A pilot study in acute subarachnoid hemorrhagic patients after aneurysm clipping
with complementary therapies of Chinese Medicine
Han-Chung Lee1,2,3, Ching-Liang Hsieh,4,5,6*, Chun-Chung Chen1, Der-Yang Cho1,
Pao-Hsuan Lin7
1
Department of Neurosurgery, China Medical University Hospital, Taichung, Taiwan.
2
Graduate Institute of Integrated Medicine, China Medical University, Taichung, Taiwan
3
School of Medicine, China Medical University, Taichung, Taiwan. 4
Graduate Institute of Acupuncture Science, China Medical University, Taichung, Taiwan. 5
Acupuncture Research Center, China Medical University, Taichung, Taiwan. 6
Department of Chinese Medicine, China Medical University Hospital, Taichung, Taiwan 7
Biostatistical Center, China Medical University, Taichung, Taiwan.
Running title: Complementary therapies in acute subarachnoid hemorrhage patients
*Corresponding author: Ching-Liang Hsieh, M.D.,Ph.D., Graduate Institute of Acupuncture
Science, China Medical University. 91 Hsueh-Shih Road, Taichung, 40402, Taiwan
TEL: 886-4-22053366 (ext. 3600) Fax: 886-4-22035191
Summary
Objectives: Acute subarachnoid haemorrhage still has high mortality and morbidity despite
the use of modern standard treatment. In Taiwan, complementary therapies of Chinese
medicine are usually used to treat stroke patients. The aim of this study was to investigate the
effect of complementary therapies of Chinese medicine on patients with acute subarachnoid
haemorrhage after aneurysm clipping.
Design: This study was designed as a pilot study. A total of 32 patients with acute
subarachnoid haemorrhage were randomly assigned to either an experimental group (EG) in
which the patients were given complementary therapies of Chinese medicine and modern
standard treatment or a control group (CG) in which patients were given modern standard
treatment only.
Main outcome measures: Glasgow Outcome Scale scores, which were assessed by an
evaluator who was blinded to the groups, 3 months after admission, and total admission days
including intensive care unit stay days.
Results: The average Glasgow Outcome Scale score 3 months after admission was 3.7±1.4 in
the EG which was higher than the score of 3.0±1.7 in the control group (p < 0.05). Average
total admission days were 34.0±24.3 for the CG which a longer stay than the 25.2±24.5 days
for the EG (p < 0.05).
subarachnoid haemorrhage are of value because they can increase Glasgow Outcome Scale
scores 3 months after admission and also because they can reduce total admission days.
Keywords: Complementary therapies; Chinese Medicine; Acute subarachnoid
Introduction
The use of complementary therapies of Chinese medicine (CM) including Chinese herbs and
acupuncture to treat patients with chronic or subacute stage of stroke is popular in Taiwan.
Salviae miltiorrhizae may increase recovery rate of patients with acute subarachnoid
haemorrhage (SAH) had been reported.1 SAH is an acute emergency. About 85% of cases
result from the rupture of an aneurysm.2 The incidence of SAH is about 6 cases per 100,000
patient years.2 The overall case mortality rate of SAH is 42% during the first 28 days.3
Although ultra-early aneurysm clipping (within 3 days after onset) is used for the treatment of
SAH, higher mortality and morbidity rates are still noted compared with the other cerebral
diseases due to occurrence of vasospasm following SAH, which may cause cerebral
ischaemia.4
Aneurysm rupture with SAH may cause inflammation resulting in fatal vasospasm and
central pyrexia.5 Patients with SAH who have complicated fever may have a prolonged stay
in the ICU and a poorer outcome.6,7 Mechanical compression of the brainstem and
hypothalamus may induce the production and release of pro-inflammatory cytokines,
including interleukin-1β (IL-1β), IL-6, tumor necrosis factor-α (TNF-α), and S100B as a
calcium-binding protein of astrocytes, causing central pyrexia.8-10 The pro-inflammatory
cytokine levels increase in brain tissues, cerebrospinal fluid (CSF), and blood in patients with
Traditional Chinese medicine (TCM) such as acupuncture, Chinese herbs, and CM
formula has been used to treat stroke for centuries in China. Our previous studies have
shown that Gastrodia elata plays a neuroprotective role in kainic acid-treated rats26 and may
reduce IL-1β and TNF-α levels in brain tissue of kainic acid-treated rats.27 Paeonol, a major
component of Moutan Cortex of Paeonia suffruticosa Andrews and the root of Paeonia
lactiflora Pall, has anti-inflammation and anti-oxidant effects in transient
ischaemia-reperfusion injury rats.28 Therefore, we designed a pilot study to investigate the
effect of complementary therapies of CM on patients with acute SAH. The therapeutic effect
was evaluated by Glascow Outcome Scale (GOS) score and total admission days including
ICU stay days. In addition, daily body temperature (BT) and intracranial pressure
(ICP) were recorded and the levels of TNF-α, IL-1β, IL-6, and S100B in cerebrospinal
fluid (CSF) were also measured.
Materials and Methods
Subjects
A total of 53 patients with acute SAH were treated at China Medical University Hospital,
Taichung, Taiwan from January 2007 and December 2007. Thirty-two patients who
underwent craniotomy were included in the study. The inclusion criteria were the following:
tomography angiography (CTA) scanning within 6 hours after the episode; 2) the
neurological deficit was between grade 2 and 4 of Hunt and Hess (H&H) grade; 3) serum
blood urea nitrogen (BUN) was ≦ 25 mg/ml, creatinine ≦ 1.8 mg/dl, and the creatinine
clearance was ≧ 50 ml/min; 4) serum SGOT, SGPT, and alkaline phosphatase were less than
three times the upper normal limit and the total bilirubin was less than 3 mg/dl; 5) serum
haemoglobin was ≧8 g/dl, platelet counts ≧ 100×103/µl,white cell counts ≧ 2×103/µl,and
the absolute neutrophils ≧1000/µl. The exclusion criteria were as follows: 1) patients with
pregnancy; 2) age < 12 years or > 70 years; 3) H&H grade of 1 and 5; 4) patients or their
families refuse participation in trial.
Study design
All the experimental procedures were according to the ethical principles dictated in the
Declaration of Helsinki. The protocol of the trial was approved by the institutional review
board of the China Medical University Hospital, Taichung City, Taiwan (DMR95:IRB80), and informed consent regarding the experimental procedures and purpose was obtained prior
to the trial.
After undergoing an aneurysm clipping operation the patients were randomized by an on duty
doctor who takes a lot of experimental group (EG) or control group (CG) from a dark box to
or a CG which received modern standard treatment only. Each group had 16 subjects.
Because this study was a pilot study, there was no basis for calculating its power and sample
size (Figure 1).
Standard treatment
Standard treatment of acute SAH according to the guidelines of the Stroke Council, American
Heart Association29 is based on clipping of the aneurysm as early as possible and the
prevention of secondary insults to the brain. External ventricular drainage was performed
routinely during aneurysm clipping. It was used not only to monitor the postoperative ICP but
also for drainage of the intraventricular haemorrhage and was maintained for not more than 7
days to avoid related infection. All patients were intubated and placed on volume-controlled
ventilation under sedation to maintain partial pressure of oxygen in arterial blood (PaO2) of
at least 100 mm Hg and arterial carbon dioxide pressure or tension (PaCO2) of approximately
35-40 mm Hg after the operation. The endotracheal tube was not removed until the
consciousness of the patient was clear and the ICP was stable. Hypertension, hyperperfusion,
haemodilution and calcium-channel blocking agent (nimodipine) were started immediately
after arrival in the ICU after surgery. ICP was treated by elevating the patient’s head by
raising one end of the bed, sedation, paralysis, and mannitol. Nutritional support was started
solutions. Oral acetaminophen was given regularly to prevent further pyrexia.
The complementary therapies of CM
The complementary therapies of CM were given every day continuously for 2 weeks after the
patients started to take food on the second day after the surgical operation. Patients were
mainly given the following four essential Chinese herbs: 1) Astragalus menbranaceus (Fisch)
Bunge (Radix Astragalli, 12 g/day; Shaanxi, China); 2) Gastrodia elata Blume (Rhizoma
Gastrodiae, 12 g/day; Sichuan, China); 3) Acorus gramineus Soland (Rhizoma Acori
Graminei, 7.5 g/day; Sichuan, China), and 4) Pheretima aspergillum (E. Perrier)
(Lumbricus, 12 g./day; Thailand). In addition, Chinese herbs such as Paeonia
suffruticosa Andr (Cortex Moutan, 12 g/day; Zhejiang, China), Lonicera japonica
Thunb (Flos Lonicerae, 19 g/day; Anhui, China), raw Rehmannia glutinosa Libosch
(Radix Rehmaniae, 12 g/kg; Henan, China), and Scutellaria baicalensis Georgi (Radix
Scutellariae, 12 g/kg; Hebei, China) were given when patients had fever or a heat
phenomenon in TCM such as quickened radial pulse (more than 85 beats/min),
tongue color was fresh red, etc. Rheum palmatum Linn (Radix et Rhizoma Rhei, 4
g/kg; Sichuan, China) and Citrus aurantium Linn (Fructus Aurantii Immaturus, 12
g/kg; Sichuan, China) were added when patients had no defecation for more than 3
shape even histological section, and decocted by the Chinese herb specialist in the
China Medical University Hospital. These herbs were mixed with 600 cc of water,
and then decocted to 300 cc. Patients were given 100 cc of the solution of herbs
three times a day.
Clinical characteristics and basic data recording
The age, gender, GCS scores, H&H grade and SAH grade of the patients were recorded on
the day of admission.
Main outcome measure
GOS scores were assessed and recorded on the personal medical record in the outpatient door
3 months (±7 days) after admission by an evaluator who was blinded to the group. The GOS
scores were divided into five grades from 1 to 5: score 1, death; score 2, vegetative state;
score 3, severe disability; score 4, moderate disability; score 5, mild or no disability.30 In
addition, total admission days were used as an outcome measure including ICU stay days.
Secondary outcome measure
The Glascow Coma Scale (GCS) scores and H&H grade were recorded on the day of
temperature (BT), taken with an ear thermometer, and daily ICP were recorded. In addition,
cytokine levels including IL-1β, IL-6, TNF-α, and S100 were measured in the cerebrospinal
fluid on the 1st and 5th day after the operation.
Daily BT and ICP recordings
The BT and ICP were monitored every 2 hours continuously for 5 days after surgery. The
average BT and ICP were calculated. The variation in BT was calculated (daily highest BT −
average daily BT)2 and the variation in ICP was calculated (daily highest ICP − average daily
ICP)2.
The measurement of cytokine levels
Three-milliliter samples of CSF were collected on the 1st and 5th days from the external
ventricle drainage of the lateral ventricle after surgery. The samples were centrifuged for 20
minutes at 2000 rpm, and the supernatant was immediately stored at −80oC until analysis.
The levels of IL-1β, IL-6, and TNF-α were determined by using a commercial enzyme-linked
immunosorbent assay (ELISA) kit (Bender MedSystems, Inc., USA) and an ELISA reader
(Dynex MRX, Virginia, USA). The sensitivity of the assay was typically 0.124 pg/ml for
IL-1β, 0.094 pg/ml for IL-6, and 0.081 pg/ml for TNF-α. The S100B level in CSF was
known concentrations. The lower limit of detection of the ELISA is 0.01 ng/ml. No
cross-reactivity or interference with other related interleukins was observed. The data were
represented in pg/ml and all assays were performed in duplicate.
Statistical Analysis
Student’s t test for unpaired test and, whenever necessary, the Wilcoxon signed rank and
repeated ANOVA were used to compare measurements. Data are expressed as means ±
standard deviations. Spearman correlation coefficients were used to compare the relationships
between the variations of daily BT and ICP and the concentrations of IL-1β, IL-6, TNF-α,
and S100B. Statistical significance was set at p < 0.05.
Results
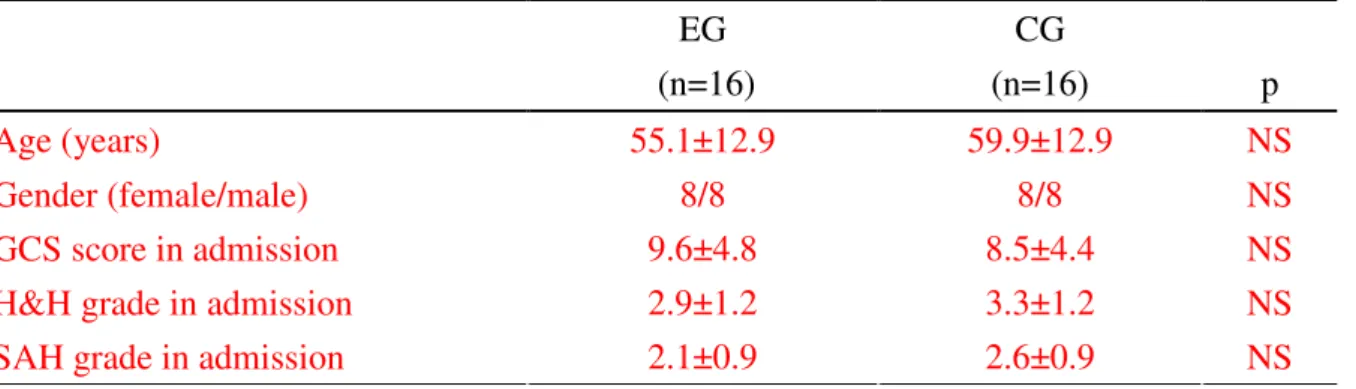
The analysis of basic data
The age, gender, GCS score on admission, H&H grade score on admission, and SAH grade
score on admission were not significantly different between the EG and CG (all p > 0.05;
Table 1).
Main outcome measures
GOS score was 3.7±1.4 in the EG which was significantly higher than the score of 3.0±1.7 in
the CG (p < 0.05).A total of four patient’s death due to intractable increased ICP in ICU, and
the complication included pulmonary infection, urinary tract infection, and gastrointestinal
tract hemorrhage etc., but no patient withdrew or adverse events were noted in the trial. The
mortality rate during the stay in the ICU was 6.25% (1/16) in the EG which was similar to the
rate of 18.8% (3/16) in the CG (p > 0.05). The mean number of ICU stay days was 16.8±8.1
for the CG which was significantly more than the 11.1±4.1 days for the EG (p<0.05). The
total number of hospital stay days was 34.0±24.3 for the CG which was significantly more
than the 25.2±24.5 days for the EG (p < 0.05).
Secondary outcome
The average GCS score at discharge was 12.3±2.5 for the EG which was significantly higher
than the score of 10.3±2.9 for the CG (p < 0.05).
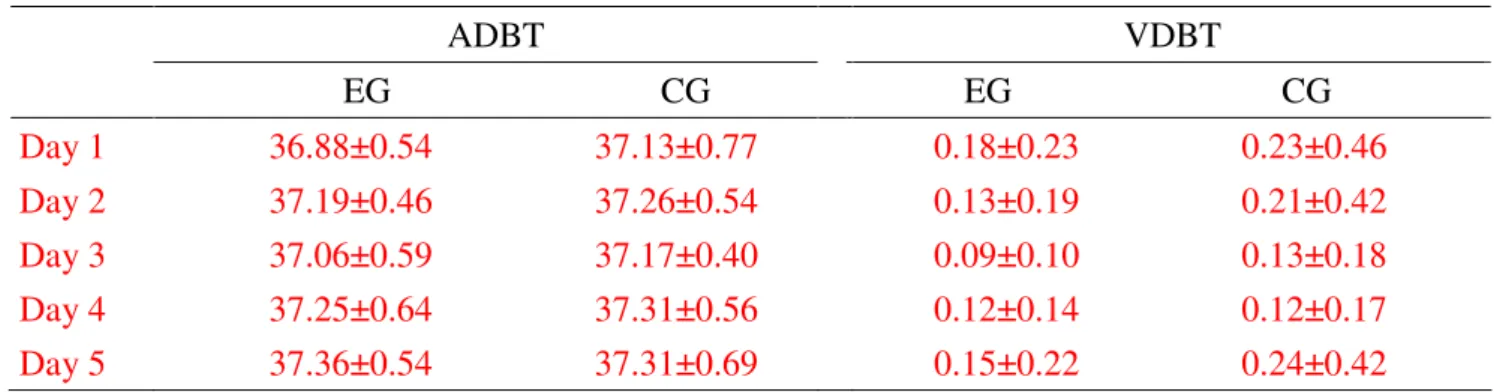
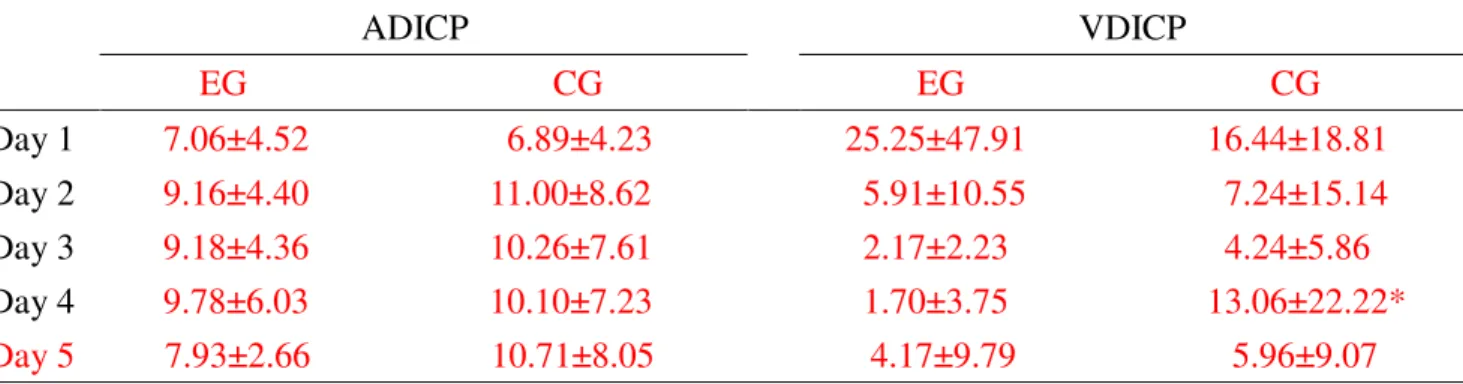
Effect of complementary therapies of CM on BT and ICP
The average daily BT and variation of daily BT, and the average daily ICP and variation of
daily ICP (VDICP) from the 1st to 5th days after surgery were not significantly different
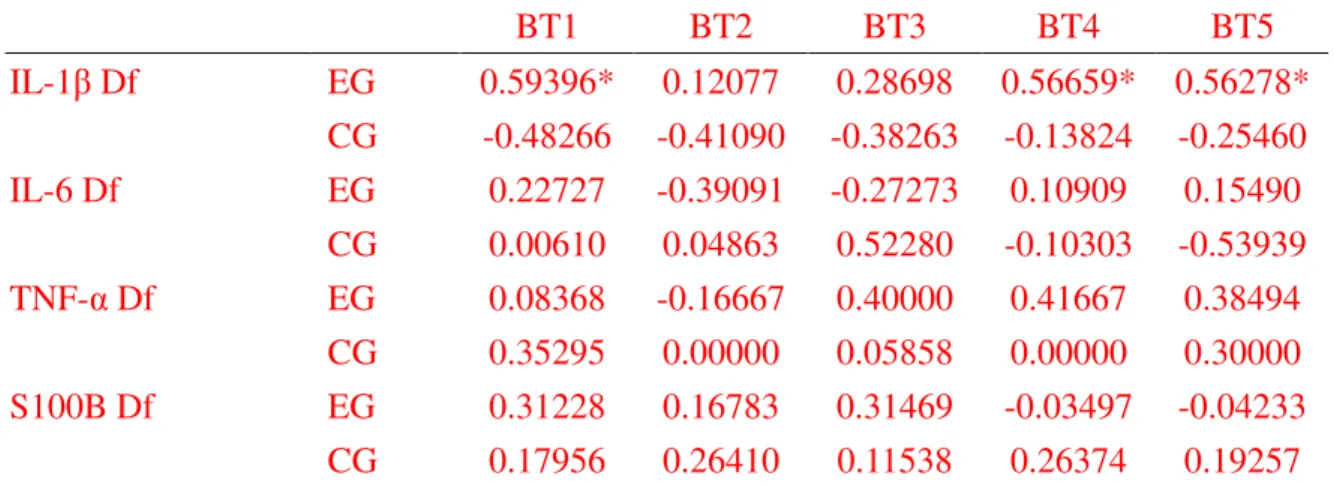
Effect of complementary therapies of CM on IL-1β, IL-6, TNF-α, and S100B in CSF
The difference between IL-1β levels on the 1st day and IL-1β levels on the 5th day was
positively correlated with the average daily BT on the 1st, 4th, and 5th days after surgery in the
EG (all p < 0.05; Table 4), whereas there was no correlation with average daily BT on the 2nd
and 3rd days after surgery in the EG (both p > 0.05; Table 4). The difference between IL-1β
levels on the 1st day and IL-1β levels on the 5th day had no correlation with the average BT on
the 1st , 2nd, 3rd ,4th, and 5th days after surgery in the CG (all p > 0.05; Table 4).
The difference between IL-6 levels on the 1st day and IL-6 levels on the 5th day had no
correlation with the average daily BT on the 1st, 2nd, 3rd ,4th, and 5th days after surgery in the
EG and CG (all p > 0.05; Table 4),
The difference between TNF-α levels on the 1st day and TNF-α levels on the 5th day had no
correlation with the average BT on the 1st , 2nd, 3rd ,4th, and 5th days after surgery in the EG
and CG (all p > 0.05; Table 4),
The difference between S100B levels on the 1st day and S100B levels on the 5th day had no
correlation with the average BT on the 1st, 2nd, 3rd ,4th, and 5th days after surgery in the EG
and CG (all p > 0.05; Table 4).
Discussion
increase the GOS score 3 months after admission, and reduce the total number of admission
days, including both ICU stay days, which suggests that complementary therapies of CM
provide an advantage in outcome for such patients. Critically ill patients with SAH
commonly have fever, a factor known to worsen neurologic injury due to vasospasm.14 In
these patients, fever and vasospasm may be both associated with the production and release
of pro-inflammatory cytokines, including IL-1β, IL-6, and TNF-α.18-22 Increased protein
levels of pro-inflammatory cytokines have been reported in brain tissues, cerebrospinal fluid,
and blood of patients with SAH, traumatic brain injury, stroke, and other neurological
conditions.23,24
Interleukin-1β is thought to play an important role in mediating inflammation and neuronal
damage after traumatic brain injury, spontaneous SAH, and stroke by enhancing the
inflammatory reactions via the release of other inflammatory mediators such as
prostaglandins, collagenase, and phospholipase A2.12Additionally, IL-1β has been implicated
in apoptotic cell death,13,14 leukocyte-endothelium adhesion,15 blood-brain barrier
disruption,16 edema formation,16,17 astrogliosis, and neovascularization.18 Experimentally,
intracerebroventricular administration of IL-1β is associated with marked stimulation of
circulating IL-6 and TNF-α levels.19,21 Inhibition of IL-1β has been shown to reduce the
incidence of central pyrexia, vessel spasm, and early edema formation.22,23 Our studies
change the average daily BT or variation of daily BT, or the average daily ICP or variation of
daily ICP except day 4 due to two patients death with increased ICP. The difference in IL-1β
concentration between the 1st day and 5th day after surgery showed a positive correlation with
daily BT on the 1st, 4th, and 5th day after surgery in the complementary therapies of CM group,
whereas there were no similar results in the CG which did not receive complementary
therapies of CM. These results suggest that complementary therapies of CM may decrease
IL-1β concentration in CSF which reduces the inflammation and fever caused by SAH.
Unfortunately, this tendency was not observed for IL-6, TNF-α, and S100B. More frequent
checking of the concentration of cytokines and a longer observation period may be helpful for
demonstrating a significant difference.
That Gastrodia elata and its component vanillyl alcohol may inhibit the
production and scavengering of oxygen free radicals, and inhibit microglia activation in
kainic acid-induced epileptic rats was shown in our previous studies.26,31,32 Aastragaloside IV
is a component of Astragalus membranaceus that can reduce the cerebral infarction area
induced by middle cerebral artery occlusion in rats, and this effect of Aastragaloside IV
results from its anti-oxidative properties.33 Acorus graminueus has the action of
resolving phlegm to open orifices in TCM, and can enhance learning and
memory.3 4 Lumbrokinase is a component of Pheretima aspergillum (earthworm), and can
inhibiting intracellular adhesion molecular-1 (ICAM-1) expression to protect against cerebral
ischaemia.35 The paeonol component of Paeonia suffruticosa may reduce the cerebral
infarction area and neurological deficit, and also has anti-oxidative action in cerebral
ischaemia-reperfusion injured rat.28The component of Lonicera japonica may inhibit
microglia activation to protect dopamingeric neurons from liopopolysaccaride (LPS)-induced
injury,36 and also may inhibit nuclear factor-κB (NF-κB) and activator protein-1 (AP-1) to
suppress inflammatory reaction in mouse alveolar macrophage.37 Catapol is a component of
Rehmannia glutinosa that may reduce lipid peroxidation and also may increase glutathione
and superoxide dismutase activities in MPP+-induced oxidative stress in mesencephalic
neurons.3 8 Catapol also can reduce the formation of intracellular reactive oxygen
species in astrocytes with H2O2-induced oxidative stress.39 The baicalin
component of Scutellaria baicalensis can mediate via binding to chemokines to
produce anti-inflammatory activity in human peripheral blood leucocytes,4 0 and
baicalein also can maintain brain mitochondrial homeostasis and function in rats
with chronic cerebral hypoperfusion-induced oxidative damage.41 Rheum
palmatum is a laxative, and its emodin component can through the inhibition of
AP-1 and NF-κB suppress matrix metalloproteinase in human cancer cells.42
Citrus aurantium is qi-regulating and is medicinal for digesting, and it has
anti-oxidation and ant-inflammation including the inhibiting generation and
scavenging of oxygen free radicals, and the inhibition of IL-1β, suggesting that these
action may improve functional recovery of SAH patients.
The present study was a pilot study and therefore there were some limitations as follows: 1)
treatment with the complementary therapies of CM had to be agreed by the patients or their
families, thus randomly assigning the patients to the CG or EG by a completely blind method
was difficult; 2) the sample size was small; 3) there was no fixed CM formula. Future
research using a randomized double-blind study design, an increased number of patients, and
a fixed CM formula is needed.
In conclusion, TCM in patients with acute SAH is a worthy extension to treatment because
they can increase GOS scores at 3 months after admission and also reduce total admission
days including ICU stay days.
Acknowledgment
This study was supported by grant DMR 95-044 from the China Medical University Hospital,
Taichung, Taiwan, and was supported in part by Taiwan Department of Health Clinical Trial
Reference
1. Gui JP. Observation of effects of salvia injection in the treatment of acute subarachnoid
haemorrhage. Modern Journal of Integrated Traditional Chinese and Western Medicine
2004; 13(6): 768.
2. van Gijn J, Rinkel GJ. Subarachnoid haemorrhage: diagnosis, causes and management.
Brain 2001; 124:249-278.
3. Ingall T, Asplund K, Mähönen M, Bonita R. A multinational comparison of subarachnoid
hemorrhage epidemiology in the WHO MONICA stroke study. Stroke 2000;
31:1054-1061.
4. Kassell NF, Sasaki T, Colohan ART, Nazar G. Cerebral vasospasm following aneurysmal
subarachnoid hemorrhage. Stroke 1985; 16(4):562-572.
5. Oliveira-Filho J, Ezzeddine MA, Segal AZ, et al. Fever in subarachnoid hemorrhage:
relationship to vasospasm and outcome. Neurology 2001; 56:1299-1304.
6. Commichau C, Scarmeas N, Mayer SA. Risk factors for fever in the neurologic intensive
care unit. Neurology 2003; 60: 837-841.
7. Gaetani P, Pasqualin A, Baena RR, Borasio E, Marzatico F. Oxidative stress in the human
brain after subarachnoid hemorrhage. J Neurosurg 1998; 89:748-754.
8. Marik PE. Fever in the ICU. Chest 2000; 117(3):855-869.
10. Kassell NF, Torner JC, Haley EC, et al. The international cooperative study on the timing
of aneurysm surgery. Part 1: overall management results. J Neurosurgery 1990; 73:18-36.
11. De Simoni MG, Sironi M, De Luigi A, Manfridi A, Mantovani A, Ghezzi P.
Intracerebroventricular injection of interleukin 1 induces high circulating levels of
interleukin 6. J Exp Med 1990; 171:1773-1778.
12. Fan L, Young PR, Barone FC, Feuerstein GZ, Smith DH, McIntosh TK. Experimental
brain injury induces expression of interleukin-1β mRNA in the rat brain. Molecular
Brain Research 1995; 30:125-130.
13. Holmin S, Schalling M, Höjeberg B, Nordqvist ACS, Skeftruna AK, Mathiesen T.
Delayed cytokine expression in rat brain following experimental contusion. J Neurosurg
1997; 86:493-504.
14. Morganti-Kossman MC, Lenzlinger PM, Hans V, et al. Production of cytokines
following brain injury: beneficial and deleterious for the damaged tissue. Molecular
Psychiatry 1997; 2:133-136.
15. Shohami E, Ginis I, Hallenbeck JM. Dual role of tumor necrosis factor alpha in brain
injury. Cytokine & Growth Factor Reviews 1999; 10:119-130.
16. Shohami E, Novikov M, Bass R, Yamin A, Gallily R: Closed head injury triggers early
production of TNFα and IL-6 by brain tissue. Journal of Cerebral Blood Flow and
17. Bell MJ, Kochanek PM, Doughty LA, et al. Interleukin-6 and interleukin-10 in
cerebrospinal fluid after severe traumatic brain injury in children. Journal of
Neurotrauma 1997; 14(7):451-457.
18. Boutin H, LeFeuvre RA, Horai R, Asano M, Iwakura Y, Rothwell NJ. Role of IL-1α and
IL-1β in ischemic brain damage. The Journal of Neuroscience 2001; 21(15):5528-5534.
19. Friedlander RM, Gagliardini V, Rotello RJ, Yuan J. Functional role of interleukin 1β
(IL-1β) in IL-1β-converting enzyme-mediated apoptosis. J Exp Med 1996; 184: 717-724.
20. Holmin S, Mathiesen T. Intracerebral administration of interleukin-1β and induction of
inflammation, apoptosis, and vasogenic edema. J Neurosurg 2000; 92:108-120.
21. Bevilacqua MP, Pober JS, Wheeler ME, Cotran RS, Gimbrone MA. Interleukin 1 acts on
cultured human vascular endothelium to increase the adhesion of polymorphonuclear
leukocytes, monocytes, and related leukocyte cell lines. J Clin Invest 1985;
76:2003-2011.
22. Quagliarello VJ, Wispelwey B, Long WJ, Scheld WM. Recombinant human
interleukin-1 induces meningitis and blood-brain barrier injury in the rat:
Characterization and comparison with tumor necrosis factor. J Clin Invest 1991;
87:1360-1366.
23. Yamasaki Y, Matsuura N, Shozuhara H, Onodera H, Itoyama Y, Kogure K. Interleukin-1
24. Giulian D, Woodward J, Young DG, Krebs JF, Lachman LB. Interleukin-1 injected into
mammalian brain stimulates astrogliosis and neovascularization. The Journal of
Neuroscience 1988; 8(7):2485-2490.
25. De Simoni MG, De Luigi A, Gemma L, Sironi M, Manfridi A, Ghezzi P. Modulation of
systemic interleukin-6 induction by central interleukin-1. Am J Physiol 1993;
265:R739-R742.
26. Hsieh CL, Chen CL, Tang NY,et al. Gastrodia elata BL mediates the suppression of
nNOS and microglia activation to protect against neuronal damage in kainic acid-treated
rats. The American Journal of Chinese Medicine 2005; 33(4):599-611.
27. Huang YH, Chang YM, Shen JJ, Hsieh CL. Relationship between the anticonvulsion
effect of Gastrodia elata and interleukin-1β, tumor necrosis factor-α and nitric oxide.
Mid-Taiwan Journal of Medicine 2005; 10 Supplement:S1-S8.
28. Hsieh CL, Cheng CY, Tsai TH, et al. Paeonol reduced cerebral infarction involving the
superoxide anion and microglia activation in ischemia-reperfusion injured rats. Journal
of Ethnopharmacology 2006; 106:208-215.
29. Bederson, JB, Connolly, ES Jr, Batjer, HH, et al. Guidelines for the management of
aneurysmal subarachnoid hemorrhage: a statement for healthcare professionals from a
special writing group of the Stroke Council, American Heart Association. Stroke 2009;
30. Jennett B, Bond M. Assessment of outcome after severe brain damage. Lancet 1975;
1(7905): 480-4.
31. Hsieh CL, Chang CH, Chiang SY, et al. Anticonvulsive and free radical scavenging
activities of vanillyl alcohol in ferric chloride-induced epileptic seizures in
Sprague-Dawley rats. Life Sciences 2000; 67:1185-1195.
32. Hsieh CL, Chiang SY, Cheng KS, et al. Anticonvulsive and free radical scavenging
activities of Gastrodia elata BL. in kainic acid-treated rats. The American Journal of
Chinese Medicine 2001; 29(2):331-341.
33. Luo Y, Qin Z, Hong Z, et al. Astragaloside IV protects against ischemic brain injury in a
murine model of transient focal ischemia. Neuroscience Letters 2004; 363:218-223.
34. Wu X, Huang Y. Review of experimental research on Calamus chemical components
functional mechanism on CNS. Journal of ZheJiang University of Traditional Chinese
Medicine 2007; 31(6):789-791.
35. Ji H, Wang L, Bi H, et al. Mechanisms of lumbrokinase in protection of cerebral
ischemia. European Journal of Pharmacology 2008; 590:281-289.
36. Chen HQ, Jin ZY, Wang XJ, Xu XM, Deng L, Zhao JW. Luteolin protects dopaminergic
neurons from inflammation-induced injury through inhibition of microglial activation.
37. Chen CY, Peng WH, Tsai KD, Hsu SL. Luteolin suppresses inflammation-associated
gene expression by blocking NF-κB and AP-1 activation pathway in mouse alveolar
macrophage. Life Sciences 2007; 81:1602-1614.
38. Tian YY, Jiang B, An LJ, Bao YM. Neuroprotective effect of catalpol against
MPP+-induced oxidative stress in mesencephalic neurons. European Journal of
Pharmacology 2007; 568:142-148.
39. Bi J, Jiang B, Liu JH, Lei C, Zhang XL, An LJ. Protective effects of catalpol against
H2O2-induced oxidative stress in astrocytes primary cultures. Neuroscience Letters 2008;
442:224-227.
40. Li BQ, Fu T, Gong WH, et al. The flavonoid baicalin exhibits anti-inflammatory activity
by binding to chemokines. Immunopharmacology 2000; 49:295-306.
41. He XL, Wang YH, Gao M, Li XX, Zhang TT, Du GH. Baicalein protects rat brain
mitochondria against chronic cerebral hypoperfusion-induced oxidative damage. Brain
Research 2009; 1249:212-221.
42. Huang Q, Shen HM, Ong CN. Inhibitory effect of emodin on tumor invasion through
suppression of activator protein-1 and nuclear factor-κB. Biochemical Pharmacology
2004; 68:361-371.
43. Su MS, Shyu YT, Chien PJ. Antioxidant activities of citrus herbal product extracts. Food
Legend
Table 1. Clinical characteristics and basic data in acute subarachnoid hemorrhagic patients EG CG (n=16) (n=16) p Age (years) NS Gender (female/male) NS GCS score in admission NS
H&H grade in admission NS
SAH grade in admission
55.1±12.9 8/8 9.6±4.8 2.9±1.2 2.1±0.9 59.9±12.9 8/8 8.5±4.4 3.3±1.2 2.6±0.9 NS
Data represent as Mean ± SD (standard deviation);n: patients number;EG:experimental group, acute subarachnoid hemorrhagic patient with complementary therapies of Chinese Medicine; CG, control group, acute subarachnoid hemorrhagic patient without
complementary therapies of Chinese medicine; GCS: Glasgow coma scale;H&H: Hunt & Hess;SAH: subarachnoid hemorrhage; NS: not significant.
Table 2. The averaged and variation daily body temperature changes in acute subarachnoid hemorrhagic patients ADBT VDBT EG CG EG CG Day 1 36.88±0.54 37.13±0.77 0.18±0.23 0.23±0.46 Day 2 37.19±0.46 37.26±0.54 0.13±0.19 0.21±0.42 Day 3 37.06±0.59 37.17±0.40 0.09±0.10 0.13±0.18 Day 4 37.25±0.64 37.31±0.56 0.12±0.14 0.12±0.17 Day 5 37.36±0.54 37.31±0.69 0.15±0.22 0.24±0.42
Data represent as Mean ± SD (standard deviation); EG:experimental group, acute subarachnoid hemorrhagic patient with complementary therapies of Chinese medicine; CG, control group, acute subarachnoid hemorrhagic patient without complementary therapies of Chinese medicine; ADBT: averaged daily body temperature; VDBT: variation of daily body temperature; Day 1: 1st day after surgical operation; Day 2: 2nd day after surgical operation; Day 3: 3rd day after surgical operation; Day 4: 4th day after surgical operation; Day 5: 5th day after surgical operation; Wilcoxon signed rank test.
Table 3. The averaged and variation daily intracranial pressure in acute subarachnoid hemorrhagic patients ADICP VDICP EG CG EG CG Day 1 7.06±4.52 6.89±4.23 25.25±47.91 16.44±18.81 Day 2 9.16±4.40 11.00±8.62 5.91±10.55 7.24±15.14 Day 3 9.18±4.36 10.26±7.61 2.17±2.23 4.24±5.86 Day 4 9.78±6.03 10.10±7.23 1.70±3.75 13.06±22.22* Day 5 7.93±2.66 10.71±8.05 4.17±9.79 5.96±9.07 Data represent as Mean ± SD (standard deviation); EG:experimental group, acute subarachnoid
hemorrhagic patient with complementary therapies of Chinese medicine; CG, control group, acute subarachnoid hemorrhagic patient without complementary therapies of Chinese medicine; ADICP: averaged daily intracranial pressure; VDICP: variation of daily intracranial pressure; Day 1: 1st day after surgical operation; Day 2: 2nd day after surgical operation; Day 3: 3rd day after surgical
operation; Day 4: 4th day after surgical operation; Day 5: 5th day after surgical operation; Wilcoxon signed rank test;*p < 0.05 compared with EG..
Table 4: The relationship of IL-1β between averaged daily body temperatures in acute subarachnoid hemorrhage patients
BT1 BT2 BT3 BT4 BT5 IL-1β Df EG 0.59396* 0.12077 0.28698 0.56659* 0.56278* CG -0.48266 -0.41090 -0.38263 -0.13824 -0.25460 IL-6 Df EG 0.22727 -0.39091 -0.27273 0.10909 0.15490 CG 0.00610 0.04863 0.52280 -0.10303 -0.53939 TNF-α Df EG 0.08368 -0.16667 0.40000 0.41667 0.38494 CG 0.35295 0.00000 0.05858 0.00000 0.30000 S100B Df EG 0.31228 0.16783 0.31469 -0.03497 -0.04233 CG 0.17956 0.26410 0.11538 0.26374 0.19257
EG:experimental group, acute subarachnoid hemorrhagic patient with complementary therapies of Chinese medicine; CG, control group, acute subarachnoid hemorrhagic patient without complementary therapies of Chinese medicine; IL-1β; Df: the difference of IL-1β concentration between 1st and 5th day; IL-6 Df: the difference of IL-6 concentration between 1st and 5th day; TNF-α Df: the difference of TNF-α concentration between 1st and 5th day; S100B Df: the difference of S100B concentration between 1st and 5th day; BT1: the average BT at the 1st day after surgical operation; BT2: the average BT at the 2nd day after surgical operation; BT3: the average BT at the 3rd day after surgical operation; BT4: the average BT at the 4th day after surgical operation; BT5: the average BT at the 5th day after surgical operation;Spearman Correlation Coefficients; *p < 0.05.
Fig.1
53 patients with acute subarachnoid hemorrhage were enrolled
21 patients were excluded 3 patients were H&H grade I 2 patients were H&H grade V 16 patients refuse Chinese medicine treatment
32 patients randomized
16 patients assigned to experimental group with complementary therapies of Chinese
Medicine
16 patients assigned to control group without complementary therapies of
Chinese Medicine
1 patient death in intensive care unit
3 patients death in intensive care unit
Glascow outcome scale were measured 3 months (± 7 days) after admission
Experimental group (n=16); Control group (n=16)
Completed trial (n=32) Statistical analysis