Date 2015/September/21
Type of manuscript: Original article
Manuscript title: Sitagliptin use and risk of acute pancreatitis in type 2 diabetes mellitus: a population-based case-control study in Taiwan
Running head: sitagliptin and acute pancreatitis Authors' full names:
Kuan-Fu Liao MD and MS
1,2,3, Cheng-Li Lin MS
4,5, Shih-Wei Lai MD
4,6, Wen-Chi Chen MD and PhD
3,71
College of Medicine, Tzu Chi University, Hualien, Taiwan
2
Department of Internal Medicine, Taichung Tzu Chi General Hospital, Taichung, Taiwan
3
Graduate Institute of Integrated Medicine, China Medical University, Taichung, Taiwan
4
College of Medicine, China Medical University, Taichung, Taiwan
5
Management Office for Health Data, China Medical University Hospital, Taichung, Taiwan
6
Department of Family Medicine, China Medical University Hospital, Taichung, Taiwan
7
Department of Urology, China Medical University Hospital, Taichung, Taiwan
Corresponding author: Shih-Wei Lai, Department of Family Medicine, China Medical University Hospital, No 2, Yuh-Der Road, Taichung City, 404, Taiwan
Phone: 886-4-2205-2121; Fax: 886-4-2203-3986 E-mail: wei@mail.cmuh.org.tw
Word count: 269 in abstract, 2028 in text, 2 tables, and 28 references
ABSTRACT
Background. There is still lack of definite evidence to establish the association between sitagliptin use and acute pancreatitis. The study aimed to test this issue in Taiwan. Methods. This case-control study was designed to analyze the database of the Taiwan National Health Insurance Program. There were 349 subjects with type 2 diabetes mellitus aged 20-84 with a first-attack of acute pancreatitis from 2009 to 2011 as the case group and 1116 randomly selected subjects with type 2 diabetes mellitus without acute pancreatitis as the control group. Both groups were matched with sex, age, comorbidities, and index year of diagnosing acute pancreatitis. Current use of sitagliptin was defined as subjects who had their last tablet of sitagliptin ≤ 7 days before the date of diagnosing acute pancreatitis. Late use of sitagliptin was defined as subjects who had their last tablet of sitagliptin between 8 to 30 days before the date of diagnosing acute pancreatitis. Never use of sitagliptin was defined as subjects who never had a sitagliptin prescription. The risk of acute pancreatitis associated with sitagliptin use was estimated by the odds ratio (OR) and 95%
confidence interval (CI) using the multivariable logistic regression model. Results.
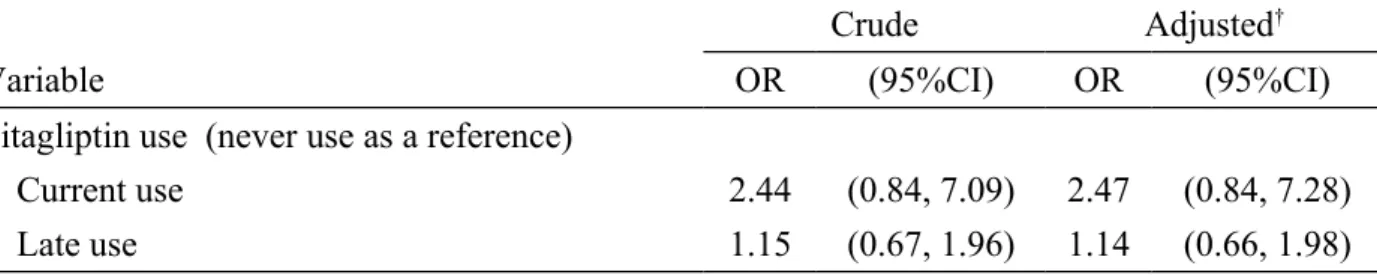
After statistical correction for potential confounders, the adjusted OR of acute pancreatitis was 2.47 for subjects with current use of sitagliptin (95% CI 0.84, 7.28), when compared with those never using sitagliptin, but without statistical significance.
The adjusted OR decreased to 1.14 for subjects with late use of sitagliptin (95% CI 0.66, 1.98), but without statistical significance. Conclusions. No significant
association is detected between sitagliptin use and acute pancreatitis in type 2 diabetes mellitus.
Keywords: acute pancreatitis; sitagliptin; Taiwan National Health Insurance
Program
INTRODUCTION
Sitagliptin is one of dipeptidyl peptidase-4 inhibitors, also called DPP-4 inhibitors, which are classified as novel oral anti-hyperglycemic agents and are commonly used to treat type 2 diabetes mellitus. Overall, sitagliptin is generally well tolerated and relatively effective and safe in patients with type 2 diabetes mellitus. However, the commonly seen adverse events of sitagliptin mainly include infections, gastro-
intestinal disorders, musculoskeletal and connective tissue disorders, angioedema and skin disorders. In addition, DPP-4 inhibitors-related acute pancreatitis has raised public health concern. Case report and adverse drug event are usually used to establish a plausible hypothesis linking the offending drug and the specific adverse effect. Till now, only few cases of acute pancreatitis were reported to be possibly related to sitagliptin use. The U.S. Food and Drug Administration (FDA) has reported that 21 people (2.44%) had acute pancreatitis among 860 people reporting to have side effects when taking sitagliptin since 2009 to 2012.
5To date, no definite conclusion is established to confirm the association between sitagliptin use and acute pancreatitis. Given acute pancreatitis having considerable morbidity and mortality, and based on the aforementioned case reports and the U.S.
FDA report, we make a plausible hypothesis that sitagliptin use may increase the risk of acute pancreatitis. If the association between sitagliptin use and acute pancreatitis can be established, physicians should take this risk into account. Therefore, we conducted a population-based case-control study using the database of the Taiwan National Health Insurance Program to examine this issue.
METHODS
Design and data source
This population-based case-control study was designed to analyze the database of
the Taiwan National Health Insurance Program. Briefly speaking, this program began in March 1, 1995, which covered almost 99% of the whole 23 million residents living in Taiwan.
6The details of the program were adequately described in previous high- quality papers.
7-9This study was approved by the Ethics Review Board of China Medical University and Hospital in Taiwan (CMUH-104-REC2-115).
Selection of cases and controls
On the basis of the International Classification of Diseases (ICD) 9th Revision, we identified subjects with type 2 diabetes mellitus aged 20-84 years with a first-attack of acute pancreatitis during the period of 2009-2011 as the case group (ICD-9 code 577.0). Subjects with type 2 diabetes mellitus without acute pancreatitis were
randomly selected from the same database as the control group. Both case and control groups were matched with sex, age (5-year interval), comorbidities, and the index year of diagnosing acute pancreatitis. The index date for each case was defined as the date of diagnosing acute pancreatitis. The index date for each control subject was a randomly assigned date within the index year of the matched case subject. To decrease biased results, subjects with chronic pancreatitis (ICD-9 code 577.1) or pancreatic cancer (ICD-9 code 157) before the index date were excluded from this study. To focus on the association between sitagliptin use only and acute pancreatitis, subjects who at least received 1 prescription for other DPP-4 inhibitors, including vildagliptin, saxagliptin and linagliptin, were also excluded from this study.
Definition of sitagliptin exposure
Based on the prescription date of each subject, the last tablet of sitagliptin can be calculated. To decrease biased results, subjects who had their last tablet of sitagliptin
> 1 month before the date of diagnosing acute pancreatitis were excluded from this
study. Therefore, only those who had their last tablet of sitagliptin within 1 month
before the date of diagnosing acute pancreatitis were included in this study. The elimination half-life of sitagliptin ranges from 8 to 14 hours in subjects with normal renal function.
10The elimination half-life increases with decreasing renal function and ranges from 16.1 to 28.4 hours in subjects with impaired renal function,
depending on the renal function preserved.
11Therefore, we used the period of 7 days as a cut-off point. Current use of sitagliptin was defined as subjects who had their last tablet of sitagliptin ≤ 7 days before the date of diagnosing acute pancreatitis or those who still had sitagliptin tablets remaining at the date of diagnosing acute pancreatitis.
Late use of sitagliptin was defined as subjects who had their last tablet of sitagliptin between 8 to 30 days before the date of diagnosing acute pancreatitis. Never use of sitagliptin was defined as subjects who never had a sitagliptin prescription.
Potential confounders
In order to control for confounding effects, comorbidities before the index date potentially related to acute pancreatitis were included as follows based on ICD-9 codes: alcohol-related disease (ICD-9 codes 291, 303, 305.00, 305.01, 305.02, 305.03, 571.0-571.3, 790.3 and V11.3), biliary stone (ICD-9 code 574), cardiovascular
disease including coronary artery disease, heart failure, cerebrovascular disease and
peripheral atherosclerosis (ICD-9 codes 410–414, 428, 430–438 and 440–448),
chronic kidney disease (ICD-9 codes 585–586 and 588.8–588.9), chronic obstructive
pulmonary disease (ICD-9 codes 491, 492, 493 and 496), hepatitis B (ICD-9 codes
V02.61, 070.20, 070.22, 070.30 and 070.32), hepatitis C (ICD-9 codes V02.62,
070.41, 070.44, 070.51 and 070.54), hyperparathyroidism (ICD-9 code 252.0), and
hypertriglyceridemia (ICD-9 codes 272.1, 272.2 and 272.4). In order to avoid subjects
who were mistakenly diagnosed or mistakenly coded by accident, only those who had
at least 2 consensus same diagnoses in the ambulatory care or at least 1 hospitalization
diagnosis were included to make sure the diagnosis validity of acute pancreatitis and other comorbidities.
Statistical analysis
We compared the differences in demographic status, sitagliptin use, and comorbidities between the acute pancreatitis cases and the controls using the Chi- square test and Fisher-exact test for categorized variables, and t-test for continuous variables. Initially, all variables were included in the univariable unconditional logistic regression model. Only those found to be significant in the univariable unconditional logistic regression model were further analyzed in the multivariable unconditional logistic regression model to estimate the odds ratio (OR) and 95%
confidence interval (CI) for risk of acute pancreatitis associated with sitagliptin use and comorbidities. All statistical analyses were performed using SAS 9.2 (SAS Institute, Cary, North Carolina, USA). Two-tailed p < 0.05 was considered statistically significant.
RESULTS
Informative profiles of the study population
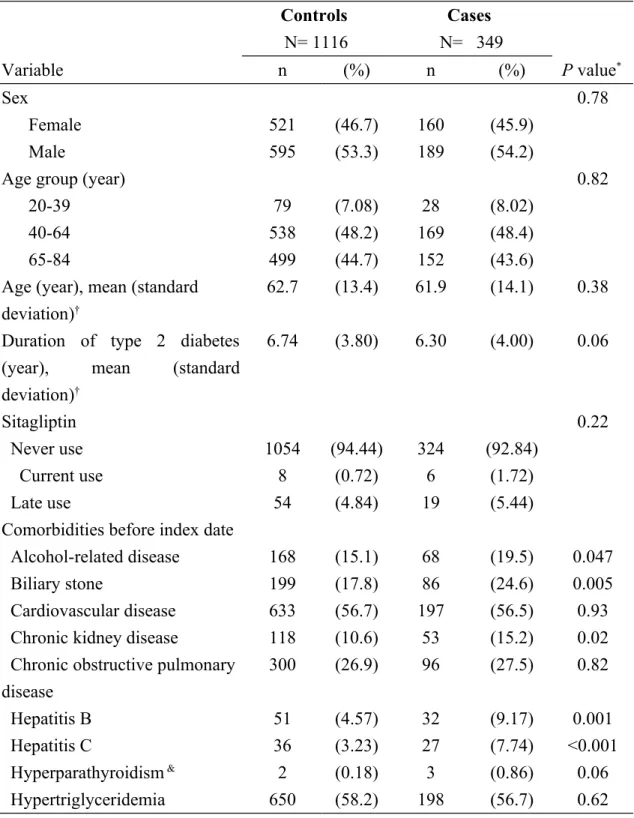
Table 1 discloses the informative profiles between the acute pancreatitis cases and the controls. In total, the study consisted of 349 subjects with acute pancreatitis as the cases and 1116 subjects without acute pancreatitis as the controls, with similar
distributions in sex, age and duration of type 2 diabetes mellitus. The mean ages
(standard deviation) were 61.9 (14.1) years in cases and 62.7 (13.4) years in controls
(t-test, P= 0.38). The mean duration of type 2 diabetes (standard deviation) were
6.30(4.00) years in cases and 6.74 (3.80) years in controls (t-test, P= 0.06). The cases
were more likely to have higher proportions of current use of sitagliptin and late use
of sitagliptin than the controls, but without statistical significance (1.72% vs. 0.72%
for current use, and 5.44% vs. 4.84% for late use, respectively, Chi-square test, P = 0.22). There were significantly higher proportions of alcohol-related disease, biliary stone, chronic kidney disease, hepatitis B, and hepatitis C in the cases than the controls (Chi-square test, P < 0.05 for all).
Acute pancreatitis associated with sitagliptin use
After adjusting for variables that were significantly related to acute pancreatitis in the univariable analysis, the multivariable logistic regression model disclosed that the adjusted OR of acute pancreatitis was 2.47 for subjects with current use of sitagliptin (95% CI 0.84, 7.28), when compared with those never using sitagliptin, but without statistical significance. The adjusted OR decreased to 1.14 for subjects with late use of sitagliptin (95% CI 0.66, 1.98), but without statistical significance.
DISCUSSION
After statistical correction for potential confounders, we noted that no significant association could be detected between sitagliptin use and acute pancreatitis, no matter current use or late use. Although this is a clinically relevant question, the literature disclosed conflicting results on the association between sitagliptin use and acute pancreatitis. To date, only few cases of acute pancreatitis were reported to be possibly related to sitagliptin use, but these cases did not have re-exposure confirmation of a causal relationship due to discontinuation of sitagliptin use or case death. In addition, The U.S. FDA mentioned that 21 people (2.44%) had acute pancreatitis events among 860 people reporting to have side effects when taking sitagliptin since 2009 to 2012, but similarly no causal relationship was confirmed.
5Several systematic studies yielded no significantly increased risk of acute pancreatitis among patients with type 2 diabetes mellitus treated with sitagliptin,
12-19which was compatible with our finding.
Recently, one study focusing on sitagliptin effect on cardiovascular outcomes in type
2 diabetes by Green et al disclosed that although the events of acute pancreatitis were
more commonly found in the sitagliptin group than that in the placebo group (23 vs.
12 events), this difference was not statistically significant.
20To the contrary, only one case-control study in the United States disclosed that patients with type 2 diabetes mellitus treated with the glucagon-like peptide 1–based therapies sitagliptin and exenatide were associated with increased odds of hospitalization for acute
pancreatitis,
21but no data was presented for a separate analysis of sitagliptin alone.
Based on the above-mentioned literature review, although the majority of available studies disclose no association between sitagliptin use and acute pancreatitis, no definite conclusion is established on the issue. More carefully designed prospective research is needed to elucidate this issue.
Some imitations of this study need to be discussed. First, due to intrinsic limitation of this database, we could not affirm whether or not patients really ingested
sitagliptin, but we included sitagliptin prescription for instead. Of course, there is the general observation that getting a prescription is not the same as ingesting it. Second, some behavior risk factors of acute pancreatitis, such as alcoholism and smoking, were not recorded in this database due to the same limitation, but we used alcohol- related disease instead of alcoholism and used chronic obstructive pulmonary disease instead of smoking. Similarly, obesity was not recorded in this database due to the same limitation. This limitation cannot be easily overcome. Third, since sitagliptin is a novel oral anti-hyperglycemic agent which is covered by the Taiwan National Health Insurance Program since 2009, the number of sitagliptin users remained small.
In addition, the database used only included the period up to 2011. That is why we
selected subjects only from 2009 to 2011. To decrease biased results, subjects who
had their last tablet of sitagliptin > 1 month before the date of diagnosing acute
pancreatitis were also excluded from this study. Due to the aforementioned reasons
and the strict inclusion criterion, that is why there were only 6 subjects with current use of sitagliptin and 19 subjects with late use of sitagliptin in the acute pancreatitis group. The relatively small number of cases remains an issue. However, this is not a solvable problem without including other databases. It may be resolved by gathering more data in the database of the Taiwan National Health Insurance Program. Fourth, data on some important potential confounders were not present, such as other
medications used and the procedure of iatrogenic endoscopic retrograde
cholangiopancreatography (ERCP). There are more than 500 drugs listed in the database of World Health Organization (WHO) to be related to acute pancreatitis.
22It seems to be relatively difficult to select all potential drugs for analysis. In addition, because of no detailed records in this database, there is no knowing that patients studied were admitted for undergoing therapeutic ERCP to treat biliary-related diseases or were admitted for other diseases but complicated by iatrogenic ERCP.
Therefore, the cause-effect association between ERCP and acute pancreatitis cannot be entirely disclosed in this study.
In spite of these limitations, some strengths of this study should be described. This is a topic of great importance. The design adequately addresses the question regarding sitagliptin use and acute pancreatitis. The methodology of this study is scientifically correct. Conclusion seems to be straightforward. The extensiveness and the ability to answer many clinical questions of the Taiwan database has been well reviewed by Hsing et al.
25There are several published studies of high quality examining the similar research question. The diagnosis accuracy of comorbidities included has been well reviewed in these studies.
26-28CONCLUSION
We conclude that no significant association is detected between sitagliptin use and acute pancreatitis among patients with type 2 diabetes mellitus. Based on
aforementioned discussion, more long-term prospective studies with large sample size are warranted to definitively resolve the uncertain questions.
Acknowledgement
This study is supported in part by Taiwan Ministry of Health and Welfare Clinical Trial and Research Center of Excellence (MOHW104-TDU-B-212-113002), China Medical University Hospital, Academia Sinica Taiwan Biobank, Stroke Biosignature Project (BM104010092), National Research Program for Biopharmaceuticals (NRPB) Stroke Clinical Trial Consortium (MOST 103-2325-B-039 -006), Tseng-Lien Lin Foundation in Taichung in Taiwan, Taiwan Brain Disease Foundation in Taipei in Taiwan, and Katsuzo and Kiyo Aoshima Memorial Funds in Japan. These funding agencies did not influence the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Specific author contributions
Kuan-Fu Liao and Shih-Wei Lai substantially contributed to the conception of the article. They planned and conducted this study. They initiated the draft of the article and critically revised the article.
Cheng-Li Lin and Wen-Chi Chen conducted the data analysis and critically revised the article.
Conflict of Interest Statement
The authors declare that they have no conflict of interest.
REFERENCES
1. Williams-Herman D, Round E, Swern AS, et al. Safety and tolerability of sitagliptin in patients with type 2 diabetes: a pooled analysis. BMC Endocr Disord 2008;8:14.
2. Karagiannis T, Boura P, Tsapas A. Safety of dipeptidyl peptidase 4 inhibitors: a perspective review. Ther Adv Drug Saf 2014;5:138-46.
3. Tsai L-Y, Ma W-Y, Hsia T-L, Pei D, Su C-C. Sitagliptin-asso-ciated acute pancreatitis: A case report. J Intern Med Taiwan 2011;22:278-82.
4. Iyer SN, Drake AJ, 3rd, West RL, Mendez CE, Tanenberg RJ. Case report of acute necrotizing pancreatitis associated with combination treatment of sitagliptin and exenatide. Endocr Pract 2012;18:e10-3.
5. eHealthMe study from FDA and social media reports. Review: could sitagliptin phosphate cause acute pancreatitis? http://www.ehealthme.com/print/ds48406887 [cited in 2014 november].
6. National Health Insurance Research Database. Taiwan.
http://nhird.nhri.org.tw/en/index.html [cited in 2015 September] [English version].
7. Lai SW, Liao KF, Liao CC, Muo CH, Liu CS, Sung FC. Polypharmacy
correlates with increased risk for hip fracture in the elderly: a population-based study.
Medicine (Baltimore) 2010;89:295-9.
8. Lai SW, Chen PC, Liao KF, Muo CH, Lin CC, Sung FC. Risk of hepatocellular carcinoma in diabetic patients and risk reduction associated with anti-diabetic therapy:
a population-based cohort study. Am J Gastroenterol 2012;107:46-52.
9. Liao KF, Lai SW, Li CI, Chen WC. Diabetes mellitus correlates with increased risk of pancreatic cancer: a population-based cohort study in Taiwan. J Gastroenterol Hepatol 2012;27:709-13.
10. Herman GA, Stevens C, Van Dyck K, et al. Pharmacokinetics and
pharmacodynamics of sitagliptin, an inhibitor of dipeptidyl peptidase IV, in healthy subjects: results from two randomized, double-blind, placebo-controlled studies with single oral doses. Clin Pharmacol Ther 2005;78:675-88.
11. Scheen AJ. Pharmacokinetics of dipeptidylpeptidase-4 inhibitors. Diabetes Obes Metab 2010;12:648-58.
12. Dore DD, Seeger JD, Arnold Chan K. Use of a claims-based active drug safety surveillance system to assess the risk of acute pancreatitis with exenatide or sitagliptin compared to metformin or glyburide. Curr Med Res Opin 2009;25:1019-27.
13. Garg R, Chen W, Pendergrass M. Acute pancreatitis in type 2 diabetes treated with exenatide or sitagliptin: a retrospective observational pharmacy claims analysis.
Diabetes Care 2010;33:2349-54.
14. Engel SS, Williams-Herman DE, Golm GT, et al. Sitagliptin: review of preclinical and clinical data regarding incidence of pancreatitis. Int J Clin Pract 2010;64:984-90.
15. Eurich DT, Simpson S, Senthilselvan A, Asche CV, Sandhu-Minhas JK, McAlister FA. Comparative safety and effectiveness of sitagliptin in patients with type 2 diabetes: retrospective population based cohort study. BMJ 2013;346:f2267.
16. Faillie JL, Azoulay L, Patenaude V, Hillaire-Buys D, Suissa S. Incretin based drugs and risk of acute pancreatitis in patients with type 2 diabetes: cohort study. BMJ 2014;348:g2780.
17. Giorda CB, Picariello R, Nada E, et al. Incretin therapies and risk of hospital admission for acute pancreatitis in an unselected population of European patients with type 2 diabetes: a case-control study. Lancet Diabetes Endocrinol 2014;2:111-5.
18. Li L, Shen J, Bala MM, et al. Incretin treatment and risk of pancreatitis in patients with type 2 diabetes mellitus: systematic review and meta-analysis of randomised and non-randomised studies. BMJ 2014;348:g2366.
19. Monami M, Dicembrini I, Mannucci E. Dipeptidyl peptidase-4 inhibitors and pancreatitis risk: a meta-analysis of randomized clinical trials. Diabetes Obes Metab 2014;16:48-56.
20. Green JB, Bethel MA, Armstrong PW, et al. Effect of Sitagliptin on Cardiovascular Outcomes in Type 2 Diabetes. N Engl J Med 2015;373:232-42.
21. Singh S, Chang HY, Richards TM, Weiner JP, Clark JM, Segal JB. Glucagonlike peptide 1-based therapies and risk of hospitalization for acute pancreatitis in type 2 diabetes mellitus: a population-based matched case-control study. JAMA Intern Med 2013;173:534-9.
22. Nitsche C, Maertin S, Scheiber J, Ritter CA, Lerch MM, Mayerle J. Drug- induced pancreatitis. Curr Gastroenterol Rep 2012;14:131-8.
23. Lai SW, Liao KF, Lin CL, Chen PC. Pyogenic liver abscess correlates with increased risk of acute pancreatitis: a population-based cohort study. J Epidemiol 2015;25:246-53.
24. Lai SW, Lai HC, Lin CL, Liao KF, Tseng CH. Chronic osteomyelitis correlates with increased risk of acute pancreatitis in a case-control study in Taiwan. Eur J Intern Med 2015;26:429-32.
25. Hsing AW, Ioannidis JP. Nationwide Population Science: Lessons From the Taiwan National Health Insurance Research Database. JAMA Intern Med
2015;175:1527-9.
26. Lai SW, Muo CH, Liao KF, Sung FC, Chen PC. Risk of acute pancreatitis in
type 2 diabetes and risk reduction on anti-diabetic drugs: a population-based cohort
27. Lai SW, Lin CL, Liao KF, Lin CY. Amiodarone use and risk of acute
pancreatitis: A population-based case-control study. Heart Rhythm 2015;12:163-6.
28. Lai SW, Lin CL, Liao KF. Actively using clopidogrel correlates with an
increased risk of acute pancreatitis in Taiwan. Int J Cardiol 2015;183:263-6.
Table 1. Informative profiles of acute pancreatitis cases and controls Controls
N= 1116
Cases N= 349
Variable n (%) n (%) P value
*Sex 0.78
Female 521 (46.7) 160 (45.9)
Male 595 (53.3) 189 (54.2)
Age group (year) 0.82
20-39 79 (7.08) 28 (8.02)
40-64 538 (48.2) 169 (48.4)
65-84 499 (44.7) 152 (43.6)
Age (year), mean (standard deviation)
†62.7 (13.4) 61.9 (14.1) 0.38
Duration of type 2 diabetes (year), mean (standard deviation)
†6.74 (3.80) 6.30 (4.00) 0.06
Sitagliptin 0.22
Never use 1054 (94.44) 324 (92.84)
Current use 8 (0.72) 6 (1.72)
Late use 54 (4.84) 19 (5.44)
Comorbidities before index date
Alcohol-related disease 168 (15.1) 68 (19.5) 0.047
Biliary stone 199 (17.8) 86 (24.6) 0.005
Cardiovascular disease 633 (56.7) 197 (56.5) 0.93
Chronic kidney disease 118 (10.6) 53 (15.2) 0.02
Chronic obstructive pulmonary disease
300 (26.9) 96 (27.5) 0.82
Hepatitis B 51 (4.57) 32 (9.17) 0.001
Hepatitis C 36 (3.23) 27 (7.74) <0.001
Hyperparathyroidism
&2 (0.18) 3 (0.86) 0.06
Hypertriglyceridemia 650 (58.2) 198 (56.7) 0.62
Data are presented as the number of subjects in each group, with percentages given in parentheses or mean with standard deviation given in parentheses.
*
Chi-square test,
&Fisher-exact test and
†t-test comparing subjects with and without
acute pancreatitis
Table 2. Acute pancreatitis associated with sitagliptin use estimated by odds ratio and 95%
confidence interval
Crude Adjusted
†Variable OR (95%CI) OR (95%CI)
Sitagliptin use (never use as a reference)
Current use 2.44 (0.84, 7.09) 2.47 (0.84, 7.28)
Late use 1.15 (0.67, 1.96) 1.14 (0.66, 1.98)
†