明新科技大學 校內專題研究計畫成果報告
以蛋白質體觀點研究運動訓練對攜鈣蛋白質與骨質密度之
關係
Proteomic views of the relation between exercise training,
calcium binding proteins and bone mineral density
Proteomic views of the relation between exercise training, calcium
binding proteins and bone mineral density
Abstract
Background
The physical and financial burden of osteoporosis can substantially diminish quality of life. Clinical evidence indicates that physical activity can prevent osteoporosis by regulating bone maintenance and stimulating bone formation and mineral accumulation. The improved muscle strength and balance achieved by physical activity can reduce the risk of falls and fractures. Regular weight-bearing physical activity is widely recommended for adult women and may help increase bone mineral density (BMD). However, studies of the effects of impact intense training programs (ITP) on BMD and physiological functions have yielded conflicting evidence. This study investigated how intense physical training impacts bone mineral density (BMD) and physiological function in female basketball athletes.
Methods
Twenty-four female subjects aged 18 to 20 years old participated in this study. Twelve subjects were athletes receiving intense training program(ITP) and twelve control subjects were sedentary non-athletes (NON, n=12). Their regional (lumbar spine L2-4 and right hip) bone mineral densities (BMDs) were measured by dual-energy X-ray absorptiometry (DXA).
Body composition, physiological function (including liver function and hormone status) were measured. Student t-test, Pearson correlation were performed to determine significant predictors of BMD at right hip and L2-L4 sites.
Results
Sedentary females had lower BMD values in L2-L4 and right hip BMD sites than those undergoing intense basketball training.
Analysis revealed that only training intensity program and seniority were related to
L2-L4 and right hip BMDs.
Conclusions
第一章 緒論
前言 (研究背景與目的)
As a serious health problem, osteoporosis that diminishes quality of life and the associated bone fractures can be a substantial physical and financial burden[1]. Physical activity is known to prevent osteoporosis by regulating bone maintenance, stimulating bone formation and mineral accumulation, strengthening muscles and improving balance, all of which can reduce the overall risk of falls and fractures[2].
Borer reported that after 9-12 months of regular exercise, young adult women often show limited improvement in bone health, possibly because of high subject attrition rates, inadequate exercise intensity, duration or frequency or because the accumulation of bone mass may be at its natural peak at this stage of physical growth[3]. The important influence of hormones and diet on bone growth and health are well known. For example, recent reports described premature bone loss associated with dietary restriction and estradiol(E2) withdrawal in exercise-induced amenorrhoea[4-6].
Skeletal integrity in young females depends largely on menstrual function and impact exercise, and female athletes should be evaluated for possible negative effects of amenorrhea on bone health[7,8]. In a recent study, a 16-week weight training protocol to develop upper arm strength proved effective for increasing bone density at the proximal phalanges in healthy young males[9].
The bone health in basketball players and the effects of intense training program(ITP) on bone metabolism have not been investigated previously. The optimal physical activity level (intensity, frequency, duration and rate of progression) for enhancing BMD in females is still unknown. Weight-bearing exercise is important for increasing BMD, and such activities should be dynamic, variable, performed rapidly and intermittently, and they should require relatively few loading cycles[6]. Although several effective interventions have been designed for improving bone mass, further research is needed to define specific exercise programs for optimizing bone structure and strength during growth. Further work is also needed to determine whether ITP exercise-induced alterations in bone mass and structure during growth are maintained into old age when fractures occur.
5
第二章 研究方法與步驟
Subjects
This study analyzed twenty-four females aged 18 to 20 years. The sample group included high school athletes (n=12) while the control group was comprised of non-athletic females (n=12). Inclusion criteria were that the female basketball athletes had participated in high-intensity activities(over 5 years of athletic activities). The measured parameters included body composition, L2- L4 BMD, right hip BMD, renal function, liver function and hormone status. The protocol was approved by human subject committee, Chinese Cultural University. All subjects signed a written consent after the procedure and possible risks had been clearly explained.
Anthropometric measurement of body composition
Anthropometric measurements were based on conventional criteria. Body weight (Wt) and height (Ht) were estimated to the nearest 0.1 kg and 0.5cm, respectively. Finally, body mass index (BMI) was calculated as body weight(kg)/body height(m)2.
ITP program activity
During the five years period, all subjects performed running activity fast walking 1h/d , jumping program 1h/d and weight-bear running activity 1h/d. The three hours of intense training was performed 5d or 6d per week . Female basketball athletes had participated in over five years high intensity activities.
Renal and liver function
From each subject, twenty-four blood samples were drawn from the antecubital vein while in the seated position. Routine complete blood counts (CBC) were taken using a Sysmex-E9000 (TOA Electronic, Inc., Tokyo, Japan), and renal and liver function tests were performed using a Hitachi 7170 instrument (Hitachi Electronic, Inc., Tokyo, Japan) by clinical chemistry laboratory staff at Cardinal Tien Hospital, Taipei County, Taiwan.
7
In each subject, triiodothyronine (T3), thyroxine (T4) were assayed in basal conditions using commercial enzymeimmunoassay(EIA) kits (CIS Diagnostics, Tronzano Vercellese-Tonino; RADIM, Pomezia-Roma, Italy; ICN Biomedicals, Inc., Costa Mesa, CA, USA).
BMD determination
The L2-L4 and right hip (at femoral neck site) BMD were measured via DXA (Norland, Norland excell, Norland Corp., WI, USA) for bone mineral densitometry, and all BMD values were also expressed as a T-score accurately reflecting BMD. The scanners were calibrated daily against the standard manufacturer-supplied calibration block to control baseline drift.
Statistical analysis
第三章 結果與討論
Results
General Characteristics
Anthropometric variables did not significantly differ between the ITP and NON groups. The ITP group underwent weight-bearing training consisting primarily of running, fast walking, jumping, maneuvering on hard surfaces and uphill running (15-18 h/wk). The duration of training over five years. Summarises the participant characteristics for all variables. No significant relationships (p>0.05) were found between two groups. Shows the BMD measurements for L2-L4 and right hip sites, , indicating that the ITP group had significantly higher BMD at both sites than the NON group.
Evaluation of physiological parameters
Further, the twenty-four females enrolled in this cross-sectional study did not significantly differ in terms of T3, T4, Alk-P, total protien or γ-GTP (Table 2).
Evaluation of BMD
Confounding factors were identified by using a stepwise linear regression procedure. Therefore, for correlation analysis, found to affect BMD at two sites were included in the subsequent analysis.
Discussion & Conclusions
Increasing BMD and preventing osteoporosis is vital for stopping or decelerating bone loss during future postmenopause[5-8]. This article describes the results of long-term intense training (ITP) in female college basketball athletes. The exercise program for these athletes emphasized low-volume weight-bearing training over five years period.
L2-L4(p<0.05 ) and hip sites(p<0.05). ITP-1 had higher BMD than ITP-2 groups. Most BMD benefits achieved by intense training or exercise during growth periods are lost when physical activity ceases[10, 11].Intense training five years, after this period of high intense activity (five years of ITP), may retain higher BMD benefits into old age. [10, 13].A reduced rate of fragility fracture in the athlete group may have been achieved because the participants had physically active lifestyles and high activity levels during training and competition, which would then increase BMD and reduce fractures[14, 15, 16].
A previous report demonstrated that multipurpose ITP programs significantly affect relevant menopausal risk factors and may therefore provide an alternative to hormone replacement therapy[16, 21].
According to the data in this study, female athletes who undergo intense training generate mechanical constraints on the body which can causes changes in bone metabolism such as increased BMD (both L2-L4 and right hip sites). The findings of similar studies elsewhere are heterogenous and depend on exercise intensity, duration and frequency[22, 27].
The two groups in this study did not differ in hormone status (in terms of T3, T4, alkaline phosphatase, total protein and r-GTP). Resistance exercise had a positive treatment effect and may help prevent skeletal change[23]. No significant relationships were observed between physiological function parameters and any measured BMD parameters in weight-bearing high impact training athletes and sedentary controls.
Study by Soot also had found that sedentary females of normal weight had lower (p<0.05 ) BMDs in sites L2-L4 and the femoral neck in comparison to females who had undergone weight-bearing ITP. This observation suggests that intense training, type of mechanical load and higher BMD have positive effects in female college basketball athletes compared to sedentary controls[12, 13]. The data for BMDs in sites L2-L4, femoral neck and total BMD of sedentary subjects in comparison to females who had undergone strength training, correlated with the results reported here.
However, the biological explanation for the reduced incidence of fracture is unclear. Proposed explanations include residual benefits to bone structural properties, muscle strength, coordination and balance[22, 27].
Each of these traits can be maintained in formally trained athletes after their active career and may help to reduce the number of fractures later in life. Therefore, based on the current evidence, a physically active lifestyle during growth stage prevents fragility fractures in old age[12, 16].
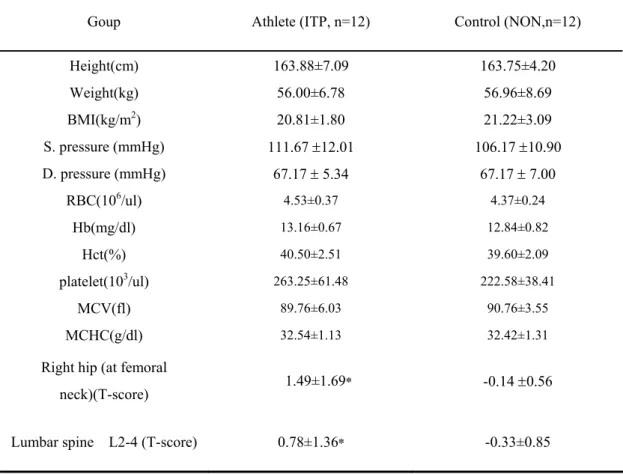
Table 1 Summarises the participant characteristics for all variables.
No significant relationships (p>0.05) were found between two groups. Hematologic parameters of the tested individuals. Parameters related to hemogram and no significantly were found in two groups. BMD in athletes with significance by independent t-test as compared with control group.The athletes group also had a significantly increased level of right hip and lumbar spine, P<0.05.
Goup Athlete (ITP, n=12) Control (NON,n=12)
Height(cm) 163.88±7.09 163.75±4.20 Weight(kg) 56.00±6.78 56.96±8.69 BMI(kg/m2) 20.81±1.80 21.22±3.09 S. pressure (mmHg) 111.67 ±12.01 106.17 ±10.90 D. pressure (mmHg) 67.17 ± 5.34 67.17 ± 7.00 RBC(106/ul) 4.53±0.37 4.37±0.24 Hb(mg/dl) 13.16±0.67 12.84±0.82 Hct(%) 40.50±2.51 39.60±2.09 platelet(103/ul) 263.25±61.48 222.58±38.41 MCV(fl) 89.76±6.03 90.76±3.55 MCHC(g/dl) 32.54±1.13 32.42±1.31
Right hip (at femoral
neck)(T-score) 1.49±1.69* -0.14 ±0.56
Lumbar spine L2-4 (T-score) 0.78±1.36* -0.33±0.85
Table 2 Description of the serum measurements of athlete and control groups. No significance were found compare athlete group than control group. Evaluation of
physiological parameters and hormone status. The twenty-four female athletes enrolled in this cross- sectional study did not differ significantly in terms of glutamate pyruvate
11
Athlete (ITP, n=12) Control (NON,n=12)
GPT(ALT)(IU/L) 15.83±4.41 15.42±4.23 BUN(mg/dl) 14.00±2.73 13.25±2.38 CRE(mg/dl) 1.01±0.07 1.03±0.08 CHO(mg/dl) 168.00±20.80 176.75±23.33 TG(mg/dl) 69.17±29.98 77.50±21.22 T3(ng/dl) 104.25±18.41 99.58±23.74 T4(ug/dl) 8.63±0.85 8.56±1.06 ALK-p(IU/L) 82.25±12.93 78.25±11.55 T-p(g/dl) 7.45±0.22 7.36±0.31 r-GTP(IU/L) 14.33±2.27 16.67±3.37
Table3 Correlation of Right hip, Lumbar Spine and Seniority.
The Lumbar spine L2-4(T-score) was significantly with Right hip BMD(T-score) in positive relation(r=0.576, p<0.05) and Seniority (r=0.781, p<0.05).
a
P < 0.05, correlation of Lumbar spine L2-4 with Right hip.
b
P < 0.05, correlation of Lumbar spine L2-4 with Seniority.
Lumbar spine L2-4 Rright hip Seniority
Lumbar spine L2-4
(T-score) 1 0.576* 0.781*
Right hip (femoral
參考文獻
1. Kemmler W, Engelke K, von Stengel S, Weineck J, Lauber D, Kalender WA:
Long-term four-year exercise has a positive effect on menopausal risk factors: the Erlangen Fitness Osteoporosis Prevention Study. J Strength Cond Res.
2007;21(1):232-9.
2. Karlsson MK: Does exercise during growth prevent fractures in later life? Med Sport Sci. 2007;51:121-36.
3. Borer KT: Physical activity in the prevention and amelioration of osteoporosis
in women : interaction of mechanical, hormonal and dietary factors. Sports
Med. 2005;35(9):779-830.
4. Martyn-St James M, Carroll S: Progressive high-intensity resistance training
and bone mineral density changes among premenopausal women: evidence of discordant site-specific skeletal effects. Sports Med. 2006;36(8):683-704.
5. Kemmler W, Engelke K, Lauber D, Weineck J, Hensen J, Kalender WA: Exercise
effects on fitness and bone mineral density in early postmenopausal women: 1-year EFOPS results. Med Sci Sports Exerc. 2002;34(12):2115-23.
6. Engelke K, Kemmler W, Lauber D, Beeskow C, Pintag R, Kalender WA:
Exercise maintains bone density at spine and hip EFOPS: a 3-year longitudinal study in early postmenopausal women. Osteoporos Int.
2006 ;17(1):133-42.
7. Kemmler W, Lauber D, Weineck J, Hensen J, Kalender W, Engelke K: Benefits
of 2 years of intense exercise on bone density, physical fitness, and blood lipids in early postmenopausal osteopenic women: results of the Erlangen Fitness Osteoporosis Prevention Study (EFOPS). Arch Intern Med. 2004.
8. Nichols JF, Rauh MJ, Barrack MT, Barkai HS:Bone mineral density in female high school athletes: interactions of menstrual function and type of
mechanical loading. Bone.2007;41(3):371-7
9. Bagrichevsky M, Martins-Filho J, Guerra-Junior G: Bone density gain at
proximal phalanges in healthy males aged 18-25 years after 16 weeks of upper-arm muscle weight training. J Sports Med Phys Fitness.
2007 ;47(4):437-42.
10. Muraki S, Yamamoto S, Ishibashi H, Oka H, Yoshimura N, Kawaguchi H,
Nakamura K: Diet and lifestyle associated with increased bone mineral density:
cross-sectional study of Japanese elderly women at an osteoporosis outpatient clinic. J Orthop Sci. 2007 ;12(4):317-20.
11. Martyn-St James M, Carroll S: High-intensity resistance training and
postmenopausal bone loss: a meta-analysis. Osteoporos Int. 2006 Jun 1[Epub
12. Nordström A, Karlsson C, Nyquist F, Olsson T, Nordström P, Karlsson M: Bone
loss and fracture risk after reduced physical activity. J Bone Miner Res.
2005;20(2):202-7.
13. Valdimarsson O, Alborg HG, Düppe H, Nyquist F, Karlsson M: Reduced
training is associated with increased loss of BMD. J Bone Mi
2005 ;20(6):906-12.
ner Res.
14. Sööt T, Jürimäe T, Jürimäe J: Areal bone density in young females with
different physical activity patterns: relationships with plasma leptin and body composition. J Sports Med Phys Fitness. 2007;47(1):65-9.
15. Sööt T, Jürimäe T, Jürimäe J: Relationships between bone mineral density,
insulin-like growth factor-1 and sex hormones in young females with different physical activity. J Sports Med Phys Fitness. 2006;46(2):293-7.
16. Yahata Y, Aoyagi K, Okano K, Yoshimi I, Kusano Y, Kobayashi M, Moji K, Takemoto T: Metacarpal bone mineral density, body mass index and lifestyle
among postmenopausal Japanese women: relationship of body mass index, physical activity, calcium intake, alcohol and smoking to bone mineral density: the Hizen-Oshima study. Tohoku J Exp Med. 2002;196(3):123-9.
17. Twiss JJ, Gross GJ, Waltman NL, Ott CD, Lindsey AM: Health behaviors in
breast cancer survivors experiencing bone loss. J Am Acad Nurse Pract.
2006;18(10):471-81.
18. Muraki S, Yamamoto S, Ishibashi H, Oka H, Yoshimura N, Kawaguchi H,
Nakamura K: Diet and lifestyle associated with increased bone mineral density:
cross-sectional study of Japanese elderly women at an osteoporosis outpatient clinic. J Orthop Sci. 2007;12(4):317-20.
19. Schulzke SM, Trachsel D, Patole SK: Physical activity programs for promoting
bone mineralization and growth in preterm infants. Cochrane Database Syst Rev. 2007;18;(2):CD005387.
20. Martyn-St James M, Carroll S: High-intensity resistance training and
postmenopausal bone loss: a meta-analysis. Osteoporos Int. 2006 Jun 1[Epub
ahead of print]
21. Daly RM: The effect of exercise on bone mass and structural geometry during
growth. Med Sport Sci. 2007;51:33-49.
22. Karlsson MK: Physical activity, skeletal health and fractures in a long term
perspective.J Musculoskelet Neuronal Interact. 2004;4(1):12-21.
23. Karlsson M: Has exercise an antifracture efficacy in women? Scand J Med Sci Sports. 2004;14(1):2-15.
24. Karlsson M: Exercise increases bone mass in children but only insignificantly
in adults. Lakartidningen. 2002;99(35):3400-5.
25. Shackelford LC, LeBlanc AD, Driscoll TB, Evans HJ, Rianon NJ, Smith SM, Spector E, Feeback DL, Lai D: Resistance exercise as a countermeasure to
disuse-induced bone loss. J Appl Physiol. 2004;97(1):119-29.
26. Layne JE, Nelson ME: The effects of progressive resistance training on bone
density: a review. Med Sci Sports Exerc. 1999;31(1):25-30.
27. Kontulainen S, Sievänen H, Kannus P, Pasanen M, Vuori I: Effect of long-term
impact-loading on mass, size, and estimated strength of humerus and radius of female racquet-sports players: a peripheral quantitative computed
tomography study between young and old starters and controls. J Bone Miner
Res. 2003;18(2):352-9.
28. Maïmoun L, Coste O, Puech AM, Peruchon E, Jaussent A, Paris F, Rossi M, Sultan C, Mariano-Goulart D: No negative impact of reduced leptin secretion
on bone metabolism in male decathletes. Eur J Appl Physiol. 2008;102(3):
343-51.
29. Rector RS, Rogers R, Ruebel M, Hinton PS: Participation in road cycling vs
running is associated with lower bone mineral density in men. Metabolism.