Synovial sarcoma of the temporomandibular joint area: report
of a case
Chin-Wan Luo, DDS, MSD,
a,bChung-Ji Liu, DDS, MSD,
c,d,eand Kuo-Ming Chang, MD
fTaipei, Taiwan
TAIPEI MEDICAL UNIVERSITY HOSPITAL, MACKAY MEMORIAL HOSPITAL, NATIONAL YANG-MING UNIVERSITY, AND MACKAY MEDICINE, NURSING AND MANAGEMENT COLLEGE
Synovial cell sarcoma is a relatively rare tumor of mesenchymal origin. It is a high-grade neoplasm that microscopically shows a monophasic or biphasic cellular pattern and includes epithelial features as well as supporting tissue features. Surgical excision is the primary mode of treatment. Postoperative radiotherapy and chemotherapy also is seen to be helpful. Between 3% and 10% of cases originate in the head and neck. A review of relevant literature shows less than 10 cases of synovial cell sarcoma of the temporomandibular joint area reported in the English literature. We report an additional case of biphasic synovial cell sarcoma arising in the temporomandibular joint area, which caused ear pain, tinnitus, and hearing loss, and we further discuss the clinical features, histopathology,
differential diagnosis, and treatment modality. (Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2007;104:e62-e65)
Synovial cell sarcoma is a malignant neoplasm that
arises not from synovial membrane, but rather from
pluripotent mesenchymal cells near or even remote
from the articular surface that is most commonly found
in the extremities.
1It comprises 8% to 10% of all soft
tissue sarcoma, and only 3% to 10% of synovial cell
sarcoma occurs in the head and neck.
2The most
com-mon sites in the head and neck are the hypopharynx and
retropharynx. It is an extremely rare tumor of the
tem-poromandibular joint area. To our knowledge, there are
only 7 cases of synovial cell sarcoma involving the
temporomandibular joint area reported in the English
literature.
2-4We present an additional case and review
the clinical features of this disease.
CASE REPORT
A 21-year-old woman appeared at the Oral and Maxillofa-cial Surgery Department of Mackay Memorial Hospital with the chief complaint of a tender mass over the right preauric-ular region for approximately 3 years. The discomfort over
the right temporomandibular region was noted for a long time, and the patient paid no attention to it until the mass became progressively larger with increased tenderness. She had visited a local otolaryngologist’s clinic several days be-fore for the ear pain, tinnitus, hearing loss, and mouth opening limitation and was referred to our department. She denied any history of trauma, facial surgery, bruxism, or clenching habits and had not had occlusal appliance therapy or any other form of nonsurgical care. Her medical history was noncontributory except for systemic lupus erythematosus. Physical examina-tion on admission revealed a tender, nonfluctuant mass in the right temporomandibular area about 3 cm in diameter. The skin overlying the swelling was intact without ulceration or induration. The range of mouth opening was limited to about 20 mm.
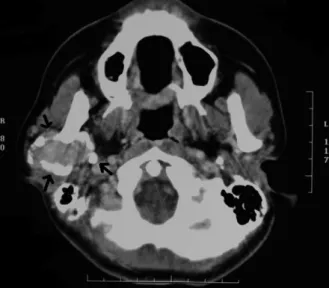
A computed tomographic scan revealed a focal soft-tissue enhancing tumor near and inferior to the right temporoman-dibular joint area posterior to the condylar neck and ramus of the right mandible. Significant ossification or calcification was seen in the periphery of this lesion. No destruction of the adjacent right mandible was seen (Fig. 1). Panoramic radiog-raphy revealed a well-circumscribed mixed radiolucent-ra-diopaque mass over the posterior border of the mandible, causing a depression in the bone (Fig. 2). There was no evidence of lymphadenopathy.
A fine needle biopsy was performed, which revealed a spindle cell tumor, favoring a benign stromal neoplasm with ossification or with reactive bony formation at the elevated periosteum.
Treatment, including surgical procedure, was discussed with the patient, and she elected to undergo operative exci-sion. The local excision was performed through the preauric-ular incision. The histological examination revealed features of synovial sarcoma with marked osseous metaplasia at the periphery. Microscopically, a biphasic tumor was identified, with predominance of hypercellular fairly uniform hyperchro-matic spindle cells with fibrosarcomalike features, which aAttending physician, Division of Oral and Maxillofacial Surgery,
Department of Dentistry, Taipei Medical University Hospital. bLecturer, Dental School, Taipei Medical University.
cChief, Department of Oral and Maxillofacial Surgery, Mackay Me-morial Hospital.
dAssociate Professor, Dental School, National Yang-Ming Univer-sity.
eAttending physician, Department of Pathology, Mackay Memorial Hospital.
fDepartment of Pathology, Taipei, Taiwan.
Received for publication Jan 21, 2007; returned for revision May 9, 2007; accepted for publication May 10, 2007.
1079-2104/$ - see front matter © 2007 Mosby, Inc. All rights reserved. doi:10.1016/j.tripleo.2007.05.009
occasionally contained epithelial-type tumor cells that formed round or slender to irregular glands (Fig. 3, A). Marked ossification forming delicate or compact bony trabeculae was seen in the hard and bony pieces (Fig. 3, B). The ossification was formed abruptly from the spindle cells without apparent osteoid formation like that seen in conventional osteosar-coma, and the term osseous metaplasia was used. The spindle cells were strongly stained for CD99 (Fig. 4, A), whereas the epithelial type of tumor cells and small numbers of spindle cells were positive for cytokeratin antigen 7 (Fig. 4, B) and epithelial membrane antigen (Fig. 4, C). The tumor cells were negative for CD34 or smooth muscle actin.
The postoperative recovery of the patient was uneventful, and all clinical signs subsided. She was discharged 10 days
after the surgical procedure. During this admission, the rou-tine tumor workup was arranged and revealed no evidence of distant metastasis. Adjuvant radiotherapy for 64.5 Gy and chemotherapy with cisplatin, epirubicin, and ifosfamide for 4 courses was completed to prevent distant metastasis and tumor recurrence. The patient has remained free of recurrent tumor 5 years after discharge. A subsequent panoramic ra-diograph revealed that the previous depression of the mandi-ble was remodeling.
DISCUSSION
Synovial cell sarcomas are found in younger patients
between the ages of 15 and 40 years, 90% are younger
than 50 years. Males appear to be slightly more
sus-ceptible than females; the average male to female ratio
is 1.2:1. Synovial sarcomas usually occur in
paraarticu-lar locations of the lower extremities.
3-6The most
com-Fig. 1. Axial computed tomography reveals a focalsoft-tissue enhancing tumor near and inferior to the right temporo-mandibular joint (arrows). Extensive ossification or calcifi-cation is seen in the periphery of this lesion.
Fig. 2. Panoramic radiograph reveals a well-circumscribed, mixed radiolucent-radiopaque mass on the posterior border of the mandible, measuring about 3⫻ 4 cm (arrows).
Fig. 3. Photomicrography. A, Plump spindle cells surround isolated glandlike epithelial component (hematoxylin-eosin, original magnification⫻100). B, Marked ossification forming delicate or compact bony trabeculae is seen in the hard and bony pieces (hematoxylin-eosin, original magnification⫻40). OOOOE
mon sites in the head and neck are the hypopharynx and
the retropharynx. It is an extremely rare tumor of the
temporomandibular joint area. There have been less
than 10 reported cases of synovial sarcoma arising in
the temporomandibular joint area.
2,3The chief complaint of most patients is a
progres-sively enlarging painless mass of several months’
du-ration. In the head and neck region, other findings
include a painful mass, dysphagia, hoarseness, and
dys-pnea, accompanied by some limited range of motion.
3-5The preoperative duration of symptoms is highly
vari-able and is most commonly 2 months to 4 years. A
history of trauma to the involved area does not appear
to be a predisposing factor.
3,4The computed tomographic imaging appearance of
synovial sarcomas of the head and neck has been
de-scribed as multilocular tumor with smooth margins and
heterogeneous enhancement after injection of contrast
medium. It may appear benign in some cases when a
well-demarcated, homogeneous lesion with
calcifica-tion is found in the head and neck.
7Findings at surgical removal yield
well-circum-scribed spherical multinodular or lobulated tumors
cov-ered by a thin fibrous membrane or pseudocapsule.
Cystic and hemorrhagic foci are present.
5The
his-topathologic findings are the same for synovial
sarco-mas arising in the head and neck and the extremities.
Synovial sarcomas classically show a biphasic cellular
pattern consisting of a stroma of fibroblast spindlelike
cells in which are scattered pale epithelial-like cells
arranged in glandular formations, nests, or cleftlike
spaces. The epithelial-like cells may be cuboidal or
columnar and form papillary projections. Synovial
sar-coma consisting of only 1 cell type, either spindle or
Fig. 4. Immunohistochemical stain. A, The spindle cells are strongly stained for CD99 immunoreactivity (original magnification ⫻100). B, The epithelial type of tumor cells and small numbers of spindle cells are positive for cytokeratin-7 immunoreactivity (original magnification⫻100). C, Epithelial type of tumor cells is also positive for epithelial membrane antigen immunoreactivity (original magnification⫻100).OOOOE
epithelial, has been described (the monophasic
vari-ant).
1,3,5,8Microcalcification has been found in some
cases.
5Pathological diagnosis is often difficult. It can
be mistaken for epithelioid sarcoma, fibrosarcoma,
ma-lignant glandular schwannoma, mama-lignant
schwan-noma, clear cell sarcoma, hemangiopericytoma, eccrine
spiradenoma, mesothelioma, pigmented villonodular
synovitis, as well as spindle cell carcinoma and
meta-static carcinoma.
4-6In addition to clinical information,
a number of immunohistochemical stains are available
to facilitate the diagnosis.
4Cytogenetic unity is also supported by the
observa-tion that a specific reciprocal chromosomal
transloca-tion t(X;18)(p11.2;q11.2) is found in each form of
synovial sarcoma.
8,9The presence of this translocation
confirms the diagnosis of difficult cases that show
un-usual histological features or are located in unun-usual
sites. Chromosome 18 contains the SYT gene, which
fuses with SSX1 or SSX2 from chromosome X. The
SYT-SSX1 fusion is associated with biphasic variants;
SYT-SSX2 fusion is associated with monophasic
vari-ants.
10Wide excision with negative margins remains the
mainstay of therapy. Most metastases are blood borne.
Regional lymph node dissection is not believed to be
necessary unless clinical lymphadenopathy or enlarged
nodes on imaging studies are present.
1,3-5With local
excision as the only mode of treatment, recurrence rates
as high as 60% to 90% have been reported in the head
and neck region.
1,2,4Postoperative radiotherapy is
ad-vocated for improved local control rates. Large doses in
the range of 65 Gy or more is considered to be necessary,
but radiotherapy alone is not recommended.
3-5,11,12Che-motherapy has been used in an attempt to prevent the
occurrence of distant metastases in advanced disease or
in patients with residual gross tumor.
3,4Metastases are
developing in approximately 50% of patients and are
most often found in the lung. Other favored sites of
tumor metastasis are lymph nodes and bone marrow.
4Although not normally a feature of sarcomas,
metasta-sis of regional lymph nodes occurs in 12.5% of cases of
the head and neck and 23% of cases in the extremities.
8The prognosis of head and neck synovial sarcoma is
poor; 5-year survival rates range from 36% to 76% and
10-year survival rates range from 11% to 63%.
3,12The
prognosis of head and neck synovial sarcoma is poor;
5-year survival rates range from 36% to 76% and
10-year survival rates range from 11% to 63%
3,12;
overall survival rates for synovial sarcoma are 55%
survival at 5 years and 38% survival at 10 years.
1Favorable prognostic factors appear to be early
diag-nosis and the performance of wide surgical excision.
The size of the primary tumor and the depth of invasion
may be the most important prognostic indicators.
Pa-tients whose primary tumor is larger than 4 cm in
diameter have a poorer outcome. Calcifying-type
syno-vial sarcoma has been reported to have a better
prog-nosis.
1,5The authors thank Dr. M.J. Buttrey for her kind review and instructive criticism of this paper.
REFERENCES
1. Carrillo R, Rodriguez-Peralto JL, Batsakis JG. Synovial sarco-mas of the head and neck. Ann Otol Rhinol Laryngol 1992; 101:367-70.
2. Mostafapour SP, Futran ND. Tumors and tumorous masses pre-senting as temporomandibular joint syndrome. Otolaryngol Head Neck Surg 2000;123:459-64.
3. White RD, Makar J Jr, Steckler RM. Synovial sarcoma of the temporomandibular joint. J Oral Maxillofac Surg 1992;50:1227-30. 4. Stadelmann WK, Cruse CW, Messina J. Synovial cell sarcoma of
the temporomandibular joint. Ann Plast Surg 1995;35:664-8. 5. Bukachevsky RP, Pincus RL, Shechtman FG, Sarti E, Chodosh
P. Synovial sarcoma of the head and neck. Head Neck 1992; 14:44-8.
6. Shmookler BM, Enzinger FM, Brannon RB. Orafacial synovial sarcoma: a clinicopathologic study of 11 new cases and review of the literature. Cancer 1982;50:269-76.
7. Rangheard AS, Vanel D, Viala J, Schwaab G, Casiraghi O, Sigal R. Synovial sarcomas of the head and neck: CT and MR imaging findings of eight patients. AJNR Am J Neuroradiol 2001;22: 851-7.
8. Cihak RA, Lydiatt WM, Lydiatt DD, Bridge JA. Synovial sar-coma of the head and neck: chromosomal translation (X;18) as a diagnostic aid. Head Neck 1997;19:549-53.
9. Bridge JA, Bridge RS, Borek DA, Shaffer B, Norris CW. Trans-location t(X;18) in orofacial synovial sarcoma. Cancer 1988;62: 935-7.
10. Koyama S, Morimitsu Y, Morokuma F, Hashimoto H. Primary synovial sarcoma of the kidney: report of a case confirmed by molecular detection of the SYT-SSX2 fusion transcripts. Pathol Int 2001;51:385-91.
11. Amble FR, Olsen KD, Nascimento AG, Foote RL. Head and neck synovial cell sarcoma. Otolaryngol Head Neck Surg 1992; 107:631-7.
12. Mullen JR, Zagars GK. Synovial sarcoma outcome following conservation surgery and radiotherapy. Radiother Oncol 1994; 33:23-30.
Reprint requests:
Chung-Ji. Liu, DDS, MSD
Department of Oral and Maxillofacial Surgery Mackay Memorial Hospital
No 92, Chung-Shan North Road, Sec. 2 Taipei, Taiwan, 10449, Republic of China [email protected]
OOOOE