1 Department of Internal Medicine, Taipei Medical University-Municipal Wan Fang Hospital, Taipei, Taiwan
2 Department of Neurosurgery, Taipei Medical University-Shuang Ho Hospital, Taipei, Taiwan
3 Department of Internal Medicine, Taipei Medical University-Shuang Ho Hospial, Taipei, Taiwan
4 School of Respiratory Therapy, Taipei Medical University
Correspondence to Ling-Ling Chiang, School of Respiratory Therapy, Taipei Medical University, 250 Wu-Xin Street, Taipei 110, Taiwan.
TEL: 27361661 ext 3512 E-mail: llchiang@tmu.edu.tw
Received: April 16, 2008 Revised: July 3, 2008 Accepted: July 17, 2008
The Experience in Respiratory Care of a Patient with Cervical Spinal Cord Injury
Shiauyee Chen
1Chien-Min Lin
2Chun-Nin Lee
3Cho-Yi Huang
1Ling-Ling Chiang
4Patients with high-level cervical spinal cord injury are at high risk of respiratory insufficiency and related complications. Paralysis of respiratory muscles often requires the use of mechani- cal ventilation. In this case, we present an approach to respiratory care for a 66-year-old male patient with C4-C5 spinal cord injury (ASIA-B). A weaning progression from mechanical ventilation was presented through the following programs: nocturnal support, intensive chest care, lung expansion therapy, speaking valve training, and inspiratory muscle training. (FJPT 2008;33(5):294-301)
Key Words: Spinal cord injury, Respiratory care, Speaking valve, Inspirtory muscle training
IntroductIon
Respiratory insufficiency and related complications are the major causes of mortality in patients with high-level spi- nal cord injury (SCI). The rate of respiratory complications in high-level (first cervical spine to fourth cervical spine, C1 to C4) SCI has been reported to be as high as 84%.1,2 For these patients, mechanical ventilation, treatment of pneumonia, use of the tracheostomy, and surgery accounted for nearly 60% of medical costs.2-4 Thus, efforts to improve respiratory function and minimize respiratory complications in these patients are crucial.
The main problems for these high-level SCI patients are
atelectasis, secretions impaction, hypoventilation, and dyspha- gia.5,6 For the respiratory muscle function, the involved main muscles for these patients include the diaphragm (innervated by spinal nerve from C3 to C5) and external intercostal muscles (T1 to T11). Additionally, the involved accessory muscles include the scalene (C4 to C8), sternocleidomastoid and trapezius (C1 to C4 and accessory nerve CNXI), and pectoralis groups (C5 to T1).2 The lesion of a high level SCI results in a restrictive ventilatory deficit and a decrease in lung volume (atelectasis).
Subsequently, the weakness of respiratory muscles and changes in the mechanical properties of the lungs and chest wall lead to paradoxical movement of the chest wall. The inefficiency of breathing and impairment of expiratory muscles also results in
FJPT 2008;33(5):294-301 Chen et al.
an ineffective cough.
It has been reported that in the first five days after injury, the prevalences of atelectaisis, pneumonia, and respiratory fail- ure are 36.4%, 31.4%, and 22.6%, respectively.2 Mechanical ventilator was provided in the initial phase immediately after the cervical spinal cord injury. A retrospective study showed that 36% of 72 patients with traumatic cervical spinal cord injury needed mechanical ventilation for respiratory failure.2 The percentage of ventilator dependence will be lower after the first year of injury because of the spontaneous neurologi- cal recovery.3 Weaning from mechanical ventilator depends on the strength of respiratory muscles, spontaneous neurological recovery, level of injury and other associating factors like copi- ous sputum and pneumonia.2-5 The weaning rate for ventilator- dependent cervical spinal cord injury was about 20% according to the report from James A, Haley Veterans hospital in Tampa ( Florida).8
In this report, we present the case of a patient with high level C4-C5 SCI with resultant ventilator dependence. The noc- turnal ventilatory support (ventilatory support provided only during night–time), use of a speaking valve (a one-way silicone diaphragm check valve that fits the tracheostomy tube), and in- spiratory muscle training (load applied on inspiratory muscle to increase strength and endurance) allowed the patient to achieve independence from the ventilator.
Case summary
The patient, a 66 y/o retired truck driver, lived indepen- dently in Ping-Shi (Taipei County). He spent a lot of time in gardening before injury. He had one son and two daughters.
This patient had a history of DM for 10 years and was not under regular medical control until 2 years ago. He had smoked 1/2 to 1 PPD, but quit smoking over 30 years before. Unfortunately, he fell off a roof (about 2-meter height) and lost consciousness on Aug. 10, 2006. He was sent to University Hospital for emer- gent medical care. Brain CT showed no signs of intracranial hemorrhage, but a cervical-spine plane film revealed a C4-5 fracture dislocation with a C4-5 locked facet and spinal cord compression. Due to the apparent C4-5 dislocation and C5-6 stenosis with positive neurological signs, an emergency opera- tion was performed on the same day. The operation procedure included (1) C4-5, C5-6 diskectomy; (2) C5 corpectomy; (3) C4-C6 interbody bone fusion; and (4) anterior cervical plate
(ACP) fixation. He was diagnosed with C4-C5 spinal cord in- jury (ASIA-B).
After the operation, this case had complications, with re- peated pneumonia, urinary tract infection, and respiratory fail- ure with ventilatory support. Tracheostomy was performed on Sep. 4, 2006 because of impending diaphragm fatigue and re- spiratory failure. Due to general health insurance and payment considerations, the patient was transferred to another hospital for further care on Nov. 18, 2006. On Jan. 16, 2007, after 3 months in that hospital, he was transferred back to our hospital.
He was still dependent on the ventilator because of recurrent bacteremia, restrictive lung disease, atelectaisis, and UTI, ac- cording to his medical records. Respiratory care and weaning were provided since Jan. 16 to Jun. 26, 2007.
On admission to intensive care unit, the patient was on a ventilator via tracheotomy. The ventilator initially was used in assisted control mandatory mode (ACMV), with the tidal vol- ume (Vt) set to achieve 550 ml, a respiratory rate of 10 breaths/
min, a positive end-expiratory pressure (PEEP) with 5 cm H2O, and FiO2 of 0.25. His consciousness was clear, and he had a body mass index of 20.9 kg/m2 (height 162 cm and weight 55 kg). He was afebrile (body temperature 37.3°). He had a heart rate of 87 beats/min and blood pressure of 117/73 mmHg. The hematocrit was 29%, the white blood cell count was 11.03 X 106cells/μL, and the red blood cell count was 3.57 X 106 cells/
μL. Other aspects of blood chemistry were normal except for a high blood urea nitrogen (24 mg/dl). He had a naso-gastric tube and foley on Jan.16, 2007. His chest X-ray is shown Fig.
1. The initial chest X-ray (Jan. 16, 2007) showed infiltration over bilateral lung fields. His muscle power were listed as fol- lows: right elbow flexor (C5): 3, left elbow flexor: 4; right wrist extensor (C6): 2, left wrist extensor: 3; right elbow extensor (C7): 1, and left elbow extensor: 2. His sensation below C5 was impaired. Spasticity was found in his bilateral upper and lower extremities (++).
Ventilator weaning was attempted in our intensive care unit. Pressure support ventilation (PSV) (pressure support 10 cmH2O, tidal volume 425 ml, rate 20 times/min, PEEP 5cm- H2O, and FiO2 of 0.25) was tried first on Jan. 23, 2007. From Jan. 29, 2007 to Feb. 2, 2007, a collar mask (5 l/min) during the daytime and PSV (pressure support 12 cmH2O, PEEP 5cmH2O, and FiO2 of 0.25) for nocturnal support were provided, and the patient was transferred to our regular neurosurgery ward. From
Feb. 3, 2007 to Feb. 9, 2007, oxygen support (5 l/min) was pro- vided at night, and only room air during the day. After Feb 9, all oxygen support was discontinued.
When the program in the ward started on Feb. 9, 2007, the patient’s saturation was 95% (room air), his heart rate was 90 beats/min, and his respiratory rate was 28 breaths/min under room air. His breathing pattern was smooth and symmetrical, with paradoxical abdominal breathing. Breathing sound showed inspiratory crackles over bilateral lower lobes. Our programs included lung expansion therapy (intermittent positive pressure breathing, IPPB, 15 –20 cmH2O, 15 minutes/time, once a day), chest physical therapy (after inhalation with bricanyl 2 ml, postural drainage with modified sidelying position, vibration with percussor, manual hyperinflation and suction, 30 minutes a time, 4 times a day), speaking valve training, and inspiratory muscle training (sandbag over anterior costal margin, starting with weight 1 kg) .
On Feb. 15, 2007, he appeared drowsy and weak during the daytime. In addition to this, we observed shallow breath- ing (36 to 40 times/min) and paradoxical movement. Noctur- nal support for ventilation was suggested. The ventilator was used in synchronized intermittent mandatory ventilation mode
(SIMV), with the tidal volume set at 500ml/min, the respira- tory rate at 6 breath/min, and the pressure support at 12cmH2O and FiO2 of 0.25 during sleep time. This nocturnal support was maintained from Feb. 15, 2007 to Apr. 17, 2007 (except during a Mar. 22 to Mar. 28 operation period for colon carcinoma). He breathed room air during the day and continued with the pro- grams listed above.
We tried a Passy-Muir Speaking Valve (PMV 007, for use with disposable ventilator tubing) after Feb. 9, 2007 (Fig. 2).
The patient had difficulty in speaking while on room air and during cuff deflation. We practiced 5 to 10 min every day after Fig 1. Initial X-ray (AP) showed infiltration involving right middle zone, bilateral basal
area, and greater severity on left lower lung field on Jan. 16, 2007.
Fig 2. Passy-Muir Speaking Valve, PMV 007, for use with disposable ventilator tubing.
FJPT 2008;33(5):294-301 Chen et al.
airway clearance and IPPB therapy. He could control his respi- ratory rate at 32 to 24 times/min. No saturation lower than 90%
or decrease of more than 5% from baseline were found.16 This patient could only tolerate for 2 to 5 minutes at the 1st trial.
By Feb. 14, the patient could vocalize clearly in fluency (speak continuously and fluently with stable saturation and heart rate) for 10 minutes. The patient gradually improved in the areas of speech time, loudness, voice quality, and articulation. Sub- sequent trials found that this patient was able to use the PMV for a longer duration, for more than 2 hours. We prolonged the speaking valve practice, except during chest care, inhalation, swallowing, and sleep. He enjoyed talking with his attendant, family, and health care professionals. He even became more ac- tive in his rehabilitation program.
After Mar. 1, 2007, this patient started oral feeding with a diet of semi-liquid food. The naso-gastric tube was removed on the same day. On Mar. 8, 2007, he could eat a bowl of cereal mixed with tofu and fish without choking. According to state- ments from the patient and his attendant, he had two episodes of aspiration followed with coughing and choking.
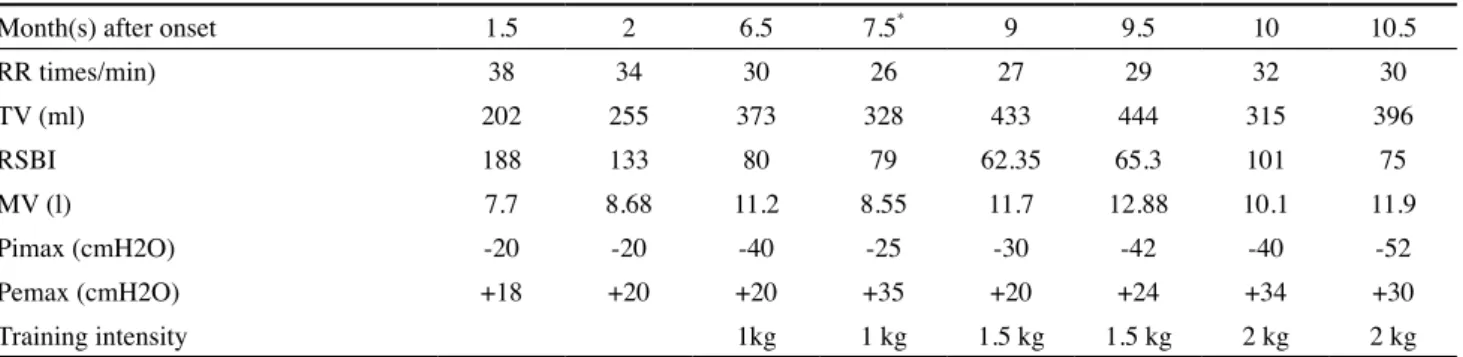
We started our inspiratory training programs with weight (1kg) on anterior costal margin on Mar. 1, 2007. The programs were executed 15 min/time two times per day, 7 days/week. His initial maximum aspiratory pressure (PI max) before training was
–40 cm H2O. The changes in parameters related to breathing pattern and respiratory muscle strength are shown in Table 1.
The program was continued except from Mar. 22 to 28, 2007, because of his operation for colon carcinoma, and from May 28, 2007 to Jun. 16, 2007, because of pneumonia. We used the SF-12 questionnaire (a tool to measure quality of life with 12 items health survey, including physical and mental summary component) for quality of life measurement. The results of the SF-12 are shown in Table 2. The results of the Barthel index showed that his functional level was totally dependent, with a score of 5 (less than 20, Apr. 18, 2007).
On May 28, 2007, the patient developed a fever. Acute tonsillitis and UTI were diagnosed. He had shortness of breath and a poor general condition. His respiratory rate increased to 30 times/min. Oxygen support with nasal cannula with 2 l/min was provided. A chest X-ray on May 28, 2007 showed increased infiltration over the right lower lobe (Fig 3.). Diflu- can100mg /4bottles/7days and Augmentin 600mg/qd/8days were prescribed, and then the oral form was tried. However, his condition worsened. On May 31. 2007, his arterial blood gas values while receiving a 0.28 fraction of inspired oxygen (FiO2) were pH of 7.37, PaCO2 of 52.9 mmHg, PaO2 of 69.6mmHg, bicarbonate of 30.5 and base excess of 4 meg/L. From May 31, 2007 to Jun. 12, 2007, nocturnal support was provided (syn-
Table 1. The improvement in parameters of respiratory function following the onset of spinal cord injury
Month(s) after onset 1.5 2 6.5 7.5* 9 9.5 10 10.5
RR times/min) 38 34 30 26 27 29 32 30
TV (ml) 202 255 373 328 433 444 315 396
RSBI 188 133 80 79 62.35 65.3 101 75
MV (l) 7.7 8.68 11.2 8.55 11.7 12.88 10.1 11.9
Pimax (cmH2O) -20 -20 -40 -25 -30 -42 -40 -52
Pemax (cmH2O) +18 +20 +20 +35 +20 +24 +34 +30
Training intensity 1kg 1 kg 1.5 kg 1.5 kg 2 kg 2 kg
*Colon CA operation
RR= respiratory rate; TV= tidal volume; Pimax= maximum inspiratory pressure; Pemax= maximum expiratory pressure; RSBI= rapid shallow breath- ing index; MV= minute ventilation
Table 2. The SF-12 score for PCS (physical component summary) and MCS (mental component summary).
Apr. 18, 2007 Jun. 25, 2007
PCS 25 30.9
MCS 54.7 66.6
chronous intermittent mandatory mode plus pressure support mode, with the tidal volume (Vt) set to achieve 500 ml, a respi- ratory rate of 6 breaths/min, a positive end-expiratory pressure (PEEP) with 5 cm H2O, and FiO2 of 0.35). On Jun. 16, 2007, after the episode of pneumonia had subsided, weight training (2kg) was begun again.
Finally, on Jun. 26, 2007, the patient could be transferred to a nursing home. He no longer required oxygen or ventilatory support. Our patient showed a mild increase in his inspiratory muscle strength (from -40cmH2O to -52 cmH2O) after nearly 4 months of training. His application for certification as handi- capped was approved. The SF-12 measurement was performed on Jun. 25, 2007 (Table 2). He had an attendant, who could transfer him from the bed to the wheelchair with high back and neck support. We had given her instruction on chest physical therapy, suction, the use of the speaking valve, and inspiratory muscle training before discharge.
dIscussIon
We present a case with C4-C5 spinal cord injury. He had the problem of respiratory insufficiency combined with repeat- ed pneumonia. The process of weaning involves liberating the patients from ventilatory support. Patients with tetraplegia may have decreased diaphragm motor function that impairs venti-
lation. Neuromuscular consequences like respiratory muscle dysfunction have an impact on the ability to be weaned from mechanical ventilation. Efforts to prevent or treat respiratory muscle weakness might have a role in reducing weaning fail- ure.7 For patients who have been through an operation and have vital capacity of less than 15 ml/kg of the ideal body weight, the weaning should be performed slowly, especially for those with a history of smoking or those older than 45 years old.
A review of the literature showed that improvement in pulmonary function was most obvious in the first 6 months fol- lowing SCI after training.8 Spontaneous recovery may persist during the first year after injury for cervical SCI.3 This case had suffered from difficulty with ventilator weaning since onset (Aug 10 2006). Actually, ventilator support had been discon- tinued for 3 weeks in November, 2006. Because of bacterial pneumonia, he was totally dependent on a ventilator until Jan.
16, 2007, when he was transferred to our hospital again.
The related clinical professionals (a doctor of neuro-sur- gery, a physical therapist, and a respiratory therapist) planned a program of ventilator weaning. We strongly suggested that nocturnal support should be maintained (slow-weaning) for two months after the observation of respiratory muscle fatigue.
In the daytime, chest physical therapy, lung expansion therapy followed by inspiratory muscle training were provided. The pa- tient achieved discontinuation of mechanical support after two months. According to Gutierrez et al’s evidence-based protocol, Fig. 3. Chest X-ray (AP) on May 27, 2007.
FJPT 2008;33(5):294-301 Chen et al.
patients with low tetraplegia have a greater chance of discon- tinuing ventilatory support than those with high tetraplegia.8 This proved true in our case. Two recent articles have indicated that SCI patients with ventilator-dependence are 39.5 times as likely to die during the first year after injury as compared to other SCI patients, and they have 2.61 times the mortality rate after that.9,10 Since ventilator-dependent tetraplegia appears to have the most severe impact on survival, periodic assessment of the possibility for ventilator weaning is important.
Most low tetraplegia patients required one or two months of training before mechanical ventilation could be discontinued.
In most cases, discontinuation was followed by an additional one or two months of daily optimization, in which patients were placed in trendelenberg, suctioned, bronchodilated, and hyperinflated using either a ventilator or Ambubag. Steps were taken to augment chest wall and lung compliance and reduce airway resistance to help patients breathe spontaneously.2 A retrospective study from Claxon et al suggested that aggres- sive secretion management is very important and may have an impact in decreasing the number of days of mechanical ven- tilation.11 The treatments should focus on lung expansion and airway clearance. In this case, we selected intermittent positive pressure breathing for lung expansion therapy combined with chest physical therapy.
The respiratory muscle training targeted specific respi- ratory muscles to improve respiratory function. A review of the literature revealed that resistance training for inspiratory muscles tends to lead to high intensity (over 75% of maximal inspiratory pressure, or even higher).12-14 Gutierrez et al8 sug- gested that in low tetraplegic patients, resistance training ap- peared to be of greatest clinical benefit to patients with C4 or C5 injuries. A qualitative systematic review from Brooks et al suggested the selection of resistance muscle trainers for at least 15 minutes, twice daily, five to seven days per week for six to eight weeks could benefit the patients. There is very little published evidence for the effectiveness of inspiratory muscle training.15 A systematic review from Van Houtte et al showed that respiratory muscle training tended to improve expiratory muscle strength, vital capacity, and residual volume. But there was not sufficient data to prove the effects on inspiratory mus- cle strength, quality of life, exercise performance, and respira- tory complications.12 Our patient showed a mild increase in his inspiratory muscle strength (from -40cmH2O to -52 cmH2O)
after nearly 4 months of training. Spontaneous recovery could also have contributed to that improvement. During this period, he had neurological recovery in right wrist extensor (muscle power 2→3).
With the attempt of weaning and accessibility of suction, the tracheostomy was performed nearly one month after onset in our patient after several failed extubations. The negative aspects of tracheostomy are difficulties in communication and impaired swallowing. In this case, we used the Passy-Muir valve to help the patients restore speaking, swallowing, and respiratory function. During this period, we subjectively ob- served that communication was quite important to the patient.
The restoration of swallowing also inspired him. Dysphagia is a contributor to respiratory deterioration after SCI.5 Communi- cation with a one-way speaking valve can lead to improvement in the patient’s sense of well-being and increase motivation for the eating process and rehabilitation. The restoration of speech function can provide improvement in the quality of life of a patient with tracheostomy. For conscious patients with intact cognitive function, the restoration of speech is important.16,17
Speech is an over-laid function of the respiratory system.
The functions of speech, swallowing, and respiration involve the structures of both the upper and the lower respiratory sys- tem. The physiological function of a one-way valve is to inhale through the tracheostomy tube and exhale through the upper airway. Because air is passed through the nasal passages, the speaking valve can aid in weaning and decannulation.18,19
The closed system allows for normalization of pharyn- geal pressure, an increase in pharyngeal/laryngeal sensation, a decrease in the risk of aspiration, and improvement in olfac- tion.18,20-22 The sense of smell could recover after the placement of a speaking valve.
Summary
As a recent paper stated, improvement in respiratory func- tion and weaning from a mechanical ventilator are extremely important to the quality of life for patients with SCI.3 In our case, we found that slow weaning combined with intensive air- way clearance techniques and lung expansion therapy could im- prove the chances of success in ventilator weaning for a patient with lower tetraplegia. Starting the respiratory muscle training as early as possible could aid in ventilator weaning. Clinical professionals should consider the importance of communica-
tion and swallowing for these patients. The early restoration of speech and swallowing can help to promote the quality of life of the patient for long-term care.
references
1. Brown RB, DiMarco AF, Hoit JD, Garshick E. Respiratory dys- function and management in spinal cord injury. Respir Care 2006;51:853-68.
2. Berlly M, Shem K. Respiratory management during the first five days after spinal cord injury. J Spinal Cord Med 2007;30:309-18.
3. Zimmer MB, Nantwi K, Goshgarian HG. Effect of spinal cord in- jury on the respiratory system: basic research and current clinical treatment options. J Spinal Cord Med 2007;30:319-30.
4. Shavelle RM, DeVivo MJ, Strauss DJ, Paculdo DR, Lammertse DP, Day SM. Long-term survival of persons ventilator dependent after spinal cord injury. J Spinal Cord Med 2006;29:511-9.
5. Wuermser L-A, Ho CH, Chiodo AE, Priebe MM, Kirshblum SC, Scelza WM. Spinal cord injury medicine. 2. Acute care manage- ment of traumatic and nontraumatic injury. Arch Phys Med Rehabil 2007;88:S55-61.
6. Kirshblum SC, Priebe MM, Ho CH, Scelza WM, Chiodo AE, Wuermser L-A. Spinal cord injury medicine. 3. Rehabilitation phase after acute spinal cord injury. Arch Phys Med Rehabil 2007;88:S62-70.
7. Boles J-M, Bion J, Connors A, Herridge M, March B, Melot C, et al. Weaning from mechanical ventilation. Eur Respir J 2007;29:1033-56.
8. Gutierrez CJ, Harrow J, and Haines F. Using an evidence- based protocol to guide rehabilitation and weaning of ventilator- dependent cervical spinal cord injury patients. J Rehabil Res Dev 2003;40:99-110.
9. Devivo MJ, Krause JS, Lammertse DP. Recent trends in mortality and causes of death among persons with spinal cord injury. Arch Phys Med Rehabil 1999;80:1411-9.
10. Krause JS, Devivo MJ, Jackson AB. Health status, community in-
tegration, and economic risk factors for mortality after spinal cord injury. Arch Phys Med Rehabil 2004;85:1764-73.
11. Claxton R, Wong D, Chung F, Fehlings M. Predictors of hospital mortality and mechanical ventilation in patients with cervical spi- nal cord injury. Can J Anaesth 1998;45:144-9.
12. Houtte SV, Vanlandewijck Y, Gosselink R. Respirtory muscle train- ing in persons with spinal cord injury: A systematic review. Respir Med 2006;100:1886-95.
13. Hill K, Jenkins SC, Philippe DL, Cecins N, Shepherd KL, Green DJ, et al. High-intensity inspiratory muscle training in COPD. Eur Respir J 2006;27:1119-28.
14. Mueller G, Perret C, Spengler CM. Optimal intensity for respira- tory muscle endurance training in patients with spinal cord injury. J Rehabil Med 2006;38:381-6.
15. Brooks D, O’Brien K. Is inspiratory muscle training effective for individuals with cervical spinal cord injury? A qualitative system- atic review. Clinical Rehabil 2005;19:237-46.
16. Hess DR. Facilitating speech in patient with a tracheostomy. Respir Care 2005;50:519-25.
17. Hoit JD, Banzett RB, Lohmeier HL, Hixon TJ, Brown R.
Clinical ventilator adjustments that improve speech. Chest 2003;124:1512-21.
18. Kaut K, Turcott JC, Lavery M. Passy-Muir speaking valve. Dimens Crit Care Nurs 1996;15:298-306.
19. Bell SD. Use of Passy-Muir tracheostomy speaking valve in mechanically ventilated neurological patients. Crit Care Nurse 1996;16:63-8.
20. Elpern EH, Okonek MB, Bacon M, Gerstung C, Skrzynski M, Village EG, et al. Effect of the Passy-Muir tracheostomy speaking valve on pulmonary aspiration in adults. Heart Lung 2000;29:287-93.
21. Suiter DM, McCullough GH, Powell PW. Effects of cuff deflation and one-way tracheostomy speaking valve placement on swallow physiology. Dysphagia 2003;18;284-92.
22. Lichman SW, Birnbaum IL, Sanfilippo MR, Pellicone JT, Damon WJ, King ML. Effect of a tracheostomy speaking valve on secre- tions, arterial oxygenation, and olfaction: a quantitative evaluation.
J Speech Hear Res.1995;38:549-55.
FJPT 2008;33(5):294-301 Chen et al.
1 萬芳醫院胸腔內科
2 署立雙和醫院神經外科
3 署立雙和醫院胸腔內科
4 台北醫學大學呼吸治療學系
通訊作者:江玲玲 台北醫學大學呼吸治療學系 110台北市信義區吳興街250號 電話:2736-1661轉3512 E-mail: llchiang@tmu.edu.tw
頸椎損傷患者之呼吸照護經驗
陳曉宜1 林乾閔2 李俊年3 黃卓禕1 江玲玲4
高位頸椎脊髓損傷患者有極高的危險性發生呼吸衰竭及相關的呼吸併發症。呼吸肌的偏癱無力常
導致病患需要仰賴呼吸器。這個個案報告我們所要呈現的是協助一位C4到C5不完全脊髓損傷的
66歲男性病患脫離呼吸器的照護過程。我們所提供的計劃包括夜間通氣支持、胸腔物理治療、肺 部擴張治療及吸氣肌訓練。