Expression of the Ipomoelin Gene from Sweet Potato
1
Pei-Ju Jih, Yu-Chi Chen, and Shih-Tong Jeng*
Department of Botany, National Taiwan University, Taipei 106, Taiwan, Republic of China
The IPO (ipomoelin) gene was isolated from sweet potato (Ipomoea batatas cv Tainung 57) and used as a molecular probe to investigate its regulation by hydrogen peroxide (H2O2) and nitric oxide (NO) after sweet potato was wounded. The expression of the IPO gene was stimulated by H2O2 whether or not the plant was wounded, but its expression after wounding was totally suppressed by the presence of diphenylene iodonium, an inhibitor of NADPH oxidase, both in the local and systemic leaves of sweet potato. These results imply that a signal transduction resulting from the mechanical wounding of sweet potato may involve NADPH oxidase, which produces endogenous H2O2 to stimulate the expression of the IPO gene. The production of H2O2was also required for methyl jasmonate to stimulate the IPO gene expression. On the contrary, NO delayed the expression of the IPO gene, whereas NG
-monomethyl-l-arginine monoacetate, an inhibitor of NO synthase, enhanced the expression of the IPO gene after the plant was wounded. This study also demonstrates that the production of H2O2 stained with 3,3⬘-diaminobenzidine hydrochloride could be stimulated by wounding but was sup-pressed in the presence of NO. Meanwhile, the generation of NO was visualized by confocal scanning microscope in the presence of 4,5-diaminofluorescein diacetate after sweet potato was wounded. In conclusion, when sweet potato was wounded, both H2O2and NO were produced to modulate the plant’s defense system. Together, H2O2and NO regulate the expression of the IPO gene, and their interaction might further stimulate plants to protect themselves from invasions by pathogens and herbivores.
Environmental stresses may lead plants to generate reactive oxygen species (ROS), which include hydro-gen peroxide (H2O2) and superoxide (O2⫺; Bolwell,
1999). Production of excessive ROS may damage cells. When ROS is generated at a controlled level, cells can use these reactive molecules as signals to activate certain genes against attacks by pathogens and herbivores (for review, see Van Breusegem et al., 2001). Therefore, plants have evolved highly orga-nized mechanisms for regulating the level of ROS to maximize benefit to themselves.
H2O2 could be generated during normal cellular
metabolism after various environmental stresses, such as an excess of light, drought, or cold (Dat et al., 2000). Mechanical wounding also stimulates the leaves of several plant species to produce H2O2
lo-cally and systemilo-cally (Bergey et al., 1999; Orozco-Ca´rdenas and Ryan, 1999). The massive production of H2O2could initiate a localized hypersensitive
re-sponse, a form of programmed cell death, which appeared to limit and block pathogen development (Levine et al., 1994). H2O2 may further activate
de-fense genes such as proteinase inhibitors I and II as it diffuses to adjacent cells (Alvarez et al., 1998; Orozco-Ca´rdenas et al., 2001). Also, the generation of H2O2
seems to be mediated by a membrane-bound
NADPH oxidase complex in plants (Lamb and Dixon, 1997; Del Rio et al., 1998; Potikha et al., 1999; Pei et al., 2000), and some chemicals that inhibit NADPH oxidase in mammals also block H2O2
pro-duction in plants (Levine et al., 1994; Auh and Mur-phy, 1995; Alvarez et al., 1998; Orozco-Ca´rdenas and Ryan, 1999).
Nitric oxide (NO) regulates diverse developmental and physiological processes in plants and is involved in growth and differentiation (Gouve´a et al., 1997; Leshem et al., 1998), senescence (Leshem et al., 1998), and seed germination (Keeley and Fotheringham, 1997; Beligni and Lamattina, 2000). Also, NO was shown to act as a signal regulating defense genes to hasten disease resistance in soybean (Glycine max; Delledonne et al., 1998). Mechanical stresses, such as centrifugation, induced Arabidopsis to produce NO, which further caused DNA fragmentation (Garce´s et al., 2001). Although the presence and functions of NO have been well studied in animals, the mechanism for the production of NO remains unclear in plants (Beligni and Lamattina, 2001). In animals, NO syn-thase (NOS) generating NO from l-Arg was identi-fied (Bredt et al., 1991), and NOS activities have also been found in pea (Pisum sativum) and maize (Zea mays; Barroso et al., 1999; Kondo et al., 1999). How-ever, no gene or protein with a sequence highly similar to animals’ NOS has been shown in plants (Beligni and Lamattina, 2001). Also, the production of NO in plants is not restricted to NOS-like activity, and NO can be generated from NO2either through a
light-mediated conversion by carotenoids (Cooney et 1This work was supported by the National Science Council
(grant no. 90 –2311–B– 002– 039 to S.-T. J.).
* Corresponding author; e-mail stjeng@ccms.ntu.edu.tw; fax 886 –2–23918940.
Article, publication date, and citation information can be found at www.plantphysiol.org/cgi/doi/10.1104/pp.102.015701.
al., 1994) or from nitrate reductase (Yamasaki et al., 1999; Yamasaki and Sakihama, 2000).
The NO molecule contains an unpaired electron and, thus, can react with ROS to affect cellular me-tabolism. Under an ordinary physiological condition, superoxide dismutase rapidly converts O2⫺ to H2O2 and an oxygen molecule. However, a large amount of NO may combine with O2⫺ to form peroxynitrite (ONOO⫺), which has been reported to damage lip-ids, proteins, and nucleic acids (Lipton et al., 1993; Yamasaki et al., 1999). Nevertheless, O2⫺ and H2O2
are more toxic than NO and ONOO⫺; therefore, NO may protect cells from destruction (Wink et al., 1993). In accordance, NO has been suggested to have dual roles, either toxic or protective, depending on its environments (Beligni and Lamattina, 1999, 2001).
Sweet potato (Ipomoea batatas cv Tainung 57) is an important crop and a major source of starch world-wide; therefore, there is a wide interest in studying the mechanisms it uses to protect against environ-mental stresses. The expression of the IPO (ipomoe-lin) gene in sweet potato was shown to be enhanced by the application of methyl jasmonate (MeJA) and mechanical wounding (Imanishi et al., 1997). How-ever, regulations of the expression of this gene and its physiological roles remain unclear. Because wound-ing could enhance the expression of the IPO gene, the effects of H2O2and NO on its expression were stud-ied. Also, the interaction between H2O2and NO in
regulating the expression of the IPO gene was investigated.
RESULTS
Stimulation of IPO Gene Expression by H2O2
The expression of the IPO gene is induced by en-vironmental stress such as mechanical wounding lo-cally and systemilo-cally (Imanishi et al., 1997). Me-chanical wounding induces plants to produce H2O2
(Bergey et al., 1999; Orozco-Ca´rdenas and Ryan, 1999). Therefore, it is an interesting topic to investi-gate whether oxidative stress like H2O2can influence the expression of the IPO gene and also whether H2O2 is involved in the signal transduction to sys-temic leaves. Glc together with Glc oxidase (G/GO) was used to provide plant tissues with a continuous generation of H2O2(Levine et al., 1994; Alvarez et al.,
1998). The cut petioles of leaves of sweet potato were immersed in 1⫻ Murashige and Skoog solutions for 12 h to reduce the wounding effect due to the sepa-ration of leaf petiole cuttings from plants. Then, G/GO was added to generate H2O2, or the leaves were wounded for comparison. For another 2, 6, and 12 h, their total RNA was isolated and analyzed by northern blotting (Fig. 1). High-level expression of the IPO gene was observed 2 h later and remained for up to 12 h after H2O2 was generated. Also, the
amount of IPO mRNA accumulation stimulated by H2O2was greater than that of the stimulation caused
by mechanical wounding at every time point tested (Fig. 1). Because neither Glc nor G/GO alone could enhance the expression of the IPO gene (data not shown), the stimulation of the expression of the IPO gene was presumably attributable to H2O2.
The activation of NADPH oxidase complex is be-lieved to participate in the accumulation of H2O2in
plants (Lamb and Dixon, 1997; Del Rio et al., 1998; Potikha et al., 1999; Pei et al., 2000). Diphenylene iodonium (DPI), a chemical inhibitor of NADPH ox-idase, could inhibit the generation of ROS and the accumulation of H2O2after plants were wounded or pathogen infected (Orozco-Ca´rdenas et al., 2001) and was used to examine further the role of H2O2 in stimulating the expression of the IPO gene. Plants with six to eight fully developed leaves were excised at the base of their stems, and DPI was added for 12 h. The third and fourth fully expanded leaves from the terminal bud were then wounded using forceps as local injuries. The first and second fully expanded leaves, which were not wounded on the same plant, were treated as the systemic leaves. Plants without DPI treatment were operated in the same way for comparison (Fig. 2). After the plants were incubated with DPI, the expression of their IPO genes was significantly decreased. DPI inhibited the expression of the IPO gene not only in the local but also in the systemic leaves. Without DPI treatment, the expres-sion of the IPO gene was stimulated by mechanical wounding locally and systematically (Fig. 2). This result suggests that the signal transduction due to the mechanical wounding of sweet potato might include the activation of NADPH oxidase, which produces endogenous H2O2 to stimulate the IPO gene. Also, the ability of DPI to block the systemic expression of the IPO gene suggests that either H2O2participates in the production of systemic signals, or H2O2 by
Figure 1. H2O2induces the expression of the IPO gene. The second
and third fully expanded leaves counting from the terminal bud of a sweet potato were excised, and their cut petioles were immersed in 1⫻ Murashige and Skoog solution for 12 h. Glc and G/GO were added to generate H2O2, or leaves were wounded (W⫹) using
for-ceps, and 2, 6 and 12 h later, their total RNA was isolated and analyzed by northern blotting to detect the amount of IPO mRNA. Ethidium bromide-stained agarose gel presents ribosome RNA (rRNA) as a loading control. Results for untreated leaves are included for comparison.
itself is the systemic signal inducing the expression of the IPO gene in the systemic leaves.
Requirement of H2O2for MeJA to Stimulate IPO Gene Expression
To evaluate the effect of H2O2on stimulation of the IPO gene by MeJA, DPI was added for 12 h before MeJA was put into solution for 2, 6, and 12 h, respec-tively (Fig. 3). Northern analysis revealed that the expression of the IPO gene was blocked at all times in the presence of DPI. Without DPI treatment, MeJA stimulated the expression of the IPO gene normally (Fig. 3). Therefore, the action of MeJA in inducing the IPO gene appeared to require the generation of H2O2. Also, in the signal transduction pathway after wounding, the position of H2O2 is downstream of that of MeJA.
Interference of NO in the Expression of the IPO Gene
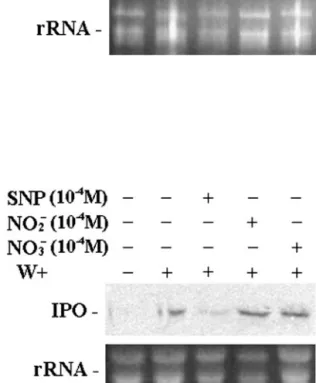
Oxidative stress produced by environments in-cludes not only H2O2but also NO. Sodium nitroprus-side (SNP), a nonenzymatic NO donor, was used to examine the function of NO in regulating the IPO gene. The cut petioles of the excised leaves were put into 1⫻ Murashige and Skoog for 12 h. SNP was added to the concentration of 10⫺2, 10⫺4, and 10⫺6m for 12 h. Northern analysis of IPO mRNA level indi-cates that SNP by itself cannot induce the expression of the IPO gene (Fig. 4A). Furthermore, SNP, sodium nitrite, and sodium nitrate were also applied to the leaf petioles of sweet potato, respectively, for 12 h. Then, leaves were wounded for another 6 h before RNA was isolated for northern analysis (Fig. 4B). The presence of SNP decreased the expression of IPO after sweet potato was wounded. Therefore, the ap-plication of sodium nitrate or sodium nitrite did not influence the accumulation of IPO mRNA induced by mechanical wounding. Therefore, SNP, the NO
do-nor, but not sodium nitrate and sodium nitrite re-duced the expression of the IPO gene.
The period of time between SNP application and wounding was extended to elucidate further the im-pact of NO on the expression of the IPO gene. The cut petioles of the excised leaves were placed in 1⫻ Murashige and Skoog for 12 h, and SNP was added to a concentration of 0.1 mm for 12 h. Leaves were then wounded for another 6 to 24 h before RNA was isolated for analysis. Although the IPO gene was not expressed in the presence of SNP at the time points of 6 and 9 h, it was surprising that it appeared at the time points of 12 and 24 h (Fig. 5). Without SNP treatment, the IPO gene was expressed from the time point of 6 to 24 h (Fig. 5). These results imply that NO did not totally inhibit the expression of the IPO gene but rather delayed it.
Figure 2. DPI inhibits the expression of the IPO gene induced by mechanical wounding. Plants with six to eight fully
developed leaves were excised at the base of their stems, and the cut stems were immersed in 1⫻ Murashige and Skoog for 12 h. The cut stems were treated with or without DPI, inhibiting NADPH oxidase to produce H2O2, at the final concentration
of 0.2 mM, and plants were incubated for another 12 h. The third and fourth fully expanded leaves counting from the terminal
bud were then wounded (W⫹) using forceps to inflict local injuries. The first and second unwounded fully expanded leaves in the same plant were treated as systemic leaves. For another 2, 6, and 12 h, their total RNA was isolated and analyzed by northern blotting to detect the amount of IPO mRNA. Ethidium bromide-stained agarose gel presents ribosome RNA (rRNA) as a loading control. Results from leaves without any treatment are included for comparison.
Figure 3. DPI inhibits the expression of the IPO gene induced by
MeJA. Leaf petiole cuttings were immersed in 1⫻ Murashige and Skoog for 12 h, and some cuttings were supplied with 0.2 mMDPI, an NADPH inhibitor, for another 12 h. All leaves were then treated with 50MMeJA, and their total RNA was extracted 2, 6, and 12 h later before analysis by northern blotting to detect the amount of IPO mRNA. Ethidium bromide-stained agarose gel presents ribosome RNA (rRNA) as a loading control. Results for untreated leaves are included for comparison.
NO Generation after Mechanical Wounding
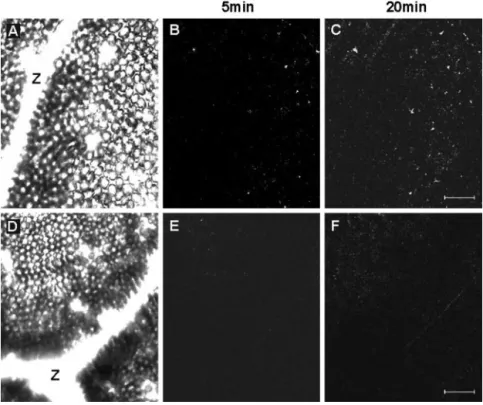
The application of exogenous NO affected the ex-pression of IPO gene; hence, it is important to under-stand whether sweet potato produces NO by itself. The fluorescent probe 4,5-diaminofluorescein diac-etate (DAF-2DA) is highly specific for NO and does not react with other ROS (Foissner et al., 2000). Therefore, the leaves of sweet potato were treated with or without NG-monomethyl-l-Arg monoacetate
(NMMA) and were probed by DAF-2DA. Then, the
leaves of sweet potato were wounded using a needle, and images of the NMMA-treated (Fig. 6, A–C) and -untreated (Fig. 6, D–F) leaves were taken by confocal scanning microscopy 5 and 20 min later. Compared with the image of leaves without NMMA treatment taken at 5 min, the image at 20 min showed that significant amounts of NO were produced 20 min after wounding (Fig. 6, B and C). Also, in the pres-ence of NMMA, the induction of NO was not ob-served (Fig. 6, E and F). These images clearly indicate that mechanical wounding stimulates the production of NO through the activation of NOS, and also that NO is a transduction signal for wounding in sweet potato.
Effects of NO on the Production and Function of H2O2 The action of H2O2in sweet potato was visualized by 3,3⬘-diaminobenzidine hydrochloride (DAB) staining. DAB binding to H2O2undergoes a polymer-ization reaction to yield a dark-brown color (Thordal-Christensen et al., 1997). Without SNP and DPI treat-ment, the dark-brown color was first observed at the midrib 0.5 h after wounding and spread to the lateral and minor veins 1 h later (Fig. 7). This finding implies that mechanical wounding induced the production of H2O2 in sweet potato within 0.5 h. However, the
midrib of the leaves treated with SNP became dark brown at least 1 h after leaves were wounded and was lighter in color than leaves without SNP treat-ment at every time point tested (Fig. 7). In the pres-ence of DPI, the color of the midrib in leaves did not change color at all, and this indicated that the dark color from DAB comes from H2O2. Therefore, NO appeared to reduce and delay the production of H2O2, thereby postponing the expression of the IPO gene.
NO not only reduces the production of H2O2 but also inhibits the function of H2O2. The cut petioles of
Figure 4. Effects of NO donor SNP, nitrite, and nitrate on the
expres-sion of the IPO gene after wounding. Leaf petiole cuttings were immersed in 1⫻ Murashige and Skoog for 12 h and were then treated with 10⫺2, 10⫺4, or 10⫺6MSNP (A) or 10⫺4MSNP, 10⫺4Msodium nitrite, 10⫺4M sodium nitrate, or water as a control for 12 h (B). Leaves in B were then wounded (W⫹) using forceps for another 6 h. Their total RNA was analyzed by northern blotting to detect the amount of IPO mRNA. Ethidium bromide-stained agarose gel pre-sents ribosome RNA (rRNA) as a loading control. Results for un-treated leaves are also included for comparison.
Figure 5. NO postpones the expression of the IPO gene induced by
mechanical wounding. Cut petioles of the excised leaves were im-mersed in 1⫻ Murashige and Skoog for 12 h, and some were treated with 0.1 mM SNP for another 12 h. All these leaves were then wounded (W⫹) using forceps, and their total RNA was analyzed by northern blotting to detect the amount of IPO mRNA 6, 9, 12, and 24 h later. Ethidium bromide-stained agarose gel presents ribosome RNA (rRNA) as a loading control. Results for untreated leaves are included for comparison.
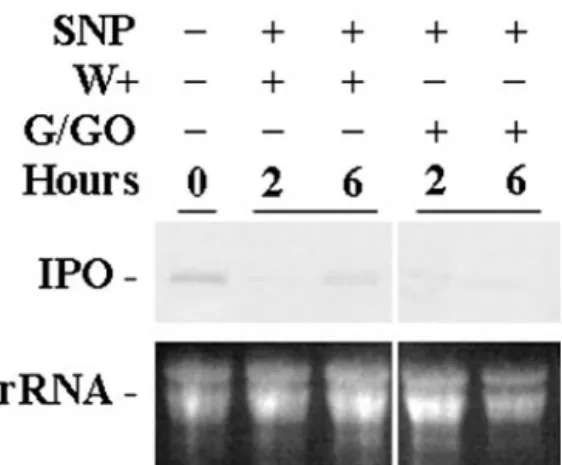
the excised leaves were treated with the NO donor SNP first, and then G/GO releasing H2O2was added
for 2 and 6 h before RNA was isolated. The result shows that in the presence of NO, the ability of H2O2 to stimulate the expression of the IPO gene was to-tally inhibited (Fig. 8). Conceivably, NO not only reduced the production of H2O2but also decreased the function of H2O2 in stimulating the IPO gene
expression.
Effects of NO Deprivation on IPO Gene Expression
No NOS in plants has been found, but NOS activity was reported in plants (Barroso et al., 1999; Kondo et al., 1999). NMMA is an NOS inhibitor and is widely used for studying the function of NO within cells (Oddis et al., 1994). Plants with six to eight fully developed leaves were excised at the base of their stems and incubated in a solution with NMMA. The Figure 6. Mechanical wounding induces NO
generation. Leaf pieces (0.5 cm2) of sweet
po-tato were immersed in loading buffer for 16 h and were added with or without 0.5 mMNMMA,
an NADPH oxidase inhibitor, for another 6 h. DAF-2DA was then added at a final concentra-tion of 10 M. After being washed by loading
buffer, leaf pieces were wounded using a needle and examined by confocal scanning laser mi-croscope, whose wavelength for excitation is 488 nm and for emission is 515 nm. Bright-field image of the wounded leaf without NMMA treatment is shown in A, and images of this area were taken by confocal scanning microscope at 5 (B) and 20 (C) min after wounding. Bright-field image of the wounded leaf treated with NMMA is shown in D, and images of this area were taken by confocal scanning microscope at 5 (E) and 20 (F) min after wounding. Z, Position of a vein. Bar⫽ 40m.
Figure 7. NO reduces the production of the
wound-induced H2O2. The first and second
fully developed leaves counting from the termi-nal bud of sweet potato were immersed in 1⫻ Murashige and Skoog for 12 h and then placed in solution with 0.1 mMSNP, 0.2 mMDPI, or 1⫻
Murashige and Skoog for another 12 h. After DAB was added, leaves were wounded (W⫹) using forceps 6 h later. For another 0, 0.5, 1, 2, and 3 h, the whole leaves were immersed in 96% (w/v) boiling ethanol for 10 min to decol-orize the chloroplast. After cooling, the leaves were stored in ethanol and photographed.
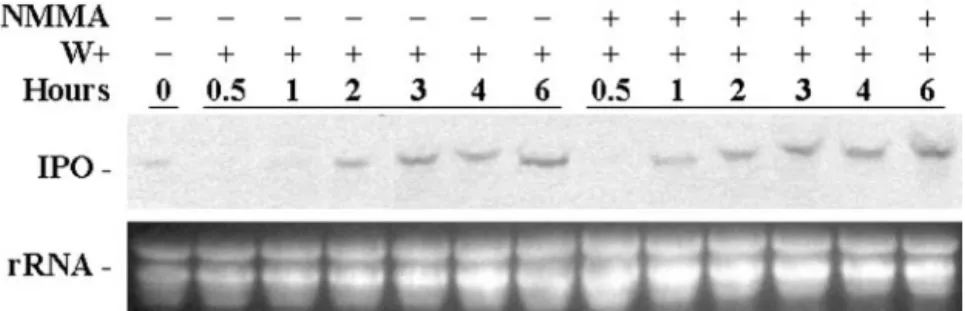
third and fourth fully expanded leaves from the ter-minal bud were then wounded using forceps as local injuries, and the first and second unwounded fully expanded leaves in the same plant were treated as the systemic leaves. Northern analysis indicates that the IPO gene was expressed in both local and sys-temic leaves with or without NMMA treatment (Fig. 9). Also, local leaves treated with NMMA had more IPO mRNA than those without NMMA treatment. This result implies that wounding the leaves of sweet potato might activate an NOS-like enzyme, which produces NO. Therefore, inhibiting NO production during wounding promotes the expression of the IPO gene, and this result is constant with that in Figure 5, which shows that the presence of NO reduced the expression of the IPO gene. This conclusion is further supported by the time point assays after wounding in the presence of NMMA (Fig. 10). In the leaves treated with NMMA, IPO mRNA was produced 1 h after wounding; however, without NMMA treatment, their IPO mRNA appeared in the leaves 2 h later after wounding (Fig. 10). This finding agrees with that of Figure 9 and indicates that inhibition of the genera-tion of NO accelerated the expression of the IPO gene after wounding.
DISCUSSION
Wounding has been shown to stimulate the pro-duction of H2O2 in tobacco (Nicotiana tabacum) and
cassava (Manihot esculenta Crantz.) plants (Repka, 1999). This study also demonstrates that H2O2 was
generated in mechanically wounded leaves of sweet potato (Fig. 7). Furthermore, among 18 plant species from six families examined, 14 of them produced H2O2in the wounded leaves (Orozco-Ca´rdenas and
Ryan, 1999). Also, DAB staining revealed the pres-ence of wound-induced H2O2in the systemic leaves of tomato (Lycopersicon esculentum) plant (Orozco-Ca´rdenas and Ryan, 1999). Thus, the generation of H2O2after wounding seems to be widespread in the
plant kingdom and protects plants from attack by herbivores.
In the presence of DPI, which inhibits the genera-tion of H2O2from NADPH oxidase, the expression of
the IPO gene in sweet potato was blocked not only in local but also in systemic leaves (Fig. 2). Two possible explanations exist. First, a systemin-like protein may be present in sweet potato and signals H2O2to
acti-vate the IPO gene in systemic leaves. In the presence of DPI, even though wounding could stimulate the production of a systemin-like protein, DPI inhibited the production of H2O2 and, thus, suppressed the
systemic expression of the IPO gene. This deduction is supported by the finding that systemin functions as a first messenger of the wounding signal to the systemic leaves, and H2O2acts downstream of
syste-min and is considered to be a second messenger in tomato (Orozco-Ca´rdenas et al., 2001). Second, DPI blocking the systemic expression of the IPO gene may suggest that H2O2participates in the production
of a systemic signal, or that H2O2itself or a related compound derived from H2O2is the systemic signal
that stimulates the expression of the IPO gene in systemic leaves. This claim is supported by a recent finding that the production of MeJA in response to wounding or systemin was required to produce a long-distance signal to systemic leaves (Li et al., 2002). Also, the function of MeJA to stimulate the IPO gene was dependent on the production of H2O2(Fig. Figure 8. NO inhibits the expression of the IPO gene induced by
H2O2. Cut petioles of the excised leaves were placed in 1⫻
Murash-ige and Skoog for 12 h, and SNP was added at a concentration of 0.1 mMfor another 12 h. These leaves were wounded (W⫹) or treated with Glc and G/GO to produce H2O2. Their total RNA was isolated
2 and 6 h later to detect the amount of IPO mRNA by northern blotting. Ethidium bromide-stained agarose gel presents ribosome RNA (rRNA) as a loading control. Results for untreated leaves are included for comparison.
Figure 9. Effect of NMMA on the expression of IPO gene in local and
systemic leaves. Plants with six to eight fully developed leaves were excised at the base of their stems, and their cut stems were immersed in 1⫻ Murashige and Skoog for 12 h. NMMA, inhibiting NOS-producing NO, was added to a final concentration of 0.5 mMfor another 12 h, and the third and fourth fully expanded leaves counting from the terminal bud were then wounded (W⫹) using forceps as local (L) injuries. The first and second unwounded fully expanded leaves in the same plant were treated as systemic (S) leaves. Their total RNA was isolated and analyzed by northern blotting to detect the amount of IPO mRNA 6 h later. Ethidium bromide-stained aga-rose gel presents ribosome RNA (rRNA) as a loading control. Result for untreated leaves is included for comparison.
3). These results perhaps imply that the production of H2O2was required for the generation of a systemic
signal to be sent to the distal leaves.
Within cells, H2O2 can be generated via several
different metabolite pathways (Dat et al., 2000). However, the inhibition of NADPH oxidase by DPI blocked the expression of the IPO gene in both local and systemic leaves after wounding (Fig. 2). This finding implies that NADPH oxidase regulates the production of H2O2in the local and systemic leaves
of sweet potato and that the wound signal must pass through the sole transducer, H2O2, to stimulate the
expression of the IPO gene. Similarly, the presence of DPI reduced the effectiveness of MeJA (Fig. 3), indi-cating that the signal transduction pathway from MeJA to stimulate IPO gene must go via H2O2. In
addition, DAB staining revealed that MeJA stimu-lated the production of H2O2 in tomato
(Orozco-Ca´rdenas and Ryan, 1999). Therefore, MeJA might activate the membrane-bound NADPH oxidase to produce H2O2, which further induced the expression of the IPO gene. Also, this pathway occurred in both local and systemic leaves.
NO has also been demonstrated to regulate plants’ defense systems against pathogens (Dangl, 1998; Durner and Klessig, 1999). The IPO gene was in-duced by mechanical wounding and MeJA (Fig. 3); therefore, it might be related to the defense system in sweet potato. SNP, an NO donor, delayed the expres-sion of the IPO gene after sweet potato was mechan-ically wounded (Fig. 5), and at the same time the presence of NMMA, an NOS inhibitor, accelerated the expression of the IPO gene (Fig. 10). These results may suggest that the presence of NO interferes with the wounding signal in stimulating the IPO gene. Also, NOS-like proteins have been identified in plants by animal anti-NOS antiserum (Kuo et al., 1995; Sen and Cheema, 1995; Barroso et al., 1999), and the accelerated expression of the IPO gene by NMMA may further indicate that NO was generated from an NOS-like protein after the sweet potato was wounded. Furthermore, NO was produced after sweet potato was wounded (Fig. 6). Therefore, sweet potato after wounding generated both H2O2and NO
within cells. H2O2induced the expression of the IPO gene, whereas NO interfered in its expression.
The NO donor SNP inhibited the expression of the IPO gene at the first few hours, and delayed its expression until 12 h after sweet potato was wounded (Fig. 5). The inhibition of SNP in the wound-inducible protein was also observed in to-mato plants. Synthesis of proteinase inhibitor I was repressed by SNP after tomato was wounded or treated with systemin (Orozco-Ca´rdenas and Ryan, 2002). However, SNP only blocked the production of H2O2induced by systemin and MeJA in tomato but
not the H2O2 generated from G/GO (Orozco-Ca´rdenas and Ryan, 2002). As a consequence, the production of proteinase inhibitor I induced by H2O2 generated from G/GO was not affected by SNP; therefore, it was concluded that NO influenced the signal pathway downstream from MeJA synthesis and upstream of H2O2 synthesis (Orozco-Ca´rdenas and Ryan, 2002). However, SNP reduced the produc-tion of H2O2 generated from mechanical wounding (Fig. 7) and inhibited the expression of the IPO gene induced by both mechanical wounding and G/GO (Figs. 5 and 8). Therefore, NO appeared to affect both the production and function of H2O2, and interacted with the components in the signal pathway upstream and downstream of H2O2synthesis in sweet potato. Upon wounding, both H2O2 and NO were
pro-duced in sweet potato (Figs. 6 and 7), and then both might participate in plants’ defense system. The in-teraction between H2O2 and NO generates at least two effects. First, H2O2and NO may react
synergis-tically to initiate a hypersensitive response, which promotes cell death in the cells infected with patho-gens and limits further invasion by the pathopatho-gens (Delledonne et al., 1998). Second, NO can react with O2⫺, which can change to H2O2within cells to form ONOO⫺; this may, in turn, damage proteins, lipids, and nucleic acids (Lipton et al., 1993) to generate antimicrobial effects (Durner and Klessig, 1999). Also, the presence of NO not only reduced the amount of H2O2 produced by sweet potato after
wounding (Fig. 7) but also postponed the expression of the IPO gene (Fig. 5). Thus, the decline in the Figure 10. NOS inhibitor NMMA accelerates the expression of the IPO gene. Cut petioles of the excised leaves were placed
in 1⫻ Murashige and Skoog for 12 h, and some petioles were treated with 0.5 mMNMMA to inhibit NOS producing NO for another 12 h. Leaves were then wounded (W⫹), and 0, 0.5, 1, 2, 3, 4 and 6 h later, their total RNA was isolated to detect the amount of IPO mRNA by northern blotting. Ethidium bromide-stained agarose gel presents ribosome RNA (rRNA) as a loading control. Results for untreated leaves are included for comparison.
amount of H2O2 might be due to the interaction
between H2O2and NO to form ONOO⫺. The expres-sion of the IPO gene might not be the first priority in protecting sweet potato from invasion by pathogens and herbivores. In accordance, NO cooperating with H2O2modulates the plant’s defense system and de-lays the expression of the IPO gene.
Mechanical wounding causes sweet potato to pro-duce MeJA, which activates NADPH oxidase to gen-erate H2O2. Wounding simultaneously causes the NOS-like protein to generate NO. H2O2and NO
co-operatively and quickly initiate the defense system, including programmed cell death at the wounded or infected cells to limit the possibility of further attack on neighboring healthy cells by pathogens or herbi-vores. Also, ONOO⫺, formed by H2O2and NO, may damage the pathogens’ proteins, lipids, and nucleic acids. Later, H2O2 activates a slow defense system, which may induce the systemic expression of genes, such as the IPO gene, to protect plants from further invasion. Thus, plants have developed delicate de-fense systems to survive in nature.
MATERIALS AND METHODS Plant Materials and Treatments
Sweet potato (Ipomoea batatas cv Tainung 57) plants were vegetatively propagated from cuttings and grown in a controlled environment (16-h/ 25°C day, 8-h/22°C night, humidity 70%, light 100mol photons m⫺2s⫺1).
Plants with six to eight fully developed leaves were used. For the experi-ments involving local and systemic leaves, plants were excised at the base of the stem with a razor blade, and their cut stems were used. Local injury was defined as mechanical wounding at the third and fourth fully expanded leaves counting from the terminal bud. In addition, the first and second fully expanded leaves, which were not wounded on the same plant, were taken as the systemic leaves. For the experiments using a single leaf, the second or third fully expanded leaves from the terminal bud was excised, and its cut petioles was used. Plants were wounded by pressing leaves with forceps.
For those experiments involving one treatment with chemical reagents, cut stems or cut petioles were incubated in 1⫻ Murashige and Skoog (Murashige and Skoog, 1962; pH 5.8) for 12 h. Then, plants were wounded or treated with 50m Glc with 2.5 units mL⫺1G/GO, SNP (10, 0.1, or 0.001
mm), 0.1 mm sodium nitrite, or 0.1 mm sodium nitrate for the time indicated in each assay. For those experiments involving two chemical treatments, cut stems or cut petioles were also incubated in 1⫻ Murashige and Skoog for 12 h. After 0.2 mm DPI, 0.1 mm SNP or 0.5 mm NMMA was added for another 12 h, and plants were wounded or treated with 50m MeJA or 50 m Glc with 2.5 units mL⫺1G/GO for the time indicated in each assay. All
reagents were from Sigma (St Louis). DPI is an inhibitor for NADPH oxidase, SNP is a nonenzymatic NO donor, and NMMA is an NOS inhibitor.
RNA Isolation and Analysis
Total RNA was isolated from liquid N2-ground leaves following the
procedure described by Chomzynski and Sacchi (1987) except that guanidium-HCl rather than guanidium thiocyanate was used. The quantity of RNA was estimated using a spectophotometer, and its quality was determined by agarose gel electrophoresis with formaldehyde. Total RNA (10g) was loaded and separated on formaldehyde-agarose gels and trans-ferred to nylon membranes before hybridization with radiolabeled probes (Sambrook et al., 1989). The radiolabeled IPO was produced in PCR using the IPO cDNA template isolated from a subtraction library (Y.-C. Chen and S.-T. Jeng, unpublished result). Prehybridization was undertaken in 5⫻ SSPE (0.05 m NaH2PO4 [pH 6.8], 0.9 m NaCl, and 5 mm EDTA), 0.5% (w/v)
SDS, 5⫻ Denhard’s solution (0.1% [w/v] Ficoll, 0.1% [w/v] bovine serum albumin, and 0.1% [w/v] polyvinyl pyrrolidine) at 65°C for 1 h. After the
radiolabeled probe was added, hybridization was performed under the same conditions for 16 h. Blots were washed twice in 0.1⫻ SSPE and 0.1% (w/v) SDS at 65°C for 15 min. Radioactive blots were displayed on the PhosphorImager (Molecular Dynamics, Sunnyvale, CA), and their autora-diographs were printed on an XLS 8600 PS printer (Eastman-Kodak, Roch-ester, NY). All experiments were repeated at least three times, and similar results were obtained.
H2O2Detection by DAB Staining
H2O2was visualized by staining with DAB (Thordal-Christensen et al.,
1997). DAB undergoes polymerization reaction to yield a dark-brown color once it encounters H2O2(Thordal-Christensen et al., 1997). The first and
second fully developed leaves counting from the terminal bud of a sweet potato were excised, and the leaf petiole cuttings were immersed in 1⫻ Murashige and Skoog for 12 h. The Murashige and Skoog solution was then added with or without 0.1 mm SNP, an NO donor, or 0.2 mm DPI, an NADPH oxidase inhibitor, for 12 h before 1 mg mL⫺1DAB was put into
solution for another 6 h. Leaves were then wounded using forceps, and 0, 0.5, 1, 2, and 3 h later, leaves were immersed in 96% (w/v) boiling ethanol for 10 min to decolorize the chloroplast but not the deep-brown polymer-ization product formed by DAB with H2O2. After cooling, the leaves were
kept in the ethanol and photographed.
Visualization of NO
Fully expanding leaves were cut into small pieces of 0.5 cm2in area and
immersed in loading buffer (10 mm Tris-KCl [pH 7.2]) for 16 h. Leaf pieces were then transferred to the loading buffer with or without 0.5 mm NMMA for another 6 h. After adding DAF-2DA (Calbiochem, La Jolla, CA) at a final concentration of 10 m, leaf pieces were incubated in the dark for 1 h (Foissner et al., 2000; Pedroso et al., 2000). The extra DAF-2DA was removed by washing leaf pieces with loading buffer for 30 min. Leaf pieces were wounded with a needle before images were taken using a TCS-SP2 confocal laser scanning microscope (Leica Lasertechnik GmbH, Heidelberg) 5 and 20 min later (excitation⫽ 488 nm and emission ⫽ 515 nm).
ACKNOWLEDGMENT
We are grateful to Dr. Chia-Yin Tsai (Department of Botany, National Taiwan University) for helpful discussion.
Received October 7, 2002; returned for revision November 28, 2002; accepted February 13, 2003.
LITERATURE CITED
Alvarez ME, Penell RI, Meijer P-J, Ishikawa A, Dixon RA, Lamb C(1998) Reactive oxygen intermediates mediate a systemic signal network in the establishment of plant immunity. Cell 92: 773–784
Auh C-K, Murphy TM(1995) Plasma membrane redox enzyme is involved in the synthesis of O2and H2O2by phytophthrona elicitor-stimulated rose
cells. Plant Physiol 107: 1241–1247
Barroso JB, Corpas FJ, Carreras A, Sandalio LM, Valderrama R, Palma JM, Lupia´nez JA, del Rı´o LA(1999) Localization of nitric-oxide synthase in plant peroxisomes. J Biol Chem 274: 36729–36733
Beligni MV, Lamattina L(1999) Is nitric oxide toxic or protective? Trends Plant Sci 4: 299–300
Beligni MV, Lamattina L(2000) Nitric oxide induces seed germination and de-etiolation, and inhibits hypocotyl elongation, three light-inducible responses in plants. Planta 210: 215–221
Beligni MV, Lamattina L(2001) Nitric oxide in plants: the history is just beginning. Plant Cell Environ 24: 267–278
Bergey DR, Orozco-Ca´rdenas ML, de Monra DS, Ryan CA (1999) A wound- and systemic-inducible polygalacturonase in tomato leaves. Proc Natl Acad Sci USA 96: 1756–1760
Bolwell GP(1999) Role of active oxygen species and NO in plant defence responses. Curr Opin Plant Biol 2: 287–294
Bredt DS, Hwang PM, Glatt CE, Lowenstein C, Reed RR, Snyder SH
(1991) Cloned and expressed nitric oxide synthase structurally resembles cytochrome P-450 reductase. Nature 351: 714–717
Chomzynski P, Sacchi N(1987) Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Bio-chem 162: 156–159
Cooney RV, Harwood PJ, Custer LJ, Franke AA (1994) Light-mediated conversion of nitrogen dioxide to nitric oxide by carotenoids. Environ Health Perspect 102: 460–462
Dangl J(1998) Innate immunity: Plants just say NO to pathogens. Nature
394:525–527
Dat J, Vandenabeele S, Vranova E, Van Montagu M, Inze D, Van Breu-segem F(2000) Dual action of the Active oxygen species during plant stress responses. Cell Mol Life Sci 57: 779–795
Delledonne M, Xia Y, Dixon RA, Lamb C(1998) Nitric oxide functions as a signal in plant disease resistance. Nature 394: 585–588
Del Rio LA, Pastori GM, Palma JM, Sandalio LM, Sevilla F, Corpas FJ, Jime´nez A, Lo´pez-Huertas E, Hernandez JA(1998) The activated oxygen role of peroxisomes in senescence. Plant Physiol 116: 1195–1200
Durner J, Klessig DF(1999) Nitric oxide as a signal in plants. Curr Opin Plant Biol 2: 369–374
Foissner I, Wendehenne D, Langebartels C, Durner J(2000) In vivo im-aging of an elicitor-induced nitric oxide burst in tobacco. Plant J 23: 817–824
Garce´s H, Durzan D, Pedroso MC(2001) Mechanical stress elicits nitric oxide formation and DNA fragmentation in Arabidopsis thaliana. Ann Bot
87:567–574
Gouve´a CMCP, Souza JF, Magalhaˆes ACN, Martins IS(1997) NO-releasing substances that induce growth elongation in maize root segments. Plant Growth Regul 21: 183–187
Imanishi S, Kito-Nakamura K, Matsuoka K, Morikami A, Nakamura K
(1997) A major jasmonate-inducible protein of sweet potato, ipomoelin, is an ABA-independent wound-inducible protein. Plant Cell Physiol 38: 643–652
Keeley JE, Fotheringham CJ(1997) Trace gas emissions and smoke-induced seed germination. Science 276: 1248–1250
Kondo R, Tikunova SB, Cho MJ, Johnson JD(1999) A point mutation in a plant calmodulin is responsible for its inhibition of nitric-oxide synthase. J Biol Chem 274: 36213–36218
Kuo WN, Kuo TW, Jones DL, Baptiste J Jr(1995) Nitric oxide synthase immunoreactivity in baker’s yeast, lobster and wheat germ. Biochem Arch
11:73–78
Lamb C, Dixon RA(1997) The oxidative burst in plant disease resistance. Annu Rev Plant Phys 48: 251–275
Leshem Y, Wills RBH, Ku VV(1998) Evidence for the function of the free radical gas, nitric oxide (NO.), as an endogenous maturation and senes-cence regulating factor in higher plants. Plant Physiol Biochem 36: 825–833
Levine A, Tenhaken R, Dixon R, Lamb C(1994) H2O2from the oxidative
burst orchestrates the plant hypersensitive disease resistance response. Cell 79: 583–593
Li L, Li C, Lee GI, Howe GA(2002) Distinct roles for jasmonate synthesis and action in the systemic wound response of tomato. Proc Natl Acad Sci USA 99: 6416–6421
Lipton SA, Yun-Beom CH, Pan Z-H, Le SZ, Vincent Chen H-S, Sucher NJ, Loscalzo J, Singel DJ, Stamler JS(1993) A redox-based mechanism for the neuroprotective and neurodestructive effects of nitric oxide and related nitroso-compounds. Nature 364: 626–632
Murashige T, Skoog F(1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15: 473–497
Oddis CV, Simmons RL, Hattler BG, Finkel MS (1994) Chronotropic effects of cytokines and the nitric oxide synthase inhibitor, l-NMMA, on cardiac myocytes. Biochem Biophys Res Commun 205: 992–997
Orozco-Ca´rdenas ML, Narva´ez-Va´squez J, Ryan CA(2001) Hydrogen per-oxide acts as a second messenger for the induction of defense genes in tomato plants in response to wounding, systemin, and methyl jasmonate. Plant Cell 13: 179–191
Orozco-Ca´rdenas ML, Ryan CA(1999) Hydrogen peroxide is generated systemically in plant leaves by wounding and systemin via the octade-canoid pathway. Proc Natl Acad Sci USA 96: 6553–6557
Orozco-Ca´rdenas ML, Ryan CA(2002) Nitric oxide negatively modulates wound signaling in tomato plants. Plant Physiol 130: 487–493
Pedroso MC, Magalhaes JR, Durzan D(2000) A nitric oxide burst precedes apoptosis in angiosperm and gymnosperm callus cells and foliar tissues. J Exp Bot 51: 1027–1036
Pei Z-M, Murata Y, Benning G, Thomine S, Klusener B, Allen GJ, Grill E, Schroeder JJ(2000) Calcium channels activated by hydrogen peroxide mediate abscisic acid signaling in guard cells. Nature 406: 731–734
Potikha TS, Collins CC, Johnson DI, Delmer DP, Levine A(1999) The involvement of hydrogen peroxide in the differentiation of secondary walls in cotton fibers. Plant Physiol 119: 849–858
Repka V(1999) Improved histochemical test for in situ detection of hydro-gen peroxide in cells undergoing oxidative burst or lignification. Biol Plant 42: 599–607
Sambrook J, Fritsch EF, Maniatis T(1989) Molecular Cloning: A Labora-tory Manual, Ed 2. Cold Spring Harbor LaboraLabora-tory Press, Cold Spring Harbor, NY
Sen S, Cheema IR(1995) Nitric oxide synthase and calmodulin immuno-reactivity in plant embryonic tissue. Biochem Arch 11: 221–227
Thordal-Christensen H, Zhang Z, Wei Y, Collinge DB(1997) Subcellular localization of H2O2in plants: H2O2accumulation in papillae and
hy-persensitive response during the barley-powdery mildew interaction. Plant J 11: 1187–1194
Van Breusegem F, Vranova´ E, Dat JF, Inze´ D (2001) The role of active oxygen species in plant signal transduction. Plant Sci 161: 405–414
Wink DA, Hanbauer I, Krishna MC, DeGraff W, Gamson J, Mitchell JB
(1993) Nitric oxide protects against cellular damage and cytotoxicity from reactive oxygen species. Proc Nat Acad Sci USA 90: 9813–9817
Yamasaki H, Sakihama Y, Takahashi S(1999) An alternative pathway for nitric oxide production in plants: new features of an old enzyme. Trends Plant Sci 4: 128–129
Yamasaki H, Sakihama Y(2000) Simultaneous production of nitric oxide and peroxynitrite by plant nitrate reductase: in vitro evidence for the NR-dependent formation of active nitrogen species. FEBS Lett 468: 89–92