimated from a large epidemiology study
5, we
increased this probability by 10 times that of the base-case
estimate to evaluate the effect of changing this probability
on the ICER of the celecoxib strategy as compared with the
naproxen strategy (Figure 2). The ICER of the celecoxib
strategy decreased to about US$3170 per QALY gained if
the probability of serious GI complications from naproxen
treatment were increased to 0.037. If the probability of
serious GI complications from naproxen treatment were
increased to > 0.0464, the celecoxib strategy would offer
patients a better quality of life, save societal resources, and
become a dominant strategy compared with naproxen.
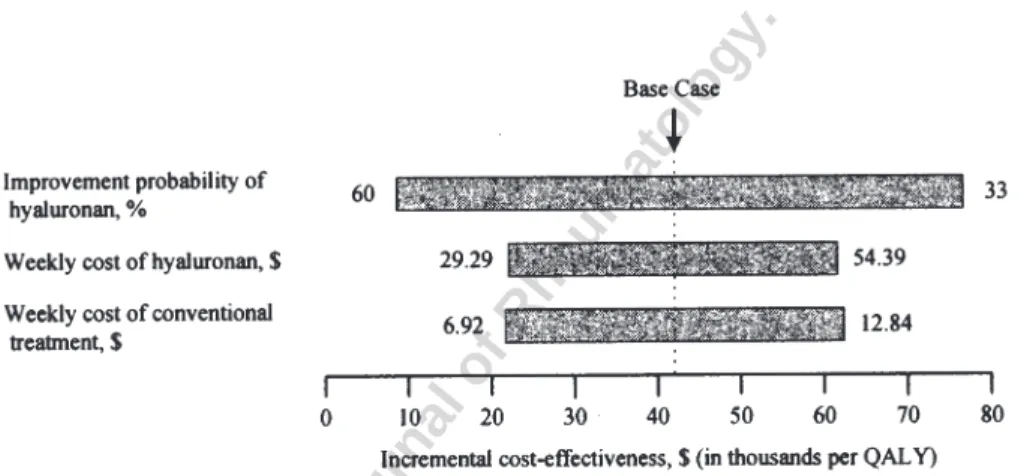
Not surprisingly, the improvement probability of HA
treatment had a potentially notable impact on the
cost-effec-tiveness of the HA strategy as compared with the celecoxib
strategy (Figure 3). When the improvement probability of
HA treatment was increased to 0.60, the ICER of the HA
strategy decreased to about US$8900 per QALY gained.
However, as the improvement probability of HA treatment
was decreased to between 0.36 and 0.297, the ICER of the
HA strategy increased far beyond the US$42,000 per QALY
gained, the base-case result. If the improvement probability
of HA treatment was decreased to < 0.297, the HA strategy
would offer patients a worse quality of life, but would still
cost more compared with celecoxib. Therefore, HA
treat-ment would be dominated by celecoxib.
The weekly treatment costs of HA and conventional
treatment had significant effects on the ICER of the HA
strategy as compared with the celecoxib strategy. The ICER
of the HA strategy decreased to about US$25,000 per QALY
gained if the weekly treatment cost of HA was decreased to
US$31 (Figure 3). If the weekly cost of conventional
treat-ment was decreased to US$7, the ICER of the HA strategy
decreased to about US$22,400 per QALY gained.
The cost of time lost from work did not have a significant
influence on the ICER. The results of base-case analysis
were insensitive to costs of outpatient and inpatient
treat-ment of GI complications.
DISCUSSION
Considering the limited resources available for healthcare, it
is important to consider the impact of incorporating new
technologies that affect patient outcomes and healthcare
expenditures. Using results from available studies and cost
data, we developed a decision-making model to simulate
possible clinical outcomes. Our results suggest that
naproxen and celecoxib treatments result in reasonable
cost-effectiveness ratios. However, the ICER of the HA strategy
was 3.3 times the 2002 Taiwan gross domestic product per
capita (US$12,588)
19and was also more than the suggested
Table 3. Base-case results. Costs are in 2002 US dollars.
Strategy Expected Incremental Effectiveness, Incremental Cost/QALY, Incremental Cost, US$ Cost, US$ QALY Effectiveness, $/QALY costs/QALY,
QALY $/QALY
Naproxen 498.98 — 0.4357 — 1145 —
Celecoxib 547.80 48.82 0.4380 0.0023 1251 21,226
Hyaluronan 678.00 130.20 0.4411 0.0031 1537 42,000
cost-effectiveness threshold, US$32,000 per QALY
(CDN$1.57:US$1), for Canada and the United States
20,21.
Taiwan has fewer health resources than Canada and the US;
therefore, adopting HA treatment for treatment of OA of the
knee may not be cost-effective in Taiwan.
The probability for improvement with HA treatment has
varied. In most randomized clinical trials, the improvement
probabilities were about 0.36
9–12. In a recent open-label
study, however, the improvement probability was 69%
13,
and the ICER of HA treatment was US$6369 per QALY
gained compared with appropriate care
22. We performed an
analysis using their improvement probability of 69% and
naproxen treatment as appropriate care. We found no large
difference from Torrance, et al’s analysis
22, as the ICER of
HA strategy would be US$8321 per QALY gained.
However, based on our clinical experience with HA
treat-ment, the high improvement probability did not seem
applicable to our patients, and therefore we decided not to
adopt it for our analysis.
Studies have shown similar efficacies of COX-2
inhibitors and naproxen for the treatment of OA
7,23,24. In our
analysis, celecoxib was found to be a more cost-effective or
even cost-saving strategy if the probability of serious GI
complications from traditional NSAID treatments was
increased. In elderly patients and patients at high risk of GI
complications, the probability of serious GI complications
could be > 0.0037. In those cases, the ICER of celecoxib
treatment was < US$21,226 per QALY gained, and
cele-coxib treatment became even more attractive. In terms of
cost-effectiveness, the results of our analyses led us to
Figure 2. One-way sensitivity analyses of the incremental cost-effectiveness of celecoxib treatmentcompared with naproxen treatment. Bars indicate the variability of the incremental cost-effectiveness ratio (x-axis) caused by changes in the value of the indicated variable, with all other variables being held constant. Labels on the horizontal bars indicate a certain range of each one-way sensitivity analysis. The range levels in this figure only indicate partial results of our one-way sensitivity analyses. Costs are in 2002 US dollars. QALY: quality-adjusted life-year.
Figure 3. One-way sensitivity analyses of the incremental cost-effectiveness of hyaluronan treatment
compared with celecoxib treatment. Bars indicate the variability of the incremental cost-effectiveness ratio (x-axis) caused by changes in the value of the indicated variable, with all other variables being held constant. Labels on the horizontal bars indicate a certain range of each one-way sensitivity analysis. The range levels in this figure only indicate partial results of our one-way sensitivity analyses. Costs are in 2002 US dollars. QALY: quality-adjusted life-year.
recommend COX-2 inhibitors for elderly patients and
patients at high risk of GI bleeding.
New medical treatments, such as HA, are often more
effective, but also more costly than traditional ones. Some
health interventions such as use of a tissue plasminogen
activator for acute ischemic stroke
25or norfloxacin for acute
uncomplicated pyelonephritis
26may appear very costly at
first, but have been proven to be cost-saving strategies in
terms of cost-effectiveness. Nevertheless, our results
suggest that the incremental effectiveness of HA treatment
might not justify its higher costs. If the weekly cost of HA
treatment could be considerably decreased, then HA would
become more attractive. In our analyses, HA treatment
would become a cost-saving strategy only if its weekly cost
fell below US$15.32.
We used an average weekly-cost approach instead of
simply adding up the components to estimate the weekly
costs, because the average weekly-cost approach produced
values closer to actual costs. Simply adding up components
may underestimate the true costs. For example, for patients
treated with celecoxib, in addition to the cost of celecoxib,
the physician fee, and the administrative fee, there were
costs for medications other than celecoxib. Those
medica-tions may include some that are relatively costly with
uncer-tain effects on OA, such as calcium supplements, antacids,
etc. The weekly cost of celecoxib ($12.52) was larger than
the total amount ($10.26) from simply adding up the cost of
celecoxib ($0.48
× 2 × 7 = $6.72), the physician fee ($5.56/2
= $2.78, with followup at 2-week intervals), and the
admin-istration fee ($1.52/2 = $0.76, with followup at 2-week
intervals). The cost of “extra” medications may be seen as a
kind of “externality” associated with the treatment strategy.
This phenomenon was most obvious for naproxen treatment
and least obvious for HA treatment among the 3 compared
strategies. We did not investigate this phenomenon closely
because it was beyond the main purpose of our study.
There were several limitations of our analysis. First, our
results might not be applicable to patients in areas where
medical financial structures and clinical practice patterns
differ markedly from those in Taiwan. Further studies are
needed to test the external validity of our analysis. Second,
our analysis was not done concurrently with any clinical
trial. The efficacy and cost data were collected from
different patient populations. We did our best to summarize
results from good-quality clinical trials and epidemiological
studies. And we also performed sensitivity analyses to
eval-uate the stability of our conclusions. We think our results are
still applicable to patients in Taiwan.
Longterm side effects of naproxen, celecoxib, and HA
were not included in our analysis. Current limited data
suggest that celecoxib is associated with fewer incidents of
longterm GI, renal, and cardiovascular toxicity than
tradi-tional NSAID
27,28. These features could make celecoxib
more favorable. We admit that our model did not cover all
possible outcomes and all possible real-world treatment
strategies. In the process, we had to balance between the
complexity and feasibility of the model. Therefore, we did
not include unlikely outcomes such as mortality events from
celecoxib or HA treatments. Ideally, the source of
quality-of-life measurements is obtained from community
prefer-ences. In our study, experts’ opinions were used instead due
to a lack of valid culture-specific and disease-specific
generic measurements in Taiwan. Future studies should
involve large-scale, multicenter prospective, randomized,
controlled analyses to assess the validity of the costs and
benefits estimated here.
With the advent of increasingly costly treatments with
greater effectiveness and fewer side effects, it is necessary
to perform cost-effectiveness analyses to justify their use,
especially when medical resources are limited. The results
of our analysis suggest that celecoxib treatment produces
reasonable cost-effectiveness ratios for patients with OA of
the knee. HA treatment, however, may not be an
economi-cally attractive choice under the current healthcare scenario
in Taiwan.
REFERENCES
1. Felson DT, Naimark A, Anderson J, Kazis L, Castelli W, Meenan RF. The prevalence of knee arthritis in the elderly: the Framingham Osteoarthritis Study. Arthritis Rheum 1987;30:914-8.
2. Chou CT, Pei L, Chang DM, Lee CF, Schumacher HR, Liang MH. Prevalence of rheumatic diseases in Taiwan: a population study of urban, suburban, rural differences. J Rheumatol 1994;21:302-6. 3. Langman MJ, Weil J, Wainwright P, et al. Risks of bleeding peptic
ulcer associated with individual non-steroidal anti-inflammatory drugs. Lancet 1994;343:1075-8.
4. Fries JF, Miller SR, Spitz PW, Williams CA, Hubert HB, Bloch DA. Toward an epidemiology of gastropathy associated with nonsteroidal antiinflammatory drug use. Gastroenterology 1989;96:647-55.
5. Singh G, Triadafilopoulos G. Epidemiology of NSAID induced gastrointestinal complications. J Rheumatol 1999;26:18-24. 6. Simon LS, Lanza FL, Lipsky PE, et al. Preliminary study of the
safety and efficacy of SC-58635, a novel cyclooxygenase 2 inhibitor: efficacy and safety in two placebo-controlled trials in osteoarthritis and rheumatoid arthritis, and studies of gastrointestinal and platelet effects. Arthritis Rheum 1998;41:1591-602.
7. Bensen WG, Fiechtner JJ, McMillen JI, et al. Treatment of osteoarthritis with celecoxib, a cyclooxygenase-2 inhibitor: a randomized controlled trial. Mayo Clin Proc 1999;74:1095-105. 8. Singh G, Ramey DR, Triadafilopoulos G. Early experience with selective COX-2 inhibitors: safety profile in over 340,000 patient years of use [abstract]. Arthritis Rheum 1999;42 Suppl:S296. 9. Brandt KD, Block JA, Michalski JP, et al. Efficacy and safety of
intraarticular sodium hyaluronate in knee osteoarthritis. Clin Orthop Rel Res 2000;385:130-43.
10. Altman RD, Moskowitz R. Intraarticular sodium hyaluronate (hyalgan) in the treatment of patients with osteoarthritis of the knee: a randomized clinical trial. The Hyalgan Study Group. J Rheumatol 1998;25:2203-12.
11. Adams ME, Lussier AJ, Peyron JG. A risk-benefit assessment of injections of hyaluronan and its derivatives in the treatment of osteoarthritis of the knee. Drug Saf 2000;23:115-30.
12. Lohmander LS, Dalen N, Englund G, et al. Intra-articular hyaluronan injections in the treatment of osteoarthritis of the knee: a randomized, double blind, placebo controlled multicentre trial. Ann Rheum Dis 1996;55:424-31.
13. Raynauld JP, Torrance GW, Band PA, et al. A prospective, randomized, pragmatic, health outcomes trial evaluating the incorporation of hylan G-F 20 into the treatment paradigm for patients with knee osteoarthritis (Part 1 of 2): clinical results. Osteoarthritis Cartilage 2002;10:506-17.
14. Ferraz MB, Maetzel A, Bombardier C. A summary of economic evaluations published in the field of rheumatology and related disciplines. Arthritis Rheum 1997;40:1587-93.
15. Williams HJ, Ward JR, Egger MJ, et al. Comparison of naproxen and acetaminophen in a two-year study of treatment of osteoarthritis of the knee. Arthritis Rheum 1993;36:1196-206. 16. MacDonald TM. Epidemiology and pharmacoeconomic
implications of non-steroidal anti-inflammatory drug-associated gastrointestinal toxicity. Rheumatology 2000;39:13-20.
17. Statistics Taiwan. Average industrial aggregate and foreign currency exchange rate. Taipei: Statistics Taiwan; 2003.
18. Torrance GW. Measurement of health state utilities for economic appraisal. J Health Econ 1986;5:1-30.
19. Statistics Taiwan. Gross domestic product per capita. Taipei: Statistics Taiwan; 2003.
20. Laupacis A, Feeny D, Detsky AS, Tugwell PX. How attractive does a new technology have to be to warrant adoption and utilization? Tentative guidelines for using clinical and economic evaluations. CMAJ 1992;146:473-81.
21. Laupacis A. Inclusion of drugs in provincial drug benefit programs: who is making these decisions, and are they the right ones? CMAJ 2002;166:44-7.
22. Torrance GW, Raynauld JP, Walker V, et al. A prospective, randomized, pragmatic, health outcomes trial evaluating the incorporation of hylan G-F 20 into the treatment paradigm for patients with knee osteoarthritis (Part 2 of 2): economic results. Osteoarthritis Cartilage 2002;10:518-27.
23. Myllykangas-Luosujarvi R, Lu HS, Chen SL, et al. Comparison of low-dose rofecoxib versus 1000 mg naproxen in patients with osteoarthritis. Results of two randomized treatment trials of six weeks duration. Scand J Rheumatol 2002;31:337-44.
24. Kivitz AJ, Moskowitz RW, Woods E, et al. Comparative efficacy and safety of celecoxib and naproxen in the treatment of osteoarthritis of the hip. J Int Med Res 2001;29:467-79. 25. Fagan SC, Morgenstern LB, Petitta A, et al. Cost-effectiveness of
tissue plasminogen activator for acute ischemic stroke. Neurology 1998;50:883-90.
26. Yen ZS, Davis MA, Chen SC, Chen WJ. A cost-effectiveness analysis of treatment strategies for acute uncomplicated pyelonephritis in women. Acad Emerg Med 2003;10:309-14. 27. Tive L. Celecoxib clinical profile. Rheumatology 2000;39:21-8. 28. Silverstein FE, Faich G, Goldstein JL, et al. Gastrointestinal
toxicity with celecoxib vs. nonsteroidal anti-inflammatory drugs for osteoarthritis and rheumatoid arthritis: the CLASS study: A randomized controlled trial. Celecoxib Long-term Arthritis Safety Study. JAMA 2000;284:1247-55.