Thermal properties of rigid rod epoxies cured with
diaminodiphenylsulfone and dicyandiamide
Wei-Fang Su
a,*, Hong-Wen Huang
a, Wei-Ping Pan
ba
Institute of Materials Science and Engineering, National Taiwan University, Taipei, Taiwan
b
Materials Characterization Center, Western Kentucky University, Bowling Green, KY 42101, USA
Abstract
The differences of thermal properties between rigid rod and flexible bisphenol A (DGEBA) epoxies were studied using modulated differential scanning calorimeter (MDSC), thermal gravimetric analyzer (TGA) and dynamic mechanical analyzer (DMA) techniques. The rigid rod epoxies of azomethine (AM), biphenol (BP) and tetramethyl biphenol (TMBP) were cured with diaminodiphenylsulfone (DDS) and dicyandiamide (Dicy), respectively. The DDS-cured epoxies have higher glass transition temperature (Tg) and decomposition temperature than the Dicy-cured epoxies due to the high functionality and rigid structure of
DDS. The AM, BP and TMBP have higher Tgthan DGEBA due to the rigid rod structure. The bulky methyl substitution on the BP
main chain of TMBP epoxy decreases the order of polymer packing and reduces the Tg. The AM epoxy has the longest rigid rod
structure, thus the AM epoxy has a higher Tgthan that of BP and TMBP epoxies.
# 2002 Elsevier Science B.V. All rights reserved.
Keywords: Thermal properties; Rigid rod epoxy; Azomethine epoxy; Biphenol epoxy; Amine curing agent
1. Introduction
Epoxy resins have been extensively used in composite materials and as adhesives. The polymers with aromatic rigid rod moiety are known to have excellent thermal and mechanical properties [1,2]. Recently, epoxy resin containing rigid rod structure has been synthesized as a new class of high performance polymers. Most of rigid rod epoxies exhibit liquid crystalline behavior. Some of rigid rod liquid crystal epoxies retained liquid crystal behavior after cured with curing agent. They have exhibited excellent thermal properties [3,4].
The thermal properties of cured epoxies are deter-mined by the structure of epoxy, cross-linking density and the functionality of epoxy and curing agent [1–6].
In this study, rigid rod epoxies, azomethine (AM), biphenol (BP) and tetramethyl biphenol (TMBP), were cured with diaminodiphenylsulfone (DDS) and dicyandiamide (Dicy), respectively. The selection of DDS was based on its high performance in the epoxy-based composite materials. The choice of Dicy was because of its low cost, good processability and elec-trical properties for printed wiring boards.
The effects of chemical structure changes on the thermal properties of cured epoxies are investigated. The results are reported and discussed in Section 3.
2. Experimental 2.1. Materials
The bisphenol A (DGEBA) (trade name DER 331) was obtained from Dow Chemical Company with an
Thermochimica Acta 392–393 (2002) 391–394
*Corresponding author.
E-mail address: suwf@ccms.ntu.edu.tw (W.-F. Su).
0040-6031/02/$ – see front matter # 2002 Elsevier Science B.V. All rights reserved. PII: S 0 0 4 0 - 6 0 3 1 ( 0 2 ) 0 0 1 2 5 - 9
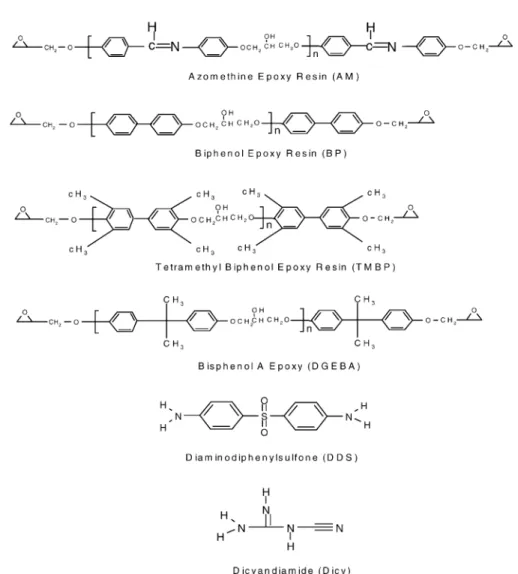
epoxy equivalent weight of 185.5. The TMBP epoxy (trade name RSS1407) was supplied from Shell Che-mical Company with an epoxy equivalent weight of185. The AM (4,40-bis(2,3-epoxypropoxy)benzylidene ani-line) with an epoxy equivalent weight of 171 and the BP (4,40-bis(2,3-epoxypropoxy)biphenyl) with an epoxy equivalent weight of 167.6 were synthesized accord-ing to literature procedure [2]. The curaccord-ing agents (DDS and Dicy) were purchased from Aldrich. The structures of epoxy resins and curing agents are shown in Fig. 1. 2.2. Preparation of cured epoxies
A DuPont DSC 9900-910 was employed for the determinations of the curing conditions. All samples
were mixed with stoichiometric amount of epoxy and curing agent. Fully cured samples exhibit no exother-mal peak and no increasing in glass transition tem-perature (Tg). The curing conditions of each sample
are shown in Table 1. 2.3. Instrumentation
A TA 2920 modulated differential scanning
calori-meter (MDSC) (5 8C/min in 50 ml/min N2) was
used to determine the Tg of cured samples. A TA
2980 dynamic mechanical analyzer (DMA) (5 8C/ min in air, 1 Hz, 20 mm amplitude) was used to measure the change in modulus and damping pro-perties. A TA 2950 thermal gravimetric analyzer
Fig. 1. Chemical structures of epoxy resins and curing agents. 392 W.-F. Su et al. / Thermochimica Acta 392–393 (2002) 391–394
(TGA) (10 8C/min in N2) was used to study the
thermal stability.
3. Results and discussion
Table 1 shows AM, BP and TMBP rigid rod epoxies need to be cured at higher temperature and longer time than flexible DGEBA epoxy. The results indicate that the rigid rod epoxies have a slower curing rate than that of flexible epoxy due to the slow molecular motions of rigid rod structure in the rigid rod epoxies. The AM epoxy has the longest rigid rod chain length, so it requires the highest post-cure temperature. The post-cure temperature of TMBP is higher than that of BP because of bulky methyl substitution on the BP main chain of TMBP.
From Table 2, AM, BP and TMBP, cured with DDS
have higher Tg than the DGEBA epoxy due to their
rigid rod structure. The AM epoxy contains the longest rigid rod chain length with the slowest molecular
movement, so the AM epoxy has the highest Tg.
The bulky substitution on TMBP epoxy decreases the ordered structure of the rigid rod moiety, so it has a lower Tgthan BP.
Table 3 indicates that AM, BP and TMBP epoxies have better thermal stability than the flexible epoxy due to their rigid rod structure. The AM epoxy exhibits
the lowest weight loss at 600 8C, which may be due to the formation of intermolecular hydrogen bonds [3]. The bulky substitution on TMBP epoxy reduces its thermal stability as compared with BP epoxy. The decomposition temperature of AM and BP epoxies are lower than that of DGEBA, the reason is not clear. However, it may be from the low curing rate of rigid rod epoxies.
Table 4 shows the DDS-cured epoxies have a higher Tgthan Dicy-cured epoxies due to the high
function-ality and rigid structure of DDS. The Dicy-cured AM
epoxy has a higher Tgthan DGEBA epoxy because its
rigid rod of AM epoxy.
4. Conclusions
The rigid rod AM, BP and TMBP epoxies exhibit
higher Tg and weight retention at high temperature
than the flexible DGEBA epoxy due to the low mole-cular motion and high thermal stability of rigid rod structure. The AM epoxy has the best thermal proper-ties among the rigid rod epoxies because of its longest rigid rod chain and possible intermolecular chain formation in the cured network. The DDS-cured epoxies have a better thermal stability than Dicy-cured epoxies because of the high functionality and rigid structure of DDS.
Table 1
Curing conditions of epoxy resin systems
Sample Pre-cure (8C/h) Post-cure (8C/h) AM/DDS 160/3þ 200/2 þ 220/2 240/1 BP/DDS 180/6 220/4 TMBP/DDS 180/2þ 220/2 230/1 DGEBA/DDS 150/4þ 180/2 þ 200/2 220/2 AM/Dicy 120/0.5þ 125/1 þ 155/1 200/1 DGEBA/Dicy 150/6þ 175/2 200/3 Table 2
Tgof DDS-cured different epoxy resin systems
Sample MDSC (8C) TMA (8C) DMA (8C) (tan d)
AM/DDS 240 219 –
BP/DDS 211 213 235 TMBP/DDS 180 – 234 DGEBA/DDS 163 208 193
Table 3
TGA results of epoxy resin systems Sample Temperature at 5 wt.% loss (8C) Percentage weight loss at 600 8C AM/DDS 339 60.33 BP/DDS 339 69.64 TMBP/DDS 336 82.70 DGEBA/DDS 377 84.60 Table 4
Tgof epoxy cured with different curing agents
Sample MDSC (8C)
AM/DDS 240
AM/Dicy 173
DGEBA/DDS 163 DGEBA/Dicy 138
Acknowledgements
Financial supports from National Science Council under grant number NSC89-2218-E-002-072 and Western Kentucky University Visiting Scholarship Funds are highly appreciated.
References
[1] W.F.A. Su, K.C. Chen, S.Y. Tseng, Bull. Coll. Eng. N.T.U. 77 (1999) 73.
[2] W.F.A. Su, K.C. Chen, S.Y. Tseng, J. Appl. Polym. Sci. 78 (2000) 446.
[3] J.Y. Lee, J. Jang, S.S. Hwang, S.M. Hong, K.U. Kim, Polymer 39 (1998) 24.
[4] J.Y. Lee, J. Jang, S.S. Hwang, S.M. Hong, K.U. Kim, Polymer 39 (1998) 24.
[5] V. Sykora, V. Spacek, I. Dobas, J. Appl. Polym. Sci. 54 (1994) 1463.
[6] C.S. Wang, J.Y. Shieh, J. Appl. Polym. Sci. 73 (1999) 353.