Susceptibilities to Amphotericin B, Fluconazole and Voriconazole of Trichosporon Clinical Isolates
Moan-Shane Tsai
1. Yun-Liang Yang
2,3. An-Huei Wang
1. Lih-Shinn Wang
4. Daniel, C. T. Lu
5. Ci-Hong Liou
1. Li-Yun Hsieh
1. Chi-Jung Wu
1,6. Ming-Fang
Cheng
7,8. Zhi-Yuan Shi
9. Hsiu-Jung Lo
1,10*1
National Institute of Infectious Diseases and Vaccinology, National Health Research
Institutes, Miaoli, Taiwan,
2
Department of Biological Science and Technology, National Chiao Tung University,
Hsinchu, Taiwan
3
Institute of Molecular Medicine and Bioengineering, National Chiao Tung
University, Hsinchu, Taiwan,
4
Division of Infectious Diseases, Buddhist Tzu-Chi General Hospital, Hulien, Taiwan
5
Division of Infectious Diseases, Lo-Hsu Foundation Inc. Lo-Tung Poh Ai Hospital,
Yilan, Taiwan
6
Division of Infectious Diseases, National Cheng-Kung University Hospital and
Medical College, Tainan, Taiwan,
7
Division of Infectious Diseases, Veterans General Hospital—Kaohsiung, Kaohsiung,
Taiwan
8
National Yang-Ming University, Taipei, Taiwan
9
Division of Infectious Diseases, Veterans General Hospital – Taichung, Taichung,
Taiwan
10
School of Dentistry, China Medical University, Taichung, Taiwan
Running title: Drug Susceptibility of Trichosporon
* Corresponding author:
Hsiu-Jung Lo, 35, Keyan Road, Zhunan Town, Miaoli County 35053, Taiwan
Telephone: 886 37246166, FAX: 886 37586457 E-mail: [email protected]
Abstract A total of 35 Trichosporon isolates were collected from the Taiwan Surveillance of Antimicrobial Resistance of Yeasts (TSARY) project from 1999 to 2006 and their identifications as well as drug susceptibilities were determined. The most frequently isolated species was T. asahii (62.9%) and the most common clinical sample that yielded Trichosporon isolates was urine (37.1%). The etiology of all seven invasive trichosporonosis was T. asahii. For the 22 T. asahii isolates, the MIC
50and MIC
90for amphotericin B were 0.25 and 1 μg/mL, respectively. Those for
fluconazole were 2 and 4 μg/mL, respectively, and for voriconazole, 0.031 and 0.063 μg/mL, respectively. When the intraclass correlation coefficients (ICCs) and
agreements were calculated, we found that the MICs of fluconazole obtained from different methods were similar and the inter-method discrepancies were low.
Nevertheless, no unanimous MIC of amphotericin B and voriconazole was obtained among different methods.
Keywords: antifungal susceptibility testing, CLSI, EUCAST, Trichosporon
Introduction
Taiwan Surveillance of Antimicrobial Resistance of Yeasts (TSARY) is a nation-wide surveillance project conducted by the National Health Research Institutes (NHRI) in Taiwan. It was instituted to monitor the trends of the distributions of clinical yeast species and the susceptibilities thereof to commonly-prescribed antifungal agents.
From 1999 to 2006, a total of 2624 yeast isolates were collected from TSARY- participating hospitals and submitted to the NHRI [1-5]. Among the enormous number of yeasts isolates collected, Candida species represented a substantial proportion. Nevertheless, non-Candida yeasts, though less-than-common in terms of number of isolates, have been consistently recovered from clinical specimens, and their clinical involvement deserves more attention. Trichosporon species is one of such.
Trichosporon species can survival in different environments, such as soil, decomposing wood, air, and water. They can also be found in human gastrointestinal tract, oral cavity, respiratory tract, and skin. Approximately 11% of healthy male and up to 14% of female were colonized by Trichosporon species on their perigenital skin [6, 7]. Although most isolates from patients are considered to be colonization or superficial infections, they have been recognized as an emerging opportunistic fungal agent for invasive infections, particularly in the setting of hematologic, solid organ malignancy, solid organ transplantation patients [8-11].
Although there were reports of in vitro antifungal susceptibility testing of clinical Trichosporon isolates [6, 8, 12-15], results yielded from a large number of clinical yeast isolates collected for surveillance purposes in Taiwan have never been reported.
In addition, the current method of in vitro antifungal susceptibility testing of
Trichosporon species has not yet been standardized by either the Clinical Laboratory
Standards Institute (CLSI) [16] or the Subcommittee of Antifungal Susceptibility Testing of the European Committee for Antimicrobial Susceptibility Testing (EUCAST) [17]. Hence, the minimal inhibitory concentration (MIC) data of
Trichosporon isolates obtained by using either CLSI or EUCAST methodologies are actually presumptive values. No studies to date have yet compared these different testing procedures on Trichosporon species to examine the inter-method discrepancy.
To better understand the in vitro susceptibility of Trichosporon species to antifungal agents, we have used different methods to determine MICs of commonly- prescribed drugs, including amphotericin B, fluconazole, and voriconazole. The MIC datasets acquired were further compared with one another by calculating the intraclass correlation coefficients (ICC) [18] and the agreements to examine the dissimilarities among the methods.
Materials and Methods
Clinical yeast isolates
Clinically significant yeast isolates from TSARY-participating hospitals were
submitted to the NHRI [3-5, 19]. There were 22, 24, and 22 hospitals participating in the survey in 1999, 2002, and 2006, respectively. For the TSARY in 1999, each hospital submitted up to 10 C. albicans and 40 non-albicans Candida species isolates.
For TSARY in 2002 and 2006, each hospital submitted all the yeast isolates from
sterile body sites. In addition, up to 10 C. albicans clinical isolates as well as 40 non-
albicans Candida species isolates obtained from non-sterile body sites were sent to
the NHRI as well. The yeast isolates were sub-cultured on Sabouraud dextrose agar
(SDA, Becton Dickinson, Cockeysville, MD, USA) to assess the purity and for
precise species identification upon arrival at the NHRI. Pure isolates were labelled and stored in vials containing 50% glycerol at −70 °C for further analyses.
Yeast identification
Yeast isolates were speciated according to the instructions of both ID 32C and the VITEK Yeast Biochemical Card (YBC) (bioMérieux, Marcy l'Etoile, France).
Identifications of all the Trichosporon isolates were further assessed by sequencing ribosomal DNA (rDNA), including ITS and/or D1/D2 regions. The fungus-specific universal primer pair ITS1 (5’-TCCGTAGGTGAACCTGCGG-3’) and ITS2 (5’- GCTGCGTTCTTCATCGATGC-3’) and another primer pair NL1 (5’-
GCATATCAATAAGCGGAGGAAAAG-3’) and NL4 (5’-
GGTCCGTGTTTCAAGACGG-3’) were employed to amplify the internal transcribed space (ITS) 1 and the D1/D2 [20, 21].
In vitro antifungal susceptibility testing
The in vitro activities of amphotericin B, fluconazole, and voriconazole were determined according to the methods of the CLSI M27-A3 document [16], the EUCAST document [17], and the modified CLSI M27-A3 protocol by using 2%- glucose supplemented RPMI 1640 broth. Amphotericin B was kindly provided by Bristol Myers Squibb, fluconazole and voriconazole were kindly provided by Pfizer.
First of all, the isolates from vials kept at −70 °C were grown in liquid medium at
35°C overnight before the cell density was adjusted. The MICs were determined at the
following time points: the CLSI methodology-after 24h (CLSI 24H) and 48h (CLSI
48H) incubation, the modified CLSI-after 24h (CLSI 24HG) and 48h (CLSI 48HG)
incubation, and the EUCAST standard-after 24h incubation (EUCAST).
The final growth of each isolate was measured using a Biotrak II plate spectrophotometric reader (Amershan Biosciences, Biochrom Ltd., Cambridge England) after incubation at 35ºC for 24h and 48h. The MIC for amphotericin B was defined as the lowest concentration capable of completely preventing cell growth. The MICs of both fluconazole and voriconazole were read as the lowest concentration that reduced the turbidity of cells by more than 50%. Strains of Candida albicans ATCC
®90028, Candida krusei ATCC
®6258, and Candida parapsilosis ATCC
®90018 were used as the quality control. The MICs of 50% and 90% of the total population were defined as MIC
50and MIC
90.
Data analysis
Agreement was defined as discrepancies between each compared MIC pair with no more than ±1 twofold dilution. For example, if an MIC value of 1 μg/mL of a clinical Trichosporon isolate was obtained at 24h, the MIC values between 0.5 and 2 μg/mL of the same antifungal agent by different methods at the same or different incubation periods were considered to be in agreement. The percentage of agreement between the MIC data sets obtained by each testing method per antifungal agent was calculated.
For convenience of computation, the MICs were transformed into log 2 values for calculating the ICCs. The calculation was performed according to the formula: ICC = (group mean square – error mean square) / (group mean square + error mean square) with Windows software PASW Statistics 18 (SPSS Inc. Chicago, IL, USA). The result has a maximum value of 1 if there is a perfect correlation and a minimum value of -1 if there is an absence of correlation [18]. A p value <0.05 was considered
statistically significant.
Data collection
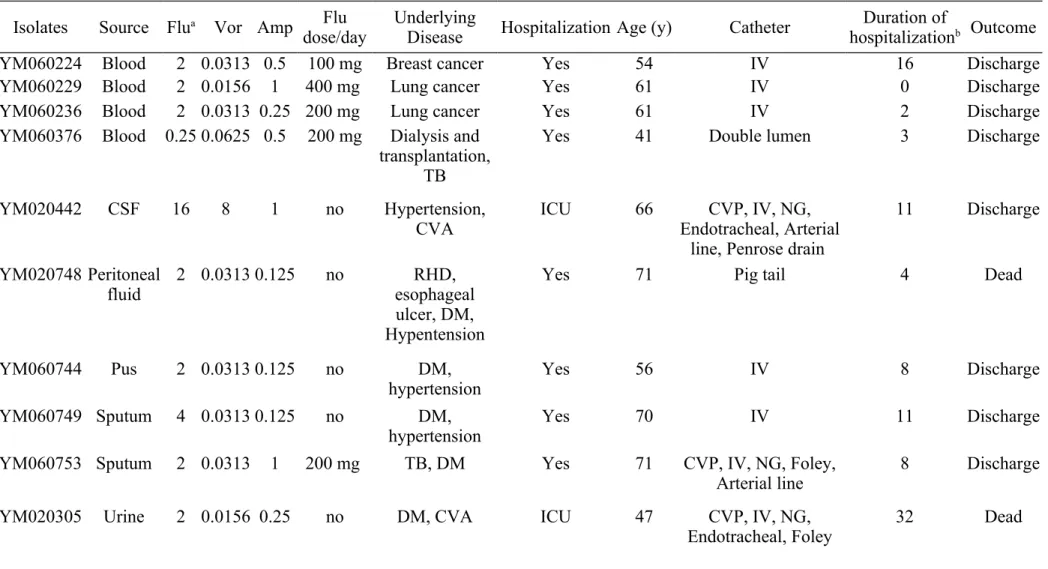
Clinical data of 13 out of 21 patients colonized/infected by T. asahii were recorded retrospectively. The information included demographic characteristics, hospital unit, length of hospitalization during when the isolate was recovered, predisposing factors within one to three months prior to isolate recovered (such as antifungal drugs, antibiotics, antituberculous agents, dialysis, catheter insertion, parenteral nutrition), underlying illnesses (such as cancer, cerebral vascular accident, diabetes,
hypertension, and pulmonary diseases), and other laboratory findings.
Results
Initially, a total of 38 Trichosporon isolates out of 2624 yeasts isolates collected by TSARYs were identified by ID 32C and/or VITEK YBC. Using specific rDNA sequences for further speciation, we were able to identify that three isolates demonstrating the same biochemical phenotypic profiles as Trichosporon species were unequivocally speciated as Candida species and they were excluded from this study. According to the rDNA sequences, the remaining 35 isolates were comprised of 8 species (Table 1). Trichosporon asahii was the most frequently isolated species, accounting for 62.9% of all Trichosporon isolates, followed by T. montevideense (14.3%), T. cutaneum (5.7%), T. dermatis (5.7%), and then T. debeurmannianum, T.
faecale, T. insectorum, and T. japonicum (2.9% for each species). The most common
clinical samples that yielded Trichosporon isolates were urine specimens (13 isolates),
accounting for 37.1% of all isolates, followed by wound discharge (17.1%), blood
samples (14.3%), respiratory tract secretion (11.4%), and 14.3% from three other
sources (Table 1). A total of seven cases of invasive trichosporonosis were identified, which were blood stream infections (5), meningitis (1), and peritonitis (1). The etiology of them was all T. asahii.
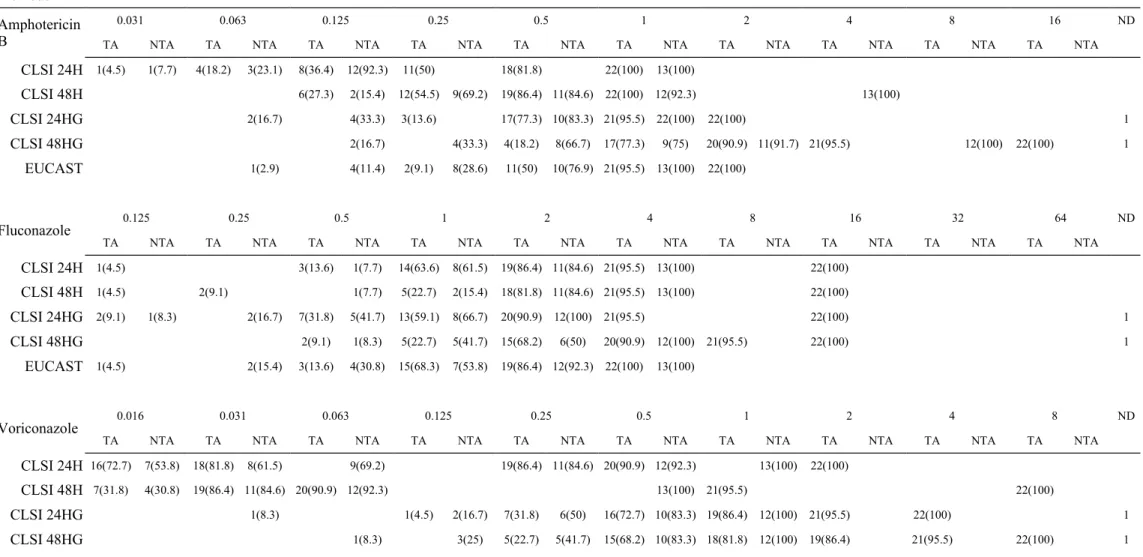
The in vitro susceptibilities to amphotericin B, fluconazole, and voriconazole were determined by different methods and the results are summarized in Table 2.
When the CLSI interpretative breakpoints of susceptibility of Candida species were employed, the majority of Trichosporon clinical isolates were susceptible to all three antifungal agents (Table 2, CLSI 48H). However, there were two exceptions. One T.
cutaneum isolate exhibited a MIC value of 4 μg/mL to amphotericin B. The other was a T. asahii isolate, recovered from cerebrospinal fluid, with MIC values of 1 μg/mL to amphotericin B, but with of 16 μg/mL and 8 μg/mL to fluconazole and voriconazole, respectively. According to the MIC results of CLSI 48H, the MIC
90of T. asahii isolates to amphotericin B, fluconazole, and voriconazole were 1 μg/mL [ranging from 0.125 to 1, mean 0.312)], 4 μg/mL (ranging from 0.125 to16, mean 1.763), and 0.063 μg/mL (ranging from 0.016 to 8, mean 0.039), respectively. The MIC
90results using fluconazole as a testing agent were uniformly found to be below 4 μg/mL with the different methods (Table 2), and the MIC data obtained were scattered over similar ranges.
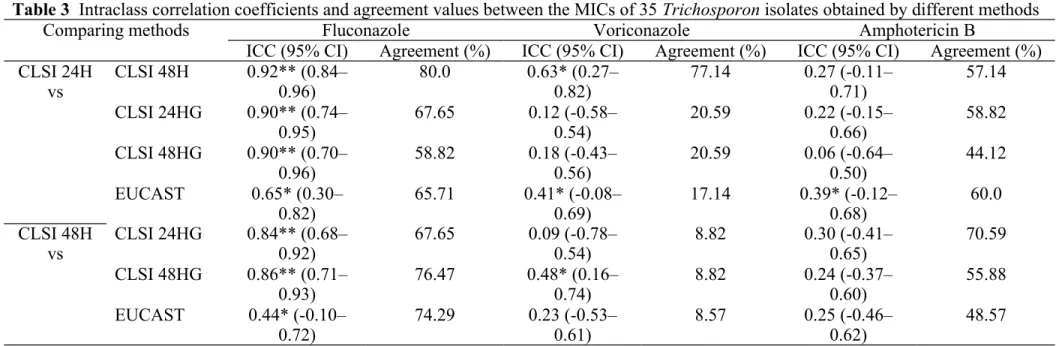
When the analysis was performed for the antifungal agents, the reproducibility of the ICC and agreement values per antifungal agent varied among the MIC results obtained by the different methods (Table 3). The agreement values among each compared methodology pair for fluconazole ranged from 58.82% to 80.0%.
Therefore, the MICs of fluconazole obtained from different methods were similar and the inter-method discrepancies were low. For voriconazole, only the results of CLSI 24H versus CLSI 48H showed a high degree of agreement (77.14%). As for
amphotericin B, the ICC values among the pair were not consistent, ranging from 0.22
to 0.39, suggesting that a high degree of variation existed among these testing methods.
The clinical characteristics of 13 patients colonized/infected with T. asahii infections are shown in Table 4. All patients but one had one or more than one
underlying diseases. A total of seven T. asahii isolates were from sterile sites. Isolates YM060229 and YM060236 were recovered from the same patient. Among the six patients, the information of one patient with invasive infections was not available.
Among the five patients reviewed, three were treated with fluconazole and discharged with improvement. Of the two patients without antifungal treatment, one died and the other was discharged. Isolates YM060229 and YM060236 were recovered on August 10 and September 15 in 2006, respectively from the same patient who received fluconazole treatment when the YM060229 isolate was recovered from blood. No other patients receive antifungal treatment within three months prior to the isolate recovery. Three patients, YM020748, YM060733, and YM060734, did not received antibiotic treatment within one month prior to isolate recovery and only two patients, YM060733 and Ym990597, did not receive antibiotic treatment when isolates were recovered. Three patients, YM990296, YM060753, and YM060376, received antituberculous treatment and one, YM060376, received dialysis prior to and when isolates were recovered.
Discussion
The spectrum of trichosporonosis encompasses a wide range of symptoms, ranging
from a minor superficial skin disease to a fulminant invasive systemic blood stream
infection [11]. Given the diverse clinical presentations of trichosporonosis and the
fatal consequences of severe Trichosporon infections, as well as for the purpose of
providing unambiguous mycological characteristics of the pathogenic yeast for clinicians as a guide for therapeutic intervention, precise taxonomical speciation of clinical Trichosporon isolates is critical. Currently-available biochemical or
morphological phenotypic methods offer little discriminatory capability in
differentiating Trichosporon species [13]. These commercialized products neither utilize unambiguous biological characteristics in the differentiation of clinical yeast isolates, nor do they have a database of sufficient numbers of Trichosporon species for comparison of the physiological traits (package inserts of both ID 32C and VITEK YBC). The results of speciation become unreliable if the phenotypic characteristics alone are employed for differentiating clinical yeast isolates. A practical approach for fungal speciation based on more defined biological characteristics other than
equivocal phenotypic ones is therefore warranted. The modern molecular method of DNA-based sequence identification, which provides accurate species as well as the large DNA sequence library provide an ideal solution for the speciation of clinically- important yeasts [14, 22-24]. In this study, either the ITS region of yeast rDNA alone or in combination with the D1/D2 region of the fungal rDNA was demonstrated to be clinically feasible and provided adequate mycological characteristics for yeast
speciation. As the number of newly-discovered opportunistic yeasts is expected to be increasing, molecular methods for differentiating clinically-important yeasts are likely to serve as accurate alternatives to conventional identification methods.
In the present study, 4 patients were in intensive care units and 11 out of 13 had antibiotic treatment and catheters when isolates were recovered. Approximately 70%
patients (9/13) had more than one underlying diseases. Diabetes was the most
common one accounting for 41.7%, followed by hypertension (33.3%), cerebral
vascular accident (25%), and tuberculosis (25%). Our findings are consistent with
previous reports that there were several groups of patients commonly associated with
trichosporonosis, such as intensive care units patients subjected to invasive medical procedures and antibiotic treatment, and patients with haematological diseases [6, 8, 15, 25].
Our results showed that T. asahii was the most frequently isolated Trichosporon species (22 isolates) in Taiwan and far outnumbered the non-T. asahii clinical isolates (13 isolates). All seven isolates from sterile sites were T. asahii, consistent with previous reports that T. asahii is the most common species among Trichosoporn genus causing invasive trichosopornosis [8, 15, 26]. Of note, amphotericin B exhibited a higher MIC
90(1 g/mL) against T. asahii than that of non-T. asahii. In earlier studies [13-15], the MIC
90of amphotericin B of Trichosporon were found to be higher than or equal to 2 μg/mL. In this study, the newer antifungal agents, echinocandins, were not tested because it is well known that basidiomycete yeasts, such as Trichosporon, are resistant to this group of antifungal agents [27], and hence they are not used for treatment of infections caused by these yeasts. Most of the MICs of voriconazole against Trichosporon isolates were clustered below 0.0625 μg/mL (Table 2). In contrast to amphotericin B, the triazole antifungal agents exhibited much more favourable in vitro activities against Trichosporon isolates [8, 15, 28-32].
Furthermore, due to the poor physiological condition of severely ill patients, a potent
agent with a much broader antifungal activity spectrum, such as voriconazole and new
azoles, isavuconazole and posaconazole, may be recommended [6, 28, 33].
When conducting TSARYs, we collected yeast isolates without detail clinical information from hospitals, resulting in some limitations in the present study. Among the 35 isolates, urine was the most common source, which is consistent with the reports that urinary tract infections can be caused by Trichosporon spp.[34-36].
Nevertheless, lacking clear and specific indications for the clinical interpretation of Trichosporon recovered from urine results in a clinical challenge for clinicians and it is also a limitation of the present study. Another limitation is that four isolates recovered from blood were from three patients. Only one, YM060376, had an
indwelling catheter. It is difficult to determine whether the improvement of the patient resulted from fluconazole treatment and/or line removal. In addition, the YM020442 isolate recovered from CSF (collected on August 5 in 2002) and was with high MICs of both fluconazole and voriconazole. Interestingly, the patient did not receive antifungal treatment and was discharged from the hospital. The chart stated “wait for another fungal culture before treating patient” indicates that the doctor may consider the fungal isolate as colonized microorganism in the CSF drainage tube. The culture result performed on August 28 was negative. Whether the drainage tube was changed between these two cultures did not stated on the chart. The outcome of patient
suggests that the YM020442 isolate is very possible to be a contaminant of drainage tube. Among 13 patients reviewed, 5 were treated with fluconazole and none was treated with any other antifungal drugs. Thus, another limitation of the present study is that we could not compare the effects of fluconazole with that of amphotericin B.
The MIC values obtained by the methods of EUCAST and CLSI were consistent for fluconazole but not for amphotericin B and voriconazole. Even though the sample size may not be sufficiently large, our data suggests that T. asahii is susceptible to fluconazole in vivo, which is correlated to in vitro susceptibility testing results.
Further studies with a larger number of Trichosporon isolates are therefore needed to
offset the correlation between in vitro drug susceptibilities obtained by different methods and in vivo clinical outcomes.
Acknowledgements
The authors would like to thank Bristol Myers Squibb for supplying the reference powder of amphotericin B, and Pfizer for fluconazole and voriconazole. They also thank the TSARY hospitals, especially the following 17, for providing clinical Trichosporon isolates: Chang Gung Memorial Hospital at Keelung, Chang Gung Memorial Hospital at Linkou, Chi-Mei Foundation Hospital, Chung Shan Medical Dental College Hospital, Tainan Municipal Hospital, Kuan-Tien General Hospital, Lo-Hsu Foundation Inc. Lo-Tung Poh Ai Hospital, Kaohsiung Military Hospital, Hua-Lien Mennonite Church Hospital, National Cheng Kung University Hospital, Hua-Lien Hospital, DOH, the Executive Yuan, Show Chwan Memorial Hospital, Tri Service General Hospital, Veterans General Hospital –Taichung, Veterans General Hospital-Kaohsiung, Asia East Memorial Hospital, and Buddhist Tzu-Chi General Hospital. This work was supported in part by the NHRI fellowship research grant to MST and by grants CL-098-PP-04 and ID-099-PP-04 from the NHRI to HJL.
Declaration of Interest: The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
References
1. Yang YL, Cheng HH, Ho YA, Hsiao CF, Lo HJ. Fluconazole resistance rate
of Candida species from different regions and hospital types in Taiwan. J
Microbiol Immunol Infect. 2003;36:187-91.
2. Yang YL, Cheng MF, Wang CW, Wang AH, Cheng WT, Lo HJ et al. The distribution of species and susceptibility of amphotericin B and fluconazole of yeast pathogens isolated from sterile sites in Taiwan. Med Mycol.
2010;48:328-34.
3. Yang YL, Ho YA, Cheng HH, Ho M, Lo HJ. Susceptibilities of Candida species to amphotericin B and fluconazole: the emergence of fluconazole resistance in Candida tropicalis. Infect Control Hosp Epidemiol. 2004;25:60-
4.
4. Yang YL, Li SY, Cheng HH, Lo HJ. Susceptibilities to amphotericin B and fluconazole of Candida species in TSARY 2002. Diagn Microbiol Infect Dis.
2005;51:179-83.
5. Yang YL, Wang AH, Wang CW, Cheng WT, Li SY, Lo HJ et al.
Susceptibilities to amphotericin B and fluconazole of Candida species in
TSARY 2006. Diagn Microbiol Infect Dis. 2008;61:175-80.
6. Colombo AL, Padovan AC, Chaves GM. Current knowledge of Trichosporon
spp. and Trichosporonosis. Clin Microbiol Rev. 2011;24:682-700.
7. Ellner K, McBride ME, Rosen T, Berman D. Prevalence of Trichosporon beigelii. Colonization of normal perigenital skin. J Med Vet Mycol.
1991;29:99-103.
8. Chagas-Neto TC, Chaves GM, Melo AS, Colombo AL. Bloodstream
infections due to Trichosporon spp.: species distribution, Trichosporon asahii genotypes determined on the basis of ribosomal DNA intergenic spacer 1 sequencing, and antifungal susceptibility testing. J Clin Microbiol.
2009;47:1074-81.
9. Hajjeh RA, Blumberg HM. Bloodstream infection due to Trichosporon beigelii in a burn patient: case report and review of therapy. Clin Infect Dis.
1995;20:913-6.
10. Tashiro T, Nagai H, Kamberi P, Goto Y, Kikuchi H, Nasu M et al.
Disseminated Trichosporon beigelii infection in patients with malignant diseases: immunohistochemical study and review. Eur J Clin Microbiol Infect
Dis. 1994;13:218-24.
11. Walsh TJ. Trichosporonosis. Infect Dis Clin North Am. 1989;3:43-52.
12. Guo LN, Xiao M, Kong F, Chen SC, Wang H, Sorrell TC et al. Three-locus identification, genotyping, and antifungal susceptibilities of medically important Trichosporon species from China. J Clin Microbiol. 2011;49:3805-
11.
13. Li HM, Du HT, Liu W, Wan Z, Li RY. Microbiological characteristics of medically important Trichosporon species. Mycopathologia. 2005;160:217-25.
14. Rodriguez-Tudela JL, Diaz-Guerra TM, Mellado E, Cano V, Tapia C, Perkins
A et al. Susceptibility patterns and molecular identification of Trichosporon
species. Antimicrob Agents Chemother. 2005;49:4026-34.
15. Ruan SY, Chien JY, Hsueh PR. Invasive trichosporonosis caused by Trichosporon asahii and other unusual Trichosporon species at a medical
center in Taiwan. Clin Infect Dis. 2009;49:e11-7.
16. Clinical Laboratory Standards Institute CLSI. Reference method for broth dilution antifungal susceptibility testing of yeasts; approved standard-third
edition. CLSI document M27-A3. Wayne, PA. 2008.
17. Testing SoASTotEECfAS. Subcommittee on Antifungal Susceptibility Testing of the ESCMID European Committee for Antimicrobial Susceptibility
Testing: EUCAST Technical Note on the method for the determination of broth dilution minimum inhibitory concentrations of antifungal agents for
conidia-forming moulds. Clin Microbiol Infect. 2008;14:982-4.
18. McGraw KO, Wong SP. Forming inferences about some intraclass correlation
coefficients. Psychological Methods. 1996;1:30-46.
19. Lo HJ, Ho AH, Ho M. Factors accounting for mis-identification of Candida species. J Microbiol Immunol Infect. 2001;34:171-7.
20. Leaw SN, Chang HC, Barton R, Bouchara JP, Chang TC. Identification of medically important Candida and non-Candida yeast species by an
oligonucleotide array. J Clin Microbiol. 2007;45:2220-9.
21. Leaw SN, Chang HC, Sun HF, Barton R, Bouchara JP, Chang TC.
Identification of medically important yeast species by sequence analysis of the
internal transcribed spacer regions. J Clin Microbiol. 2006;44:693-9.
22. Kalkanci A, Sugita T, Arikan S, Yucesoy M, Ener B, Otag F et al. Molecular identification, genotyping, and drug susceptibility of the basidiomycetous yeast pathogen Trichosporon isolated from Turkish patients. Med Mycol.
2010;48:141-6.
23. Nakajima M, Sugita T, Mikami Y. Granuloma associated with Trichosporon asahii infection in the lung: Unusual pathological findings and PCR detection
of Trichosporon DNA. Med Mycol. 2007;45:641-4.
24. Taj-Aldeen SJ, Al-Ansari N, El Shafei S, Meis JF, Curfs-Breuker I, Theelen B et al. Molecular identification and susceptibility of Trichosporon species isolated from clinical specimens in Qatar: isolation of Trichosporon dohaense
Taj-Aldeen, Meis & Boekhout sp. nov. J Clin Microbiol. 2009;47:1791-9.
25. Girmenia C, Pagano L, Martino B, D'Antonio D, Fanci R, Specchia G et al.
Invasive infections caused by Trichosporon species and Geotrichum capitatum in patients with hematological malignancies: a retrospective
multicenter study from Italy and review of the literature. J Clin Microbiol.
2005;43:1818-28.
26. Suzuki K, Nakase K, Kyo T, Kohara T, Sugawara Y, Shibazaki T et al. Fatal
Trichosporon fungemia in patients with hematologic malignancies. Eur J
Haematol. 2010;84:441-7.
27. Bayramoglu G, Sonmez M, Tosun I, Aydin K, Aydin F. Breakthrough Trichosporon asahii fungemia in neutropenic patient with acute leukemia
while receiving caspofungin. Infection. 2008;36:68-70.
28. Araujo Ribeiro M, Alastruey-Izquierdo A, Gomez-Lopez A, Rodriguez- Tudela JL, Cuenca-Estrella M. Molecular identification and susceptibility testing of Trichosporon isolates from a Brazilian hospital. Rev Iberoam Micol.
2008;25:221-5.
29. McGinnis MR, Pasarell L, Sutton DA, Fothergill AW, Cooper CR, Jr., Rinaldi MG. In vitro activity of voriconazole against selected fungi. Med Mycol.
1998;36:239-42.
30. Paphitou NI, Ostrosky-Zeichner L, Paetznick VL, Rodriguez JR, Chen E, Rex JH. In vitro antifungal susceptibilities of Trichosporon species. Antimicrob
Agents Chemother. 2002;46:1144-6.
31. Perparim K, Nagai H, Hashimoto A, Goto Y, Tashiro T, Nasu M. In vitro susceptibility of Trichosporon beigelii to antifungal agents. J Chemother.
1996;8:445-8.
32. Uzun O, Arikan S, Kocagoz S, Sancak B, Unal S. Susceptibility testing of
voriconazole, fluconazole, itraconazole and amphotericin B against yeast
isolates in a Turkish University Hospital and effect of time of reading. Diagn
Microbiol Infect Dis. 2000;38:101-7.
33. Thompson GR, 3rd, Wiederhold NP, Sutton DA, Fothergill A, Patterson TF.
In vitro activity of isavuconazole against Trichosporon, Rhodotorula, Geotrichum, Saccharomyces and Pichia species. J Antimicrob Chemother.
2009;64:79-83.
34. Febre N, Silva V, Medeiros EA, Wey SB, Colombo AL, Fischman O.
Microbiological characteristics of yeasts isolated from urinary tracts of intensive care unit patients undergoing urinary catheterization. J Clin
Microbiol. 1999;37:1584-6.
35. Mori Y, Hiraoka M, Katsu M, Tsukahara H, Mikami Y, Mayumi M. Marked renal damage in a child with hydronephrosis infected by Trichosporon asahii.
Pediatr Nephrol. 2005;20:234-6.
36. Wolf DG, Falk R, Hacham M, Theelen B, Boekhout T, Scorzetti G et al.
Multidrug-resistant Trichosporon asahii infection of nongranulocytopenic
patients in three intensive care units. J Clin Microbiol. 2001;39:4420-5.
Table 1 The distribution of Trichosporon species
Specimens from body sites (no.)
Blood CSF Peritoneal fluid Respiratory tract
secretion
aUrine Wound
discharge Nail
scrapings Others
bTrichosporon species (no.) 5 1 1 4 13 6 3 2
T. asahii (22) 5 1 1 4 8 3
T. montevideense (5) 2 2 1
T. cutaneum (2) 1 1
T. dermatis (2) 1 1
T. debeurumannianum (1) 1
T. faecale (1) 1
T. insectorum (1) 1
T. japonicum (1) 1
CSF, cerebral-spinal fluid.
aRespiratory tract secretion included three samples from sputum and one from Bronchial washing.
b
Other sources included one T. cutaneum from a stool sample, and T. faecale not specified.
Table 2 In vitro antifungal susceptibility testing results of 22 Trichosporon asahii and 13 non-T. asahii clinical isolates Antifungals
Cumulative isolates (%) of T. asahii and non-T. asahii susceptible at MICs (µg/mL) Methods
Amphotericin B
0.031 0.063 0.125 0.25 0.5 1 2 4 8 16 ND
TA NTA TA NTA TA NTA TA NTA TA NTA TA NTA TA NTA TA NTA TA NTA TA NTA
CLSI 24H 1(4.5) 1(7.7) 4(18.2) 3(23.1) 8(36.4) 12(92.3) 11(50) 18(81.8) 22(100) 13(100)
CLSI 48H 6(27.3) 2(15.4) 12(54.5) 9(69.2) 19(86.4) 11(84.6) 22(100) 12(92.3) 13(100)
CLSI 24HG 2(16.7) 4(33.3) 3(13.6) 17(77.3) 10(83.3) 21(95.5) 22(100) 22(100) 1
CLSI 48HG 2(16.7) 4(33.3) 4(18.2) 8(66.7) 17(77.3) 9(75) 20(90.9) 11(91.7) 21(95.5) 12(100) 22(100) 1
EUCAST 1(2.9) 4(11.4) 2(9.1) 8(28.6) 11(50) 10(76.9) 21(95.5) 13(100) 22(100)
Fluconazole 0.125 0.25 0.5 1 2 4 8 16 32 64 ND
TA NTA TA NTA TA NTA TA NTA TA NTA TA NTA TA NTA TA NTA TA NTA TA NTA
CLSI 24H 1(4.5) 3(13.6) 1(7.7) 14(63.6) 8(61.5) 19(86.4) 11(84.6) 21(95.5) 13(100) 22(100)
CLSI 48H 1(4.5) 2(9.1) 1(7.7) 5(22.7) 2(15.4) 18(81.8) 11(84.6) 21(95.5) 13(100) 22(100)
CLSI 24HG 2(9.1) 1(8.3) 2(16.7) 7(31.8) 5(41.7) 13(59.1) 8(66.7) 20(90.9) 12(100) 21(95.5) 22(100) 1
CLSI 48HG 2(9.1) 1(8.3) 5(22.7) 5(41.7) 15(68.2) 6(50) 20(90.9) 12(100) 21(95.5) 22(100) 1
EUCAST 1(4.5) 2(15.4) 3(13.6) 4(30.8) 15(68.3) 7(53.8) 19(86.4) 12(92.3) 22(100) 13(100)
Voriconazole 0.016 0.031 0.063 0.125 0.25 0.5 1 2 4 8 ND
TA NTA TA NTA TA NTA TA NTA TA NTA TA NTA TA NTA TA NTA TA NTA TA NTA
CLSI 24H 16(72.7) 7(53.8) 18(81.8) 8(61.5) 9(69.2) 19(86.4) 11(84.6) 20(90.9) 12(92.3) 13(100) 22(100)
CLSI 48H 7(31.8) 4(30.8) 19(86.4) 11(84.6) 20(90.9) 12(92.3) 13(100) 21(95.5) 22(100)
CLSI 24HG 1(8.3) 1(4.5) 2(16.7) 7(31.8) 6(50) 16(72.7) 10(83.3) 19(86.4) 12(100) 21(95.5) 22(100) 1
CLSI 48HG 1(8.3) 3(25) 5(22.7) 5(41.7) 15(68.2) 10(83.3) 18(81.8) 12(100) 19(86.4) 21(95.5) 22(100) 1
EUCAST 2(15.4) 3(23.1) 1(4.5) 6(46.2) 8(36.4) 10(76.9) 17(77.3) 12(92.3) 22(100) 13(100)
TA, Trichosporon asahii, NTA, non-Trichosporon asahii; CLSI 24H and CLSI 48H, the MIC results obtained by the CLSI method at 24h and 48h, respectively; CLSI 24HG and CLSI 48 HG, the MIC results obtained by the modified CLSI method at 24h and 48h, respectively; EUCAST, the MIC results obtained by the EUCAST method; ND, not determined.
Table 3 Intraclass correlation coefficients and agreement values between the MICs of 35 Trichosporon isolates obtained by different methods
Comparing methods Fluconazole Voriconazole Amphotericin B
ICC (95% CI) Agreement (%) ICC (95% CI) Agreement (%) ICC (95% CI) Agreement (%) CLSI 24H
vs CLSI 48H 0.92** (0.84–
0.96) 80.0 0.63* (0.27–
0.82) 77.14 0.27 (-0.11–
0.71) 57.14
CLSI 24HG 0.90** (0.74–
0.95) 67.65 0.12 (-0.58–
0.54) 20.59 0.22 (-0.15–
0.66) 58.82
CLSI 48HG 0.90** (0.70–
0.96) 58.82 0.18 (-0.43–
0.56) 20.59 0.06 (-0.64–
0.50) 44.12
EUCAST 0.65* (0.30–
0.82) 65.71 0.41* (-0.08–
0.69) 17.14 0.39* (-0.12–
0.68) 60.0
CLSI 48H
vs CLSI 24HG 0.84** (0.68–
0.92) 67.65 0.09 (-0.78–
0.54) 8.82 0.30 (-0.41–
0.65) 70.59
CLSI 48HG 0.86** (0.71–
0.93) 76.47 0.48* (0.16–
0.74) 8.82 0.24 (-0.37–
0.60) 55.88
EUCAST 0.44* (-0.10–
0.72) 74.29 0.23 (-0.53–
0.61) 8.57 0.25 (-0.46–
0.62) 48.57
*p<0.05; **p<0.01.
ICC, intraclass correlation coefficient; CI, confidence interval; CLSI 24H and CLSI 48H, the MIC results obtained by the CLSI method at 24h and 48h, respectively; CLSI 24HG and CLSI 48 HG, the MIC results obtained by the modified CLSI method at 24h and 48h, respectively;
EUCAST, the MIC results obtained by the EUCAST method.
Table 4 Clinical Characteristics of 13 patients colonized/infected by Trichosporon asahii isolates Isolates Source Flu
aVor Amp Flu
dose/day Underlying
Disease Hospitalization Age (y) Catheter Duration of
hospitalization
bOutcome
YM060224 Blood 2 0.0313 0.5 100 mg Breast cancer Yes 54 IV 16 Discharge
YM060229 Blood 2 0.0156 1 400 mg Lung cancer Yes 61 IV 0 Discharge
YM060236 Blood 2 0.0313 0.25 200 mg Lung cancer Yes 61 IV 2 Discharge
YM060376 Blood 0.25 0.0625 0.5 200 mg Dialysis and transplantation,
TB
Yes 41 Double lumen 3 Discharge
YM020442 CSF 16 8 1 no Hypertension,
CVA ICU 66 CVP, IV, NG,
Endotracheal, Arterial line, Penrose drain
11 Discharge
YM020748 Peritoneal
fluid 2 0.0313 0.125 no RHD,
esophageal ulcer, DM, Hypentension
Yes 71 Pig tail 4 Dead
YM060744 Pus 2 0.0313 0.125 no DM,
hypertension Yes 56 IV 8 Discharge
YM060749 Sputum 4 0.0313 0.125 no DM,
hypertension Yes 70 IV 11 Discharge
YM060753 Sputum 2 0.0313 1 200 mg TB, DM Yes 71 CVP, IV, NG, Foley,
Arterial line 8 Discharge
YM020305 Urine 2 0.0156 0.25 no DM, CVA ICU 47 CVP, IV, NG,
Endotracheal, Foley 32 Dead
YM060733 Urine 2 0.0313 0.125 no Hypertension,
Dementia, BPH Yes 80 IV 1 Discharge
YM060734 Urine 4 0.0313 0.125 no DU, CVA ICU 81 no 0 Dead
Ym990296 Bronchial
washing 1 0.0156 0.5 unknown TB ICU 73 Tracheostomy,
Endotracheal, Foley, Pig tail
16 Discharge
Ym990597 Urine 2 0.0156 0.125 no none No 32 no 0 Discharge
a