Research Express@NCKU - Articles Digest
Research Express@NCKU Volume 11 Issue 9 - December 11, 2009 [ http://research.ncku.edu.tw/re/articles/e/20091211/4.html ]
Long-term efficacy and safety of adefovir dipivoxil for
the treatment of hepatitis B e antigen-positive chronic
hepatitis B
Ting-Tsung Chang
Department of Medicine, College of Medicine, National Cheng Kung University ttchang@mail.ncku.edu.tw
Hepatology, 2008, 48:750-8.
A
n estimated 400 million people worldwide are chronically infected with hepatitis B virus (HBV). More than one-half of reported cases with hepatocellular carcinoma (HCC) and approximately one-third of reported cases of cirrhosis are caused by chronic hepatitis B. HCC and cirrhosis, in turn, result in approximately one million deaths each year. Chronic hepatitis B (CHB) patients with an elevated viral load have the highestrisk of progressing to these life-threatening complications. To avoid or minimize liver disease progression, current CHB treatment recommendations now stress the importance of long-term maintenance of HBV DNA suppression. Long-term safety and efficacy data for antiviral medications are indispensable to guide the risk-benefit assessment for a patient.
Approved agents available for the treatment of CHB can be divided into two groups: interferons and nucleoside/ nucleotide analogues (NAs). Treatment with standard or pegylated interferon has been shown to result in durable serologic responses (HBe seroconversion) in HBeAg-positive patients, but these therapies are limited by response (seroconversion) rates of between 20 and 40%, injectable administration, and a high incidence of adverse events. NAs include lamivudine, adefovir dipivoxil (ADV), entecavir, telbivudine, and tenofovir. Lamivudine and telbivudine have demonstrated efficacy and long-term safety, but the benefits of treatment diminish as resistance after long term therapy. The most recently published CHB treatment recommendations now classify lamivudine and telbivudine as not preferred due to its high rate of resistance. Published data on the long-term efficacy and safety of adefovir and entecavir in HBeAg-positive patients are limited.
The current report presents efficacy results through week 240 for all patients who received ADV 10 mg during year 1 of the study, and safety results for the subset of patients who were enrolled in the long-term safety and efficacy study (LTSES) and received up to 5 years of ADV. However, during year 2, study medication had been misallocated during the second year of the study due to an error in the interactive voice response system used to assign blinded treatment. Sixty-five patients given ADV in year 1 elected to continue in a long-term safety and efficacy study (LTSES). At enrollment, the 65 LTSES patients were a median 34 years old, 83% male, 74% Asian, 23% Caucasian, median baseline serum hepatitis B virus (HBV) DNA 8.45 log10 copies/mL, and median baseline alanine aminotransferase
(ALT) 2.0 x upper limit of normal (ULN). Virological Response (figure 1)
The median change from baseline in serum HBV DNA for these patients who received adefovir at study weeks 48, 144, 192, and 240 were -3.44, -3.69, -3.55, and -4.05 log10copies/mL, respectively. 39% had serum HBV DNA < 1000 copies/mL at study week 240.
Research Express@NCKU - Articles Digest
Figure 1. Median serum HBV DNA concentration by study visit.
Figure 2. Median serum ALT concentration by study visit.
Biochemical Response (figure 2)
The median change from baseline in serum ALT concentrations for these patients who received adefovir at study weeks 48, 96, 144, 192, and 240 were -43, -18, -49.5, -41, and -50 IU/L, respectively; 66% had normalized (ULN) serum ALT concentrations at study week 240.
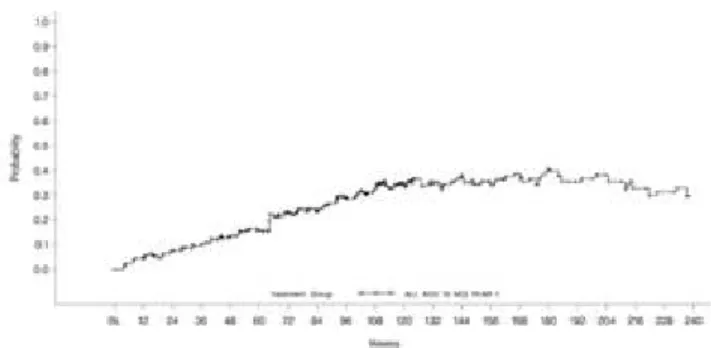
Figure 3. Plot of probability of HBeAg seroconversion by two-state Markov model
Serological Response (figure 3)
The proportion of patients with e-seroconversion increased steadily from baseline with the estimated Markov probability values of HBeAg seroconversion, 13%, 29%, 37%, 35%, and 30% for weeks 48, 96, 144, 192, and 240, respectively.
Fifteen patients had baseline and end of follow-up liver biopsies; improvements in necroinflammation and fibrosis were seen in 67% and 60% of these patients, respectively. Adefovir-associated substitutions, A181V
or N236T, were detected in 16 of 38 (42%) patients in the setting of virologic failure (confirmed increase of ≥1 log10
HBV DNA copies/mL above nadir or never suppressed below 103 copies/mL) There were no serious adverse events
related to ADV. In summary, the results of this long-term cohort study show that among HBeAg-positive patients, long term therapy with ADV achieves and maintains virological, biochemical, serological, and histological improvement. ADV was also well-tolerated.
References:
CHANG TT, Suh DJ. 2008 Current approaches for treating chronic hepatitis B: when to start, what to start with, and when to stop. Hepatology International 2:S19-27