行政院國家科學委員會專題研究計畫 成果報告
以蛋白質體學方法尋找台灣腎細胞癌病患之特殊腫瘤相關
抗原
計畫類別: 個別型計畫 計畫編號: NSC92-2314-B-002-337- 執行期間: 92 年 08 月 01 日至 93 年 07 月 31 日 執行單位: 國立臺灣大學醫學院泌尿科 計畫主持人: 闕士傑 共同主持人: 賴明坤 計畫參與人員: 王國忠 報告類型: 精簡報告 處理方式: 本計畫可公開查詢中 華 民 國 93 年 12 月 28 日
Surface Enhanced Laser-assisted De/ionization Time-Of-Fly (SELDI-TOF) mass spectrometry resolved distinct profile of Renal Cell Carcinoma (RCC) patient’s serum
Kao-Chung Wang, Shih-Chieh Jeff Chueh and Min-Kuen Lai
Department of Urology, National Taiwan University Hospital, 7, Chung-Shan S. Rd. Taipei, Taiwan 100
Introduction:
Renal cell carcinoma (RCC) is a potentially lethal malignancy. Many of the RCC are asymptomatic and are discovered incidentally during routine physical checkup. Therefore, effective screen marker is needed for RCC. Recently, MN/CA9 had been reported as renal cell carcinoma (RCC) marker. Further challenge of MN/CA9 to immunotherapy reveals fare results, which imply additional tumor marker (s) for RCC.
Tumor markers for malignant disease had been very useful either for diagnosis or prognosis. Recently, surface enhanced laser desorption/ionization-time of flight-mass spectrometry (SELDI-TOF-MS) had been used to screen neoplasms (Kozak et al 2003, Qu et al 2002) as well as alcohol abuse (Nomura et al 2004).
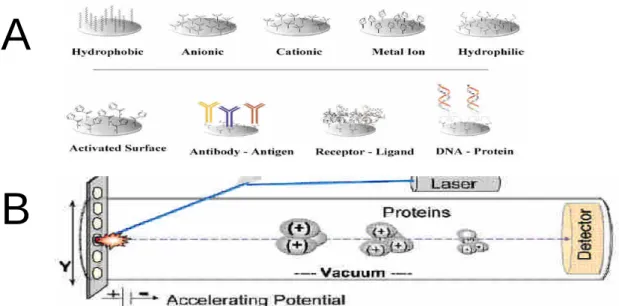
SELDI-TOF is a novel type of mass spectrometry. It uses aluminum chips with special coating surface to capture specific protein of interest (Figure 1A). After wash off non-specific binding protein, the chips is sent to a vacuum chamber and
bombarded with laser. The proteins on the aluminum surface is excited with the matrix and fly though the chamber. The detector in SELDI-TOFrecordsprotein’s mass according to the time-of-fly and expressed as m/z (Figure 1B). The SELDI-TOF technology has been used directly to detect protein fingerprints in complex biological samples, such as urine (Vlahou et al 2001) and serum (Rosty et al 2002).
In this study, we used SELDI technology to compare paired serum samples obtained before and after nephrectomy for RCC patients. After analysis, five clusters of polypeptides with reciprocal behavior had been found.
Materials and methods
We used serum from 36 patients to establish the model. Patients diagnosed with RCC grade I/II were included. For every patient, serum samples collected before nephrectomy were grouped as pre-operative, two months or later in the outpatient clinics, were collected as post-operative. All samples were store at -80oC until analysis.
An aliquot of the stored sera was used for the SELDI-TOF MS analysis. In our study, every ProteinChip were tested, however, a cationic exchanger (WCX2) were
chosen for the analysis. TOF mass spectra were generated in a Ciphergen Protein Biology System II by averaging 56 laser shot per spot with an intensity of 115 to 145 and detector sensitivity to 8. All spectra were compiled to be analyzed, and qualified mass peaks (signal-to-noise ratio >20) with mass-to-charge ratios (m/z) between 2000 and 20000 were auto-detected. Peak clusters were completed using second pass peak section (signal-to-noise ratio >2, within 0.3% mass window), and statistical estimated peaks were added. All these were performed using ProteinChip Software 3.0.1
(Ciphergen).
Serum from eight metastatic RCC patients’and 16 non-RCC patients were collected and subjected the same experimental procedure as the model samples.
Results:
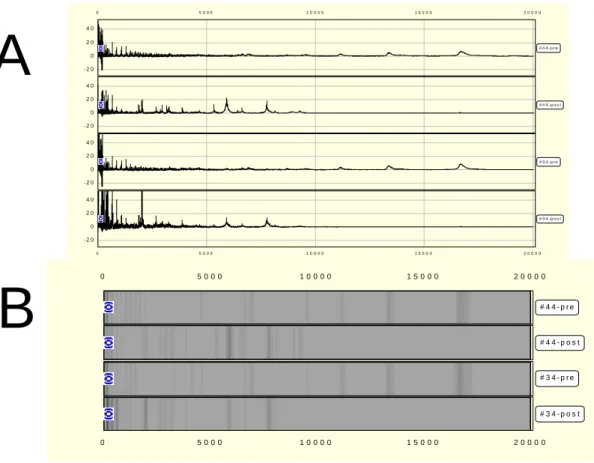
We obtained mass spectra for a total of 72 serum samples from SELDI analysis using WCX2 arrays. Figure 2 shows representative spectra of proteins retained on the WCX2 protein chips. The Mass records can be expressed by trace view (Figure 2A) or gel view (Figure 2B). This model samples was analyzed with BioWizard software (Ciphergen) and showed 5 clusters with statistical significance. It was noted that 16.7 kDa and 13.4 kDa peaks, prominent in pre-operative sera, significantly decreased in post-operative sera. On the contrary, lower molecular weight peaks of 7.7 kDa, 5.9 kDa, and 2.0 kDa were prominent in post-operative but very few in the pre-operative sera (Figure 3A).
When compare non-RCC patients sera with the model samples, they had very similar profile with the post-operative sera (Figure 3B). Since the early stage of RCC tumor mass were removed by surgery, post-operative profile should represent tumor free status. We pooled the non-RCC patients’profileto themodelsamplesand analyzed again, the non-RCC profiles closely resemble those post-operative in model samples (Figure 3B).
The purification and identification of these polypeptides are in progress.
Discussion:
The reproducibility of the SELDI spectra on a single chip (intra-assay) and between chips (inter-assay) was reported by Adam et al (2002). They used seven proteins in the range of 3-10 kDa to calculate the coefficient of variance for a pooled normal serum sample. The peak location was 0.02% and 0.03% for intra-assay and inter-assay respectively, and the intra- and inter-assay normalized intensity (peak height) was 12.1% and 20.5% respectively. SELDI seems to be a good screening tool rather than a quantitative instrument.
relationship imply strong co-relation with RCC. The higher molecular weight 16.7 kDa and 13.4 kDa clusters may represent tumor associated proteins. However, the lower molecular weight 7.7 kDa, 5.9 kDa, and 2.0 kDa may represent proteins that has been suppressed by the tumor. Although non-RCC patients also expressed these polypeptides, they have the same profile as those of post-operative sera. The results suggestavaluablecriteriato differentiateRCC in patient’sserum.
The metastatic RCC sera showed no different profile between pre- or
post-operative samples (data not shown), and the profile similar to post-operative of RCC patients’.Additionalmarker(s)between non-RCC, post-operative, and
metastatic serum are under investigation.
Figure 1 . (A) Various coatings of ProteinChip array are available for sample preparation. The chemically modified surfaces are used to retain proteins based on their specific physical properties. (B) Schematic diagram of the SELDI Ciphergen mass spectrometer.
Acknowledgement: This project was supported by National Science Council, Taiwan, ROC (NSC92-2314-B-002-337). SELDI-TOF analyses were performed by the Proteomics & Protein Function Core lab. located at the Center for Genomic Medicine, National Taiwan University, supported by Promotion of Research-oriented university program from the Ministry of Education.
A
Figure 2: Representative SELDI-tof output of two patients serum with WCX2 ProteinChip in trace view format (A) and gel view format (B).
Figure 3: (A) BioWizard software shows significant profiles of 5 clusters of polypeptides
with m/z of 16.7K, 13.4K, 7.7K, 5.9K, and 2.0K. Two reciprocal pre- and post-operative
serum display very distinctive profile (B) Pooling of non-RCC patient’sserum results reveal
similar profile as those of post-operative group.
0 5 0 0 0 1 0 0 0 0 1 5 0 0 0 2 0 0 0 0 0 5 0 0 0 1 0 0 0 0 1 5 0 0 0 2 0 0 0 0 # 4 4 - p r e # 4 4 - p o s t # 3 4 - p r e # 3 4 - p o s t - 2 0 0 2 0 4 0 - 2 0 0 2 0 4 0 - 2 0 0 2 0 4 0 - 2 0 0 2 0 4 0 0 5 0 0 0 1 0 0 0 0 1 5 0 0 0 2 0 0 0 0 0 5 0 0 0 1 0 0 0 0 1 5 0 0 0 2 0 0 0 0 # 4 4 - p r e # 4 4 - p o s t # 3 4 - p r e # 3 4 - p o s t
A
B
-4 -2 2 4 2000 4000 6000 8000 10000 12000 14000 16000 18000 20000 Log Normalized IntensityM/Z post pre -5 2000 4000 6000 8000 10000 12000 14000 16000 18000 20000 LogNormalizedIntensity M/Z non-RCC post pre
B
A
References:
Appalaneni V, Yellinedi S, Baumann MA. Diagnosis of malignant ascites in prostate cancer by measurement of prostate specific antigen. American Journal of the Medical Sciences 2004, 327, 262-3.
Kaufman H, Schlom J, Kantor J. A recombinant vaccinia virus expressing human carcinoembryonic antigen (CEA). International Journal of Cancer 1991, 48, 900-7
Kozak KR, Amneus MW, Pusey SM, Su F, Luong MN, Luong SA, Reddy ST, and Farias-Eisner R. Identification of biomarkers for ovarian cancer using strong
anion-exchange ProteinChips: Potential use in diagnosis and prognosis. PNAS 2003, 100(21), 12343-8
Markman M, Webster K, Zanotti K, Peterson G, Kulp B, Belinson J. Examples of the marked variability in the relationship between the serum CA-125 antigen level and cancer-related symptoms in ovarian cancer. Gynecologic Oncology 2004, 93, 715-7
Nomura F, Tomonaga T, Sogawa K, Ohashi T, Nezu M, Sunaga M, Kondo N, Iyo M, Shimada H, and Ochiai T. Identification of novel and downregulated biomarkers for alcoholism by surface enhanced laser desorption/ionization-mass spectrometry. Proteomics 2004, 4, 1187-94
Qu Y, Adam BL, Yasui Y, Ward MD, Cazares LH, Schellhammer PF, Feng Z, Semmes OJ, and Wright Jr.GL. Boosted decision tree analysis of surface-enhanced laser desorption/ionization mass spectral serum profiles discriminates prostate cancer from noncancer patients. Clinical Chemistry 2002, 48, 1835-43.
Rosty C, Christa L, Kuzdzal S, Baldwin WM, Zahurak ML, Carnot F, Chan DW, Canto M, Lillemoe KD, Cameron JL, Yeo CJ, Hruban RH, Goggins M. Identification of hepatocarcinoma-intestine-pancreas/pancreatitis-associated protein I as a biomarker for pancreatic ductal adenocarcinoma by protein biochip technology. Cancer Research 2002, 62, 1868-75
Vlahou A, Schellhammer PF, Mendrinos S, Patel K, Kondylis FI, Gong L, Nasim S, Wright Jr GL. Development of a novel proteomic approach for the detection of transitional cell carcinoma of the bladder in urine. American Journal of Pathology 2001, 158, 1491-502.