Research Express@NCKU Volume 9 Issue 2 - June 5, 2009 [ http://research.ncku.edu.tw/re/articles/e/20090605/4.html ]
How to defense virus infection in fish: grouper
Mx confers resistance to nodavirus and
interacts with coat protein
Young-Mao Chen
a, Yung-Lin Su
a, Pei-Shiuan Shie
a, Shao-Ling Huang
a,
Huey-Lang Yang
a, b, c, and Tzong-Yueh Chen
a, b, c,*aInstitute of Biotechnology, College of Bioscience and Biotechnology bResearch Center of Ocean Environment and Technology
cAgriculture Biotechnology Research Center, National Cheng Kung University, Tainan,
70101, Taiwan
ibcty@mail.ncku.edu.tw
Develepmental and Comparative Immunology 32:825-836, 2008
I
n recent years, with the rapidly developing aquaculture industry, viral diseases have affected severely many highly valued species such as grouper causing heavy economic losses in Taiwan and SE Asia. Until now, one bottleneck of the grouper farming industry is the control of fish disease, especially the larva stage of grouper that have hazarded by the virus disease NNV (nerves necrosis virus), these diseases on grouper larva cause over 99.7% fatality rate. But we know that develop the grouper vaccines or increase immunity could be prevent the diseases.Fish interferons (IFNs) are the main cytokines induced in the innate immune response directed against viral infection. Gene products regulated by IFNs are the major effectors of IFN-mediated biological actions; antiviral
products among the IFN-stimulated genes include dsRNA-activated protein kinase (PKR), guanylate binding protein (GBP), Mx proteins, and 2′-5′ -oligoadenylate synthetase (OAS). These IFN-inducible antiviral proteins inhibit viral replication at the levels of penetration, uncoating, mRNA synthesis, protein synthesis, and assembly. One of IFN-inducible antiviral proteins, Mx proteins are members of the
superfamily of dynamin-like GTPases involved in intracellular membrane remodeling and intracellular trafficking, and whose primary function is the propensity to self-assemble into defined structures capable of binding activity. They seem to act as force-generating molecules capable of self-assembly into rings and stacks of interconnected rings in solution. Mx proteins have been used as molecular markers for IFN production and virus infection in mammals and fish. In addition, antiviral pathways of Mx-mediated inhibition of viruses have been identified, in which one of the pathways involving the intrinsic structure of fish Mx species contributes to the intracellular localization of Mx proteins, with a differing antiviral spectrum of Mx proteins. The viral protein partner in the Mx-virus interaction correlates with their specificity of antiviral activity. Various cellular factors that are dependent on the cell type in which the factor is expressed might account for the contrasting results of antiviral activity. Grouper Mx (gMx) proteins can inhibit nodavirus propagation. However, molecular mechanism of this antiviral function is unknown, and the viral target is still poorly characterized.
cells, and interferes with viral antigens in fish system. Grouper cells that overexpress Mx are highly resistant to viral infection. However, the role and the antiviral mechanisms of gMx protein in host cell defenses against nodavirus infection in grouper cells remains unclear.
Our team focuses at the host-virus interaction study over six years. Piscine nodavirus, a member of the Betanodavirdae family, is the causative agent of viral nervous necrosis (fish encephalitis) that results in high mortality rates in hatchery-reared larvae and juveniles of marine fishes in Taiwan, Japan, Australia, and Europe. Betanodaviruses are neuropathogenic and inflicts conspicuous damage characterized by
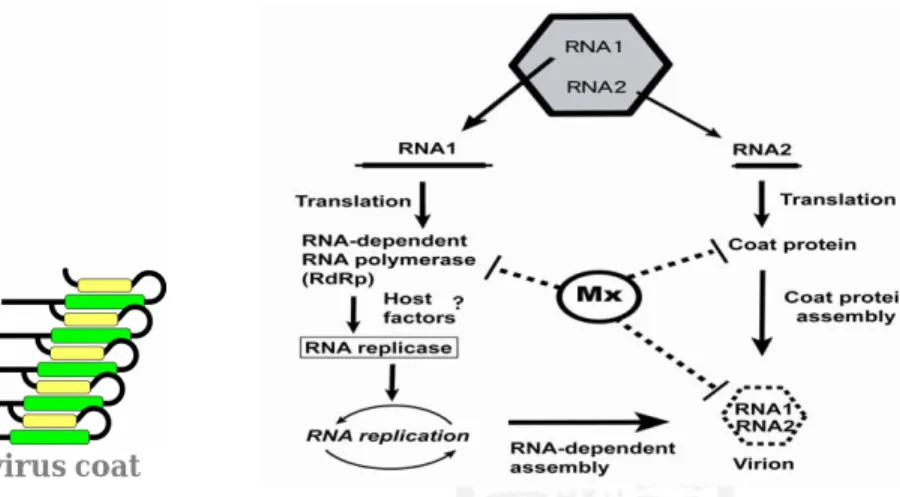
vacuolation and degeneration of neurons throughout the central nervous system. This piscine nodavirus is a non-enveloped, icosahedral capsid with a genome comprised of two positive-sense single-stranded RNA strands: 3.1 kb RNA1 and 1.4 kb RNA2. RNA1 encodes an RNA-dependent RNA polymerase (RdRp), while RNA2 encodes coat protein. In addition, a subgenomic RNA3 transcribed from the 3’ end of RNA1 encodes B2 protein. We found nodavirus coat protein which is multifunction, multidomain protein that is the focus of intense study as an effector for numerous viral functions and the induction of molecular processes. Nodavirus coat protein influences viral self-assembly and budding during the latter stages of viral
replication. Coat protein contains the signal required for nucleolar localization; through virus infection into the cells, coat protein was demonstrated to spread within the nucleolus and cytoplasm. Correspondingly, the transport machinery of the host assists virus replication. Grouper nervous necrosis virus coat protein contains three domains: (i) basic amino acids residues in the N-terminal region comprising residues 1-50, which may be involved in the protein-RNA interaction necessary for encapsidation, (ii) N-terminal domain residues 83-216 that form a β-sandwich that provides structural scaffolding, and (iii) residues 217 to the C-terminus, which likely form a surface-protruding domain involved in specific biological processes such as host cell recognition. However, while coat proteins are postulated to move from the endoplasmic reticulum through the nuclear pore complex to the nucleolus, little is actually known about the transport process. Several functions of Mx protein may contribute to the link between reduced viral yields and protein association. Fish interferon-induced proteins are a downstream effector molecule of the diverse biological actions of interferons and are thought to mediate many regulatory functions toward antiviral responses. In the previous study, we have been cloned the full nucleotide and amino acid sequences of an Epinephelus
coioides Mx cDNA and promoter from nodavirus-infected grouper. The grouper Mx gene has been shown to
be inducible in vivo by injection of live fish with fish pathogen nodavirus. The expression of Mx mRNA from healthy grouper by RT-PCR showed that the Mx mRNA was constitutively expressed at low levels in the eye, gills and heart, and minimally expressed in blood, brain, kidney, spleen, muscle, liver and
intestine. The gMx gene transcript began to increase 6 h after nodavirus injection and peaked at 72 h in the brain. The levels of expression of gMx from brain of a nerves necrosis virus (NNV)-infected grouper over a 96 hours time course. However, the molecular mechanisms of gMx for antiviral function in grouper are less studied and unclear, these problems will be solved.
Fig. 1. Immunological subcellular localization of grouper Mx proteins in nodavirus infected or poly[I:C] induced Mx expressing grouper cells. Grouper Mx proteins
were inducible after treatment with either
nodavirus (MOI=0.1) or poly[I:C] (0.1 mg mL-1) for
24 h. Cells were fixed, stained with polyclonal rabbit anti-grouper Mx and Texas-Red 594 labeled sheep anti-rabbit immunoglobulins.
effect on nodavirus infection (Fig.2). These results show that the promoter activity of gMx protein could be inducible by poly[I:C] and may have the antivirus activity. In this study, we explored the possibility that grouper Mx carried out its physiological function by participating in or interfering with translocation events. Based on the results supported the Mx protein may interact with the proteins in NNV. Specifically, we investigated the hypothesis that grouper Mx-related induction expression is involved in coat protein binding and perturbance of its intracellular localization. The N-terminus of coat protein contains a functional nucleolus localization signal (NLS) that is required for many of its activities including viral RNA replication, package signal, translocation, and virion assembly. We used the Far-western blotting to identify NNV proteins which
Fig. 2. Nodavirus activity of the mx-expressing clones. (A) Cells were seeded in a 96-well plate
until subconfluent growth developed before stimulation with 0.1–10 μg mL-1 poly [I:C] directly added
to culture medium for 24 h before challenge with nodavirus. Cell viability was determined by MTT assay. (B) Cells grown in 96-well plates were lipofectin-transfected with 1–2 μg mL-1 of poly [I:C] for 24 h
before challenge with nodavirus. Cell viability was determined four days later using the MTT assay. Two independent experiments were performed in triplicate. (C) Stable Mx-expressing clones were obtained via transfection with the pcDNA3.1mx-gfp and pcDNA3.1mx-his6 constructs and subsequent neomycin selection. The expression of Mx protein fused to GFP, and V5/His epitope-tagged grouper Mx proteins was determined in GF-1 cells. Western blots were performed to detect expression of the GFP and Mx-His6 proteins. The asterisk to the right of the figure indicates an unidentified cross-reacting protein, and
nodavirus, the Mx-expressing clone (pcDNA3.1mx-gfp), permanently Mx-expressing clone
(pcDNA3.1mx-his6), and untreated cells (control) were seeded in 96-well plates until subconfluence and cell viability was determined using the MTT assay. Two independent experiments were performed in triplicate. (E) Growth rates of Mx-expressing cell lines. Equal numbers of cells from each line were seeded onto 24-well plates. Cell were trypsinized and counted from three wells for each line every day after plating. (F) Viability of Mx-expressing cells after nodavirus infection. At each time point, viability was determined by the trypan blue exclusion method. GF-1 cells were used as a control (squares) for viability in cells treated with nodavirus. The stable clones of expressing Mx were GF-1 cells containing the grouper mx-gfp expression plasmid are indicated by the triangles and mx-his6 expression plasmid are denoted by circles. In panels E and F, the data is representative of one of three independent experiments (mean ± SEM).
Fig. 3. Interaction between nodavirus coat
protein and grouper Mx protein. Fig. 4. The potential anti-nodavirus mechanism of grouper Mx.
Over the past years, piscine nodavirus has devastated the grouper (Epinephelus spp.) culture industry in Taiwan and other Asian countries and so a better understanding of fish natural defense mechanisms against such pathogens is needed. We hope that knowledge of grouper Mx protein will contribute to the
![Fig. 1. Immunological subcellular localization of grouper Mx proteins in nodavirus infected or poly[I:C] induced Mx expressing grouper cells](https://thumb-ap.123doks.com/thumbv2/9libinfo/9027020.312878/3.918.485.818.22.181/immunological-subcellular-localization-proteins-nodavirus-infected-induced-expressing.webp)
![Fig. 2. Nodavirus activity of the mx-expressing clones. (A) Cells were seeded in a 96-well plate until subconfluent growth developed before stimulation with 0.1–10 μg mL -1 poly [I:C] directly added to culture medium for 24 h before challenge with noda](https://thumb-ap.123doks.com/thumbv2/9libinfo/9027020.312878/4.918.95.795.57.894/nodavirus-activity-expressing-subconfluent-developed-stimulation-directly-challenge.webp)