doi:10.1136/adc.68.3.408
1993;68;408-411 Arch. Dis. Child.
L M Huang, C Y Lee, M H Chang, J D Wang and C Y Hsu
cytomegalovirus, and human herpesvirus-6.
Primary infections of Epstein-Barr virus,
http://www.fetalneonatal.com/cgi/content/abstract/68/3/408
Updated information and services can be found at:
These include: References
http://www.fetalneonatal.com/cgi/content/abstract/68/3/408#otherarticles
1 online articles that cite this article can be accessed at:
Rapid responses
http://www.fetalneonatal.com/cgi/eletter-submit/68/3/408
You can respond to this article at:
service Email alerting
the top right corner of the article
Receive free email alerts when new articles cite this article - sign up in the box at
Notes
http://journals.bmj.com/cgi/reprintform
To order reprints of this article go to:
http://journals.bmj.com/subscriptions/
go to: Archives of Disease in Childhood To subscribe to
Primary
infections of
Epstein-Barr
virus,
cytomegalovirus,
and human herpesvirus-6
Li-Min Huang,Chin-YunLee,Mei-HweiChang, Jung-Der Wang, Ching-Ying Hsu
Abstract
A total of 121 infants entered a cohort sero-logicalstudy ofprimary infections with herpes-viruses. All of them had seven samples of blood available: the first sample was taken soonafterbirth, the other six were taken at 1, 2, 3, 6, 12,and 14 months of age. One sample of maternal blood was collected immediately afterdelivery. All blood samples weretested for antibodies against cytomegalovirus, Epstein-Barrvirus, and human herpesvirus-6 (HHV-6).
Primarycytomegalovirusinfectionoccurred
early; the cumulative infection rates were
1-7%,
8*3%,
18-3%, 25%,52*5%,
and 65% by the ages of 1, 2, 3, 6, 12, and 14 months, respectively. Epstein-Barrvirus infection was not seen before 3 months of age and slowlyemergedthereafter, reaching a cumulative rate of 1-7%, 11-6%, 21-5% at the ages of 6, 12, and 14months, respectively. Primary HHV-6 infectionwasalso arareeventin thefirst three months of life, butpeaked between 6 and 12 months of age. No detectable risk factorswere
associated withprimaryEpstein-Barrvirus or HHV-6infection. The risk factors associated
withcytomegalovirus infection includedbreast
feeding,fewerchildreninhousehold, andcare byababysitter.
(ArchDisChild 1993;68:408-411)
hope to understand better the biology and mode
ofspread ofthese three viruses.
Subjectsand methods
SUBJECTS
From January 1988, 121 newborns were recruited to enter a study for the efficacy of recombinanthepatitisBvaccine. Their mothers were all hepatitis B surface antigen (HBsAg)
positive; 109 of them were hepatitis e antigen
(HBeAg) positive.All infantshad receivedfour
doses of recombinant hepatitis B vaccines
(Engerix-B, SmithKline and Biologicals,
Belgium)at0, 1, 2, and 12 monthsofage.On the first day of life, 0 5 mlofhepatitis B
immuno-globulin was given to infants born to HBeAg
positive
mothers.
METHODS
Everybabywasbledonsevenoccasions: atbirth
(after hepatitisBimmunoglobulin injection)and
at 1, 2, 3, 6, 12, and 14 months ofage. The mother's blood was drawn soon after delivery.
Sera obtainedwerestoredat-20°Cuntil tested. The collectionofcaseinformation was detailed
before.6 CollegeofMedicine, National Taiwan University, Department of Paediatrics Li-MinHuang Chin-Yun Lee Mei-HweiChang Department of Public Health and Internal Medicine
Jung-DerWang Department of
Paediatrics, Taipei MunicipalMaternaland Child HealthHospital,
Taiwan
Chin-YingHsu
Correspondenceto:
DrLi-Min Huang,9000 RockvillePike,Room306, Building4, NIAID,National InstitutesofHealth,MD
20892,USA.
Accepted10September1992
There are at least six members of the human
herpesviridae: herpes simplex virus type I and
II, varicella zoster virus, cytomegalovirus,
Epstein-Barr virus, and human herpesvirus-6 (HHV-6).Allhumanherpesvirusesaresimilarin
structure andgeneralgenome layout. Theyare all capable of inducing human diseases and
maintaining a latent state in the host after
primary infection.' The primary infections of differentherpesviruses usuallyoccurinthe first
fewyearsoflife,butwithsubstantialdifferences
amongthem. In thisstudywefocusonthe latter three viruses: cytomegalovirus, Epstein-Barr virus, and HHV-6. Cytomegalovirus is an
important aetiological agent of congenital and
perinatal virus infections,23 to which
Epstein-Barr virus has been rarely linked.4 It has been
speculated that, in addition to environmental
factors,virusreactivationduringpregnancyand maternal antibody have important roles in the
timing ofprimary infection.' HHV-6 is a new member of human herpesviruses and was first characterised in 1986.1 Earlier we have shown that primary HHV-6 infections usually occur between six and 12 months of life.6 Here we
present the cohort study of cytomegalovirus,
Epstein-Barr virus, and HHV-6. Bycomparing
the different patterns ofprimary infections we
CONTROLS
A total of100 childrenof1 yearold, who were
randomly selected from our well baby clinic,
werebledonceafterinformedconsentgiven by
their parentsorguardians.Theirsera weretested
for antibodiesagainstcytomegalovirus,
Epstein-Barrvirus,and HHV-6toserve ascontrols.
ANTIBODYDETERMINATIONS
IgGandIgMantibodiesagainstcytomegalovirus (anticytomegalovirus) were tested by enzyme linked immunosorbent assay(ELISA). IgG
anti-cytomegalovirus was tested with a kit from Behring,Holland. The results wereexpressedin anticytomegalovirus titres according to the
manufacturer's instruction and titres lower than
100 were considered negative. IgM anticyto-megaloviruswastestedwithakitfor Whittaker
M A Bioproduct, Maryland, USA. The result
wasexpressedaspositiveornegative.
IgG antibodyagainstEpstein-Barrvirus
(anti-Epstein-Barr virus) viral capsid antigen was
tested by indirect immunofluorescence
assay.7
P3HR-1
cellswereinducedby5'-iododeoxyuri-dine and then were fixed onto slides with cold
acetone.Theremainingproceduresfollowed the standard immunofluorescence assay. IgG
Primary infections ofEpstein-Barr virus,cytomegalovirus, and human herpesvirus-6
Table1 Persistence timeof maternally derived antibodies.Figuresinparentheses show
cumulative percentage.Some infantswereinfected bycytomegalovirusbefore complete disappearance ofmaternalderivedcytomegalovirus antibodies. Theseinfantswere notincluded
in the evaluationforcytomegalovirus in this table
Persistence time(months)
0-1 1-2 2-3 3-6 6-12 12-14 >14 Epstein-Barr virus 6(5) 26(28) 30(54) 37(87) 10(96) 3(98) 2(100) Cytomegalovirus 1(1) 8(12) 14(29) 35(74) 9(86) 6(94) 5(100)
HHV-6 28(32) 28 (64) 16(83) 15(100) 0(100) 0(100) 0(100)
tested by indirect immunofluorescence assay and has beendetailedbefore.8Thereciprocal of the highest serum dilution that gave positive fluorescence stain was taken as the titre of that serum. A titre lower than 10 was defined as
seronegative.
RISKFACTORS
In the identification of risk factors, life table analysis was usedforunivariateanalysisandCox regression formultivariateanalysis. The
follow-ing factors were examined: (1) sex, (2) birth body weight, (3) Apgar scores, (4) agesof father and mother,(5)socioeconomic classes of parents, (6) breastfeeding,(7) maternalserological statusfor
HHV-6, Epstein-Barr virus, and cytomegalo-virus, (8) the caregiver- that is, who the baby was taken care ofby mainly- mother, grand-parents, or babysitter, (9) administration of
hepatitis B immunoglobulin, (10) number of
exposed children including those in the home and ofthe babysitter, (11) household density (spaceofhome divided by the numberof people living there), (12) use of air conditioner, (13) behaviour facilitating the exchange ofsaliva as estimated by the frequency the baby put its
fingersinto another person's oral cavity, and (14) numberofneedle injections.
Results
MOTHERS' ANDMATERNALLY DERIVED ANTIBODIES
Almost all the mothers were seropositive for cytomegalovirus (117/121; 96 7%) and
Epstein-0) 4 -m c ._ 0 4._ 0) E) E 0 1*0_ 0-9 _ 0-8 0-7 06 0-5 0-4 0-3 02 0*1 HHV-6 ...
.Epstein-Barr
virus ---Cytomegalovirus 64-5% 52-5%70.2%
165 0% _ _ _ _ _ _J1 - 250% -16-3% r
-
21-5% - -- 13-2% 8-3 . ... 11-6% IT I I I IIBarrvirus(115/121; 95%)while 80%(97/121)of mothers were seropositive for HHV-6. About
half of babies were born with antibody titres against these three viruses that were equal to
maternal titres. Ofthe remaining half, roughly
onequarterof babies hadhigherandonequarter had lower antibody titres compared with their mothers.
Maternally derived HHV-6 antibody
dis-appearedatthefastestrate(table1);bytheageof 6months,allbabieslost theirpassiveIgG anti-HHV-6. Maternally derived antibodies against
Epstein-Barrvirus andcytomegalovirus decayed
at a slower rate; the median durations for maternally derived antibodies against
Epstein-Barr virus andcytomegalovirus to persistwere
both between threeand six months.
TIMEOFPRIMARYINFECTION
The timeofprimary infectionsis shown infig1.
All cases ofprimary HHV-6 and Epstein-Barr
virus infectionswere diagnosed by
seroconver-sion, thatis, fromseronegative to seropositive. Seventy nine children wereinfected with
cyto-megalovirusby the age of 14months;39
serocon-vertedagainstcytomegalovirusand40developed rising anticytomegalovirus titresin consecutive
serum samples. In the 40 cases diagnosed by rising anticytomegalovirus titres, 23 of them hadapositiveIgManticytomegalovirus. A
two-foldrise incytomegalovirustitreswasconsidered
significant. Fivecaseshadatitrerisingbetween 1-3 to twofold and also without positive IgM
antibody. The sera from these five cases were tested two to three times to make sure of the
antibody rising. One possible reason for the
antibody rising was because of cross reaction
to other viruses such as Epstein-Barr virus or
HHV-6. But no Epstein-Barr virus or HHV-6
infectionwasnoted in thatspecifictimeinterval,
thusmakingcrossreaction lesslikely. Theywere all seropositive against cytomegalovirus up to
14monthsofage, thatwasalso lesslikely from
their titresimmediately afterbirth.Considering
all theevidence,wediagnosedthese fivecasesas
havingcytomegalovirus infection.
No primary HHV-6 infection was noted before 2 monthsofage nor was there
Epstein-Barr virus infection before 3 months of age.
Primary cytomegalovirus infection occurred early, and by the age of 3 months 18-3% of infants had been infected and the proportion increased to 25% by 6 months of age. The
monthlyinfectionrates areshown in table2.The
highest risk period for cytomegalovirusinfection
was between 1 and 3 months of age; that for
HHV-6 was between 6 and 12 months of age. The number of primary Epstein-Barr virus
infectionsslowlyincreasedfrom3monthsofage onward.
Table 2 Monthly infectionratesofEpstein-Barrvirus,
cytomegalovirus,and HHV-6(percent)
Ageinterval(months)
0-1 1-2 2-3 3-6 6-12 12-14 Epstein-Barrvirus 0 0 0 0-6 1-7 2-5 Cytomegalovirus 1-7 6-6 10 2-2 4-6 6 3 HHV-6 0 0 1 4 8-5 3 - 1 2 3 4 5 6 7 8 9 10 11 121314 Age(months)
FigureI Cumulativeratesofcytomegalovirus, Epstein-Barrvirus,andHHV-6infections.
V.V. . . .. . . -.- -.- -.- -.-
-.-409
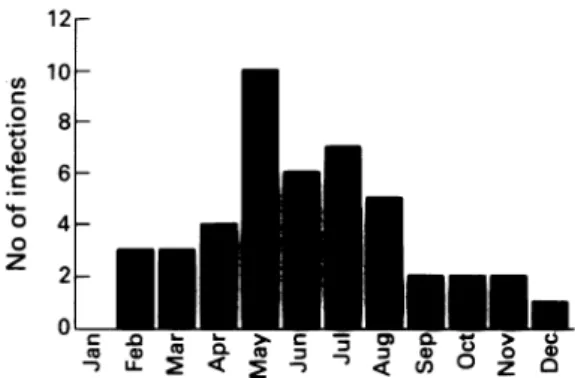
12r 0 G) a) %._ 0 0 z 10 8 6 4 2 01 C Dl X- X- >.C , > 0 a) M.L M -) 0) 0 )
Figure2 Monthly distribution ofprimary cytomegalovirus infections.
MONTHLYDISTRIBUTION OF CYTOMEGALOVIRUS INFECTION
Except for the infections occurring between 6
and 12 months of age, we could accurately
estimate the time ofprimary infection.
There-fore, it is possible to study the monthly
dis-tribution. As the number of cases of primary
Epstein-Barr virus infectionwassmall andmost
HHV-6 infections occurred between 6 and 12 months ofage,only the monthly distribution of
primary cytomegalovirus infections was
calcu-lated. This result is shown infig 2. We observed
a clustering ofprimary cytomegalovirus
infec-tions from ApriltoAugust.
PREVALENCE OF SEROPOSITIVITY IN THECONTROL
GROUP
A total of 100 childrenaged 1 yearentered this
studyas acontrolgroup. ThehepatitisBstatus
of their mothers was unknown and they were chosenon arandom base. Theseropositiverates
for Epstein-Barr virus and cytomegalovirus in
these 100 children were 19% and 56%
respec-tively. Thesefigureswerecloseto the
seroposi-tive rates in study infants at 1 year of age,
15-7% and 64-2% for Epstein-Barr virus and
cytomegalovirusrespectively.The differences of
seropositive ratesbetweenstudycases and
con-trolswerenotstatisticallysignificant (p=0-64 for
Epstein-Barr virus and0-27forcytomegalovirus by X2 test with Yates's correction). The
differ-encesof theantibody titreswere notsignificant
either forcytomegalovirus(p=0-12by Student's
ttest after logarithmic transformation and p=
0-62byWilcoxon ranksumtest)orEpstein-Barr virus (p=0-54 by Student'st testafter
logarith-mic transformation and p=0-86 by Wilcoxon rank rum test). The two groups were also
comparablewithregardstothestatusofHHV-6
antibody.6
RISK FACTORS OF PRIMARY CYTOMEGALOVIRUS, EPSTEIN-BARRVIRUS, AND HHV-6 INFECTION
Wedid notfindanyrisk factor associated with eitherprimaryHHV-66orprimary Epstein-Barr virus infection. Life table analysis using the
generalised Wilcoxon testidentified the
follow-ing factors, which were associated with the
primary cytomegalovirus infection: (1) breast
feeding (p=0-0006), (2) carer (highest risk for those cared forbyababysitterand lowest riskby
mother, p=0-017), (3) number of children in
household(fewerchildren associated with higher
risk, p=0 03), and (4) household density(high density having higher risk,p=0 03).
Cox regression was used for multivariate analysis andsignificant factors entered into the model were breastfeeding (odds ratio 2-18, 95% confidence interval(CI) 1-3to3-66),number of children in household (odds ratio 0-75 for per extra one child, 95% CI 0-58 to 0-96), and a dummy variable of carer as grandparents against babysitter (risk higher for caretaker being baby-sitter than being grandparents; odds ratio 2-43, 95% CI 1-05 to5-64).
Discussion
Inthis study three distinct patterns of primary herpesvirus infection were observed. A substan-tialproportion (18-3%)of primary cytomegalo-virus infections occurred in the first three months oflife, then the risk of cytomegalovirus infection levelledoff by the age of 14 months. PrimaryEpstein-Barr virus infections occurred muchlater than that ofcytomegalovirus. During the first three months oflife no Epstein-Barr virus infection was noted; after that, the risk of Epstein-Barr virus infectionroseslowly and was maintained at a low levelat 14 months ofage. The pattern of primary HHV-6 infection differed from that of Epstein-Barr virus or cytomegalovirus infections. The infection of HHV-6 occurred late, similar to Epstein-Barr
virus,butsurged between6and12months of age (8- 5% of infants infected each month). Two thirds of theinfants had beeninfectedbythe age of 1 year and the risk of HHV-6 infection droppedto3% between 12 and14months ofage. As the host factors were the same, extrinsic
factors wereresponsible for the three different patterns.
We suggest that the passive antibody the infant receives from itsmother,the time for the infantto encounterthevirus,and theamountof virus theinfant isexposedto, all determine the infection patterns. As humanherpesviruses can-not survive long in the environment or on a
fomite, direct contact with human excretion is necessary for virus spread.' The first possible
contact happenswhenthe babypasses through
the birth canal and the second major one is
ingestionof breast milk. A virus present in the birth canal and human breast milk would bean
important agent of congenital and perinatal
infections. Cytomegalovirus has been shown
to replicate well in the birth canal during the third trimester of pregnancy9 and exist in
human breast milk.'"0 The situations for
Epstein-Barr virus and HHV-6 are different;
excretionofEpstein-Barrvirus in the birth canal
and breast milk is minimal ornil.'2 3 Although
information about HHV-6 islimited, it hasbeen shownfromserologythat thereisnoreactivation ofHHV-6inlatepregnancy.'4Thesefactsmight
explain the few infections of Epstein-Barr virus andHHV-6 in the first three months of life. After thattime,the levelofmaternallyderived
antibody
and the amount of virusthe baby encountered determined the infection rate. Inthis study all maternally derived HHV-6 antibodies
Primary infections ofEpstein-Barr virus,cytomegalovirus, and human herpesvirus-6 411
derived Epstein-Barr virus antibody persisted muchlonger as shown in table 1. Normal adults usually excrete large amounts of HHV-6.'s 16 In one study, by using the polymerase chain reaction, it was shown that the excretion of HHV-6 in theoropharynxwasricher than thatof Epstein-Barr virus.'7 Early loss ofpassive anti-body and high doseofvirus in the environment could explain why such a large proportion of infants (50%) became infected with HHV-6 between 6 and 12 months, while persistent
passive antibody and relatively low amount of virus make theprimary Epstein-Barr virus
infec-tionlag behind.
Although most babies in thisstudywereborn to HBeAg positive HBsAg carriermothers, we
believed thattheyweresimilartootherchildren inrespect to primary Epstein-Barr virus,
cyto-megalovirus,and HHV-6infections.Agroupof randomly selected infants were shown to be similar to the study cases both in seropositive
rates andantibodytitres. Besides, the factthat
hepatitisB immunoglobulin injectionwas not a
significant factor fromeither univariateor multi-variate analysis also suggests that the status of
maternal hepatitis B infection and
immuno-globulin
injectiondid not have amajorinfluence.The threefactorssignificantlyassociatedwith primary cytomegalovirus infection in the Cox modelwerebreastfeeding,numberofchildren in the household, and carer. Breast feeding
increased theriskofcytomegalovirusinfection, a fact that has been reported before and may be due to the presenceof cytomegalovirus in breast milk and more intimate relationship between
babyanditsmother.'"I One ofthe reasonswhy
large number of childrenin the householdand having a grandparent as carer were associated with a lesser risk of cytomegalovirus infection might be the age of carer. The excretion of
cytomegalovirusin womenisinverselyrelatedto age after
puberty.'8
The genital excretion ofcytomegalovirus drops from15% in teenage girls
to anundetectable level in women older than 31 years. Urinary excretion of cytomegalovirus drops from 8% in younggirls to nil in women older than 26 years. No excretion of cytomegalo-virus in postmenopausal women is detected.
Usually grandparents areolder thanbabysitters
andwomenwith more children in the household
areolder than those with fewer.Therefore, it is
possible that these two risk factors in the Cox model project the effect of age of the person
directly takingcareofthe baby.
No risk factor was identified for primary
Epstein-Barr virus infection in this study. We
followedupthisgroupof children to 14 months
ofage andonly21-5%ofthem were infected with
Epstein-Barr virus. A study of older children is likely to be more appropriate in identifying risk factors associated with Epstein-Barr virus
infection.
We observed clustering of primary cyto-megalovirus infections from April through August. The connection of season and primary
cytomegalovirusinfection has not beenreported before.Thisprospective study designfacilitated theidentification of theexactonsetof infection. Previousstudiesusuallyrelied onthe reporting
of cases. The reason for this seasonal variation is not clear.Whether awarmtemperature activates the latent cytomegalovirus in seropositive
carriers or facilitates spread is unknown but is worthyof further study.
In summary, the three human herpesvirus studied, Epstein-Barr virus, cytomegalovirus,
and HHV-6 all behaved differently in causing primary infection. Cytomegalovirus caused infection soon after birth and the risk remained stable until the age of14 months. HHV-6 was mostprevalent when the baby was between 6 and 12monthsof age. Epstein-Barr virus was not a common pathogen before 14 months of age compared with cytomegalovirus and HHV-6. Possible decisivefactors might be reactivation in late pregnancy, level and duration of maternal
antibody,and the presenceof virus in saliva and breastmilk.
1 Straus SE.Introduction toherpesviridae. In: Mandell GL, DouglasRG, Bennett JE, eds. Principles andpracticeof infectious diseases. 3rd Ed. New York: Churchill
Living-stone, 1990:1139-44.
2 Griffith PD, Baboonian C. Aprospectivestudy ofprimary cytomegalovirus infection duringpregnancy: final report.
BrJObstet Gynaecol 1984;91:307-15.
3 ChandlerSH,AlexanderER, Holms KK.Epidemiology of
cytomegalovirusinfection in aheterogeneouspopulationof pregnantwomen.J Infect Dis1985;152: 249-95. 4ArvinAM, Yeager AS. Otherviralinfectionsof the fetus and
newborn. In: Remington JS, Klein JO, eds. Infectious diseasesofthefetusandnewborninfant.3rd Ed.Philadelphia:
WBSaunders,1990:517-9.
5 SalahuddinSZ, Ablashi DV,MarkhamPD,etal.Isolationofa
newvirus, HBLV, in patients with lymphoproliferative
disorders.Science 1986;234: 596-601.
6Huang LM, Lee CY, Chen JY, et al. Primary human
herpesvirus-6 infectionsin children: a prospective study.
JInfectDis1992;165: 1163-4.
7 HinumaY,KonnM, Yamaguchi K,WudarskiDJ,Blakeslee
JR Jr, GraceJT Jr. Immunoflurescenceandherpes-type
particlein theP3HR-1Burkittlymphomacell line. J Iirol 1967;1:1045-51.
8Huang LM, LeeCY, Lin KS,etal. Humanherpesvirus-6 associated with fatal hemophagocvtic syndrome. Lancet
1990;336:60-1.
9Stagno S, Reynolds DW,Tsiantos A, etal. Cervical
cyto-megalovirusexcretion inpregnant and nonpregnant women: suppression inearly gestation.JInfectDis 1975; 131:522-7.
10Leinikki R, Heinonen K, Pettay0.Incidence of cytomegalo-virusinfectioninearlychildhood.ScandJ7Infect Dis1972;4: 1-5.
11 Stagno S, Reynolds DW,PassRF, AlfordCA. Breast milk and
the risk ofcytomegalovirus infection.NEngijIfed1980;
302: 1073-6.
12 VisintineAJ, Gerber P, Nahmias AJ.Leukocvtetransforming
agent (Epstein-Barr virus) in newborn infantsand older
individuals.JPediatr1976; 89: 571-5.
13Chang RS, SetoDY.PerinataltransmissionbyEpstein-Barr virus.Lancet1979; ii: 201.
14Balachandra K,Ayuthaya PIN,Auwanit W, et al. Prevalence ofantibodytohuman herpesvirus6in women and children. Microbiol Immunol 1989; 33: 515-8.
15 Pietroboni GR, Harnett GB, Bucens MR, Honess RW.
Antibodytohumanherpesvirus6 insaliva. Lancet 1988; i: 1059.
16 Levy JA, Ferro F, Greenspan D, Lennette ET. Frequent
isolation of HHV-6 fromsaliva andhighseroprevalenceof the virus in thepopulation.Lancet1990; 335: 1047-50. 17Gopal MR,Thomson BJ,FoxJ,Tedder RS,Honess RW.
Detection by PCR ofHHV-6and EBV DNA in blood and
oropharynxofhealthyadults andHIV-seropositives. Lancet 1990;i:1598-9.
18 KnoxGE, PassRF, Reynolds DW, Stagno S,Alford CA.
Comparative prevalence ofsubclinicalcytomegalovirusand
herpes simplexvirus infection in the genital andurinary
tracts of low income, urban women. J Infect Dis 1979; 140: 419-22.