DOI 10.1378/chest.07-0005 2007;132;532-539
Chest
Chuen-Ming Shih, Te-Chun Hsia and Chih-Yen Tu
Chia-Hung Chen, Wu-Huei Hsu, Hung-Jen Chen, Wei Chen,
*
Chronic and End-Stage Renal Disease
Thoracic Empyemas Between Patients With
Different Bacteriology and Prognosis of
http://chestjournal.chestpubs.org/content/132/2/532.full.html services can be found online on the World Wide Web at:
The online version of this article, along with updated information and
ISSN:0012-3692
) http://chestjournal.chestpubs.org/site/misc/reprints.xhtml (
written permission of the copyright holder.
this article or PDF may be reproduced or distributed without the prior Dundee Road, Northbrook, IL 60062. All rights reserved. No part of Copyright2007by the American College of Chest Physicians, 3300 Physicians. It has been published monthly since 1935.
is the official journal of the American College of Chest
Different Bacteriology and Prognosis of
Thoracic Empyemas Between Patients
With Chronic and End-Stage Renal
Disease*
Chia-Hung Chen, MD; Wu-Huei Hsu, MD, FCCP; Hung-Jen Chen, MD; Wei Chen, MD; Chuen-Ming Shih, MD; PhD; Te-Chun Hsia, MD; and Chih-Yen Tu, MD
Background: Bacterial infections are a well-documented complication in patients with chronic kidney disease (CKD) and end-stage renal disease (ESRD). However, there are no previous studies of the empyemas that can develop in these patients.
Methods: This retrospective study investigated the bacteriology and outcomes of empyema in stage 4 CKD (predialysis) and ESRD patients receiving long-term dialysis and treated in a tertiary university hospital from January 2001 to March 2006.
Results: Eighty-four stage 4 CKD patients and 40 ESRD patients had empyemas. Most empyemas (nⴝ 77, 62%) were secondary to pneumonia. Empyema culture findings were positive in 102 patients (82%): 87 microorganism were isolated in pleural fluid from 67 stage 4 CKD patients, and 39 microorganisms were isolated in pleural fluid from 35 ESRD patients. Aerobic Gram-negative organisms (nⴝ 58, 67%), especially Klebsiella pneumoniae (n ⴝ 20, 34%), were the predominant pathogens in stage 4 CKD patients; aerobic Gram-positive organisms (nⴝ 21, 54%), especially Staphylococcus aureus (nⴝ 14, 67%), were the main pathogens in ESRD patients. Compared to stage 4 CKD patients, ESRD patients had a significantly higher percentage of catheter infections (pⴝ 0.002) and aerobic Gram-positive organism bacteremia (p ⴝ 0.001), as well as a lower aerobic Gram-negative organism infection rate (p < 0.001) and a lower infection-related mortality rate (pⴝ 0.022).
Conclusion: Stage 4 CKD patients and ESRD patients with empyema have different causative pathogens and outcomes. In ESRD patients, the dialysis catheter or the dialysis process appear to alter the microbiological flora responsible for empyema. This finding has clinical implications
that clinicians need to consider. (CHEST 2007; 132:532–539)
Key words: bacteriology; chronic kidney disease; empyema; end-stage renal disease
Abbreviations: CKD⫽ chronic kidney disease; CMUH ⫽ China Medical University Hospital; CPPE ⫽ complicated
parapneumonic effusion; ESRD⫽ end-stage renal disease; GNB ⫽ Gram-negative bacilli; GPC ⫽ Gram-positive cocci; ORSA⫽ oxacillin-resistant Staphylococcus aureus; PD ⫽ peritoneal dialysis; PE ⫽ pleural empyema
C
hronic kidney disease (CKD), including end-stage renal disease (ESRD), is associated with an increased susceptibility to infection due to advancedage, immune dysfunction, underlying disease, and the dialysis procedure itself.1 CKD and ESRD
pa-tients, per se, have a greater risk of pulmonary infections.2,3 Among pulmonary infections, pleural
empyema has a high mortality. It is usually a com-plication of pneumonia but may arise from
hematog-*From the Division of Pulmonary and Critical Care Medicine, Department of Internal Medicine, China Medical University Hospital, Taichung, Taiwan.
The authors have no conflicts of interest to disclose.
Manuscript received January 2, 2007; revision accepted April 17, 2007. Reproduction of this article is prohibited without written permission from the American College of Chest Physicians (www.chestjournal. org/misc/reprints.shtml).
Correspondence to: Chih-Yen Tu, MD, Department of Internal Medicine, China Medical University Hospital, No. 2, Yude Rd, Taichung, Taiwan; e-mail: [email protected]
DOI: 10.1378/chest.07-0005
Original Research
enous seeding from an extrapulmonary focus.4
How-ever, little is known about the exact clinical characteristics and the bacteriology of empyemas in CKD patients, whether or not they are treated with maintenance dialysis.
To allow effective recommendations to be made and to improve the empirical therapeutic strategy for stage 4 CKD and ESRD patients with empyemas, we retrospectively reviewed the clinical features, caus-ative pathogens, and outcomes of stage 4 CKD and ESRD patients receiving maintenance dialysis who had empyemas develop from January 2001 to March 2006 at a tertiary medical center. In this retrospec-tive study, CKD patients were classified into predi-alysis (stage 4 CKD) and postdipredi-alysis (ESRD with long-term dialysis) groups to determine if the bacte-riology and outcomes of empyema were different between these two groups.
Materials and Methods
Enrolled Patients
China Medical University Hospital (CMUH), a 1,677-bed facility, is a tertiary medical center located in central Taiwan. On average, our dialysis unit services 565 patients a month, including 340 patients receiving hemodialysis and 225 patients on perito-neal dialysis (PD). We retrospectively reviewed the computerized medical records of stage 4 CKD and ESRD patients receiving maintenance dialysis who had been admitted to CMUH with a diagnosis of thoracic empyema. From January 2001 through March 2006, 633 patients with empyemas were admitted to CMUH. Patients were excluded if the empyemas were traumatic, the patients had undergone repeated thoracenteses, or the chart was incomplete. Of these patients with empyema, 84 had stage 4 CKD and 40 had ESRD undergoing hemodialysis or PD for⬎ 3 months.
Data Collection
The protocol was approved by the institutional review board of the CMUH. The following data were collected for each patient: age, gender, underlying disease, initial hemogram, biochemistry results, bacteria grown on pleural effusion cultures, treatment strategies, surgical interventions, and outcomes.
Definitions
CKD is the standardized nomenclature used for chronic diseases of the kidney. The National Kidney Foundation has defined the term CKD as evidence of kidney damage that persists for ⱖ 3 months.5 CKD severity is graded according to renal
function, based on the estimates of the creatinine clearance calculated using the Cockcroft-Gault equation.6Stage 4 CKD is
defined as an estimated glomerular filtration rate from 15 to 29 mL/min. ESRD (stage 5) is defined as an estimated glomerular filtration rate⬍ 15 mL/min.
Based on Light’s criteria and the literature,7simple
compli-cated parapneumonic effusion (CPPE) was defined as a pleural fluid lactic dehydrogenase level⬎ 1,000 IU/L, or a glucose level ⬍ 40 mg/dL, or a positive Gram stain or culture, with no fluid
loculation and no frank pus. Complex complicated parapneumo-nic effusion was defined as pleural fluid that met the criteria of simple CPPE, but in addition the pleural fluid was loculated. Simple pleural empyema (PE) was defined as pleural fluid that contained frank pus in a single loculus. Complex PE was defined as multiloculated pleural fluid that contained frank pus. Hospital-acquired infection-related PE was defined as an infection that occurred⬎ 48 h after admission with no evidence of infection on admission.8 Pneumonia was defined clinically; febrile patients
with purulent sputum, leukocytosis, and a new onset of pulmo-nary infiltrates on chest radiograph or CT were considered to have pneumonia. A catheter infection was defined as a positive culture finding with the same organism from a catheter segment and a peripheral blood sample.9A bacteriologic diagnosis based
on the microbiological examination of the pleural fluid and other samples was also made. Bacteremia was defined as the isolation of a bacterial pathogen from two or more blood culture samples. The bacteria found in the pleural effusion were classified as follows: aerobic Gram-positive, aerobic Gram-negative, anaero-bic, or polymicrobial bacteria. Polymicrobial infection was de-fined as the isolation of more than one strain of pathogen on the pleural effusion culture.
The administration of antimicrobial treatment was defined as inadequate if the antibiotics did not cover the infectious patho-gens or, if due to resistance, the pathopatho-gens were not susceptible
in vitro to the antibiotics. Patients in whom the infection was the
cause of death were considered to have suffered infection-related mortality.
Statistical Analysis
The data were compiled and analyzed using commercial statistical software (SPSS for Windows, version 10.0; SPSS; Chicago, IL). All continuous variables are reported as mean⫾ SD and were compared using a two-tailed Student t test. Categorical variables are reported as the number of patients and percentage. Differences in categorical variables were examined using Fisher exact test. Multivariate stepwise logistic regression analysis was used to identify independently significant factors that predicted the percentage of Gram-positive and Gram-negative organisms and infection-related mortality. All tests of significance were two sided; pⱕ 0.05 was considered statistically significant. Odds ratios and 95% confidence intervals were also calculated.
Results
Patient Characteristics, Underlying Disease, and Clinical Features
During the study period, empyemas or CPPEs developed in 84 stage 4 CKD patients and 40 ESRD patients receiving maintenance dialysis (88 men and 36 women). In the stage 4 CKD group, 11 patients had complex PE, 4 had simple PE, 60 had complex CPPE, and 9 had simple CPPE. In the ESRD group, 5 patients had complex PE, 2 had simple PE, 28 had complex CPPE, and 5 had simple CPPE. Overall, culture results were positive in 102 patients (82%), including 67 stage 4 CKD patients (66%) and 35 ESRD patients (34%). The average age of the 102 culture-positive PE or CPPE patients was 67⫾ 14 years (range, 25 to 91 years). Most patients (n⫽ 93, 91%) had a chronic underlying disease or associated
medical conditions; the most common concomitant conditions were diabetes mellitus (42%), hyperten-sive cardiovascular disease (18%), cerebrovascular disease (16%), and malignancy (15%). There was no significant difference in underlying diseases between the stage 4 CKD group and the ESRD group. Of the 35 culture-positive ESRD patients with empyema, 29 were receiving hemodialysis, and 6 were receiving PD. The mean duration of dialysis therapy was 18⫾ 26 months (range, 3 to 96 months). Table 1 shows the clinical characteristics.
Table 2 shows the etiology of the pleural empy-emas by group. In both groups, pneumonia was the predominant cause of empyema: 56 patients (67%)
in the stage 4 CKD group, and 21 patients (52%) in the ESRD group. There were no statistically signif-icant differences in the other causes of empyema between the groups, except that catheter infection was significantly more frequent in ESRD patients (25%) than in stage 4 CKD patients (6%).
Microbiology Results and Their Relationship to Clinical Characteristics
Overall, 87 organisms, including 81 aerobic bacte-ria, 4 anaerobic bactebacte-ria, and 2 tuberculosis bactebacte-ria, were isolated from 67 stage 4 CKD patients (Table 3). The most common bacteria were aerobic
Gram-Table 1—Clinical Characteristics of 102 Patients With Positive Culture Empyemas in CKD Stage 4 and ESRD With
Hemodialysis or PD* Patient Characteristics All Patients (n⫽ 102) CKD Stage 4 (n⫽ 67) ESRD (n⫽ 35) p Value HD (n⫽ 29) PD (n⫽ 6) Mean age, yr 66.8⫾ 13.6 68.8⫾ 13.6 62.6⫾ 14.0 65.2⫾ 8.2 NS Male gender 71 (70) 42 (63) 15 (52) 4 (67) NS Body length, cm 158.4⫾ 20.9 159.1⫾ 17.1 155.2⫾ 30.3 164.7⫾ 5.7 NS Body weight, kg 57.4⫾ 11.3 57.8⫾ 11.7 55.9⫾ 11.3 58.7⫾ 6.0 NS Dialysis duration, mo 0 16.1⫾ 26.4 26.7⫾ 21.6
Other underlying conditions
Diabetes mellitus 43 (42) 26 (39) 12 (41) 5 (83) NS
Malignancy 15 (15) 10 (15) 5 (17) 0 (0) NS
Cerebrovascular disease 16 (16) 11 (16) 5 (17) 0 (0) NS Hypertensive cardiovascular disease 18 (18) 10 (15) 7 (24) 1 (17) NS Congestive heart failure 11 (11) 9 (13) 2 (7) 0 (0) NS
COPD 7 (7) 5 (7) 1 (3) 1 (17) NS
Previous pulmonary tuberculosis 3 (3) 2 (3) 1 (3) 0 (0) NS
Liver cirrhosis 13 (13) 12 (18) 1 (3) 0 (0) NS
Systemic lupus erythematous 2 (2) 0 (0) 2 (7) 0 (0) NS Peptic ulcerative disease 5 (5) 4 (6) 1 (3) 0 (0) NS
Long-term steroid use 2 (2) 2 (3) 0 (0) 0 (0) NS
Alcoholism 1 (1) 0 (0) 1 (3) 0 (0) NS
No other underlying diseases 9 (9) 3 (4) 5 (17) 1 (17) NS *Data are presented as No. (%) or mean⫾ SD unless otherwise indicated. HD ⫽ hemodialysis; NS ⫽ not significant.
Table 2—Etiologies of PE Between the Groups of Chronic and ESRD*
Infection All Patients (n⫽ 124)
Patient Group p Value CKD Stage 4 (n⫽ 84) ESRD (n⫽ 40) Pneumonia 77 (62.2) 56 (66.7) 21 (52.5) 0.13 Infective endocarditis 1 (0.8) 1 (1.1) 0 (0) 0.49 Septic arthritis 4 (3.2) 3 (3.6) 1 (2.5) 0.75 Catheter infection 15 (12.1) 5 (6.0) 10 (25) 0.002 Urosepsis 2 (1.6) 1 (1.1) 1 (2.5) 0.59 Skin infection 1 (0.8) 0 (0) 1 (2.5) 0.15 Osteomyelitis 2 (1.6) 1 (1.1) 1 (2.5) 0.59 Liver abscess 2 (1.6) 2 (2.4) 0 (0) 0.33 Intra-abdominal abscess 5 (4.0) 4 (4.8) 1 (2.5) 0.55 Mediastinitis 2 (1.6) 2 (2.4) 0 (0) 0.33 Unknown 13 (10.5) 9 (10.8) 4 (10) 0.90
negative organisms (n⫽ 59, 68%). Of these, three of the most common Gram-negative pathogens ac-counted for 42 of the 87 positive-culture cases (48%): Klebsiella pneumoniae (n⫽ 20, 23%), Esch-erichia coli (n⫽ 12, 14%), and Pseudomonas aerugi-nosa (n⫽ 10, 11%). The second-most-common or-ganisms were aerobic Gram-positive oror-ganisms (23%), especially Streptococcus viridans (9%), which was the major Gram-positive empyema-causing or-ganism.
In the 35 ESRD patients who had culture-positive empyemas, 47 organisms were recovered, including 43 aerobic bacteria and 4 anaerobic bacteria. The predominant bacteria were aerobic Gram-positive organisms (n⫽ 27, 77%), of which S aureus (n ⫽ 14, 33%) and Enterococcus spp (n⫽ 7, 16%) were responsible for 78% of the aerobic Gram-positive organism infections. The second-most-common or-ganisms were aerobic Gram-negative oror-ganisms
(n⫽ 16, 34%); among these, K pneumoniae (n ⫽ 5, 12%) was the major causative organism.
Isolates from the 102 patients with positive-cul-ture empyemas were further categorized as commu-nity acquired (n⫽ 62) and hospital acquired (n⫽ 40) [Fig 1, Table 3]. The causative microorgan-isms in the community-acquired empyema group differed from those in the hospital-acquired empy-ema group. In the community-acquired empyempy-ema group, K pneumoniae (n⫽ 19, 31%) was the most common infecting organism; while in the hospital-acquired empyema group, oxacillin-resistant Staph-ylococcus aureus (ORSA) was predominant (n⫽ 10, 25%). The causative microorganisms responsible for community-acquired and hospital-acquired infec-tions differed between the stage 4 CKD and the ESRD patients. In the stage 4 CKD patients, aerobic Gram-negative pathogens were the main organisms responsible for both community-acquired and
hos-Table 3—Bacteriology of Positive-Culture Effusions in 67 CKD Stage 4 Patients and 35 ESRD Patients*
Isolates, by Class
CKD Stage 4 HD PD All Patients
CA HA CA HA CA HA CA HA Aerobic GPC 13 7 8 13 5 1 26 21 ORSA 0 3 2 7 0 0 2 10 Oxacillin-sensitive S aureus 1 2 3 2 0 0 4 4 Coagulase-negative staphylococcus 0 0 1 0 0 0 1 0 Streptococcus pneumoniae 3 0 2 1 1 0 6 1 S viridans 6 2 0 0 1 0 7 2 Streptococcus group D 1 0 0 0 0 0 1 0 Streptococcus group F 1 0 0 0 0 0 1 0 -Streptococcus 1 0 0 0 0 0 1 0 Enterococcus spp 0 0 0 3 3 1 3 4 Aerobic GNB 33 25 6 8 1 1 40 34 K pneumoniae (ESBL) 0 2 0 0 0 0 0 2 K pneumoniae (non-ESBL) 15 3 4 1 0 0 19 4 E coli (ESBL) 1 1 0 1 0 0 1 2
E coli (non ESBL) 8 2 0 0 0 1 8 3
Pseudomonas aeruginosa 3 7 2 1 1 0 6 8 Burkholderia cepacia 1 3 0 1 0 0 1 4 Enterobacter cloacae 1 1 0 1 0 0 1 2 Enterobacter gergoviae 0 1 0 0 0 0 0 1 Proteus mirabilis 1 1 0 1 0 0 1 2 Acinetobacter baumannii 0 2 0 1 0 0 0 3 Acinetobacter lowffi 0 0 0 1 0 0 0 1 Salmonella enteritidis C2 2 0 0 0 0 0 2 0 Serratia marcescens 1 0 0 0 0 0 1 0 Comamonas testosteroni 0 1 0 0 0 0 0 1 Citrobacter freundii 0 1 0 0 0 0 0 1 Gram-positive bacilli 1 0 0 0 0 0 1 0 Nocardia 1 0 0 0 0 0 1 0 Anaerobes 5 1 1 3 0 0 6 4 Peptostreptococcus magnus 0 0 0 1 0 0 0 1 Bacteroides fragilis 2 0 0 2 0 0 2 2 Fusobacterium nucleatum 1 1 1 0 0 0 2 1 Prevotella denticola 1 0 0 0 0 0 1 0 Bifidobacterium spp 1 0 0 0 0 0 1 0 Tuberculosis 2 0 0 0 0 0 2 0
pital-acquired empyemas. The most common caus-ative pathogen responsible for community-acquired empyema was K pneumoniae (n⫽ 15, 45%); the most common causative pathogen in hospital-ac-quired empyema was P aeruginosa (n⫽ 7, 28%). However, in ESRD patients, aerobic Gram-positive organisms were the main pathogens for both com-munity-acquired and hospital-acquired empyemas. S aureus (n⫽ 14, 67%) was the most common organ-ism isolated. In the ESRD group receiving PD, eight bacterial pathogens were recovered from six pa-tients; the predominant bacteria were also aerobic Gram-positive organisms (n⫽ 6, 75%), especially Enterococcus (n⫽ 4, 50%).
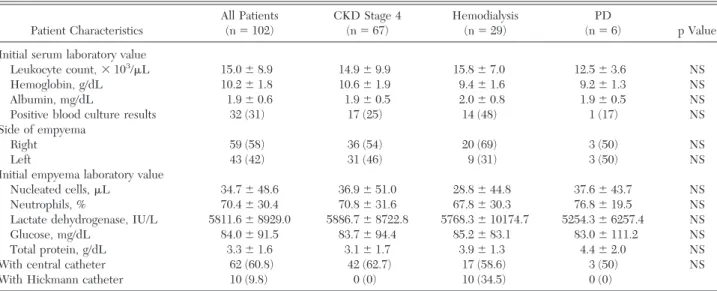
Laboratory data are shown by group in Table 4.
There were no statistically significant differences between the stage 4 CKD and ESRD groups. Treatments and Outcomes of Patients With Culture-Positive Empyemas
A total 110 patients (89%) received a pigtail catheter implant, and 14 patients (11%) required chest tube drainage as judged by the attending physician. Among these 110 patients with pigtail catheter drainage, 9 patients (8%) required fibrino-lytic therapy because of a poor response to simple pigtail catheter drainage of CPPEs or empyemas. Of these patients receiving fibrinolytic therapy, seven patients were clinically improved and the other two
Figure 1. Bacteriology of 102 culture-positive effusions in patients with community-acquired and hospital-acquired infections. GPB⫽ Gram-positive bacilli; TB ⫽ tuberculosis; OSSA ⫽ oxacillin-sen-sitive S aureus.
Table 4 —Comparison of Laboratory Values of Serum and Pleural Effusion Between CKD Stage 4 and ESRD
Patients Receiving Hemodialysis and PD*
Patient Characteristics All Patients (n⫽ 102) CKD Stage 4 (n⫽ 67) Hemodialysis (n⫽ 29) PD (n⫽ 6) p Value Initial serum laboratory value
Leukocyte count,⫻ 103/L 15.0⫾ 8.9 14.9⫾ 9.9 15.8⫾ 7.0 12.5⫾ 3.6 NS
Hemoglobin, g/dL 10.2⫾ 1.8 10.6⫾ 1.9 9.4⫾ 1.6 9.2⫾ 1.3 NS Albumin, mg/dL 1.9⫾ 0.6 1.9⫾ 0.5 2.0⫾ 0.8 1.9⫾ 0.5 NS Positive blood culture results 32 (31) 17 (25) 14 (48) 1 (17) NS Side of empyema
Right 59 (58) 36 (54) 20 (69) 3 (50) NS
Left 43 (42) 31 (46) 9 (31) 3 (50) NS
Initial empyema laboratory value
Nucleated cells,L 34.7⫾ 48.6 36.9⫾ 51.0 28.8⫾ 44.8 37.6⫾ 43.7 NS Neutrophils, % 70.4⫾ 30.4 70.8⫾ 31.6 67.8⫾ 30.3 76.8⫾ 19.5 NS Lactate dehydrogenase, IU/L 5811.6⫾ 8929.0 5886.7⫾ 8722.8 5768.3⫾ 10174.7 5254.3⫾ 6257.4 NS Glucose, mg/dL 84.0⫾ 91.5 83.7⫾ 94.4 85.2⫾ 83.1 83.0⫾ 111.2 NS Total protein, g/dL 3.3⫾ 1.6 3.1⫾ 1.7 3.9⫾ 1.3 4.4⫾ 2.0 NS With central catheter 62 (60.8) 42 (62.7) 17 (58.6) 3 (50) NS With Hickmann catheter 10 (9.8) 0 (0) 10 (34.5) 0 (0)
patients were treated using video-assisted thoraco-scopic surgery due to poor control of the infection and/or poor drainage function. The successful rate of fibrinolytic therapy was 78%.
Thirty-two patients had positive blood culture findings. The incidence of aerobic Gram-positive coccus (n⫽ 21, 66%) bacteremia was higher than that of aerobic Gram-negative bacilli (GNB) [n⫽ 11, 34%; Table 5]. The rate of bacteremia was higher in the ESRD group (43%, 15 of 35 patients) than in the stage 4 CKD group (25%, 17 of 67 patients). Overall, the infection-related mortality rate was 43% (53 of 124 patients); 40 of 84 stage 4 CKD patients (48%) died, and 13 of 40 ESRD patients (33%) died. GNB infection (n⫽ 32, 63%) was the predominant cause of infection-related mor-tality.
Discussion
To the best of our knowledge, this is the first study reported in the English-language literature that fo-cused on empyemas in stage 4 CKD (predialysis) patients and ESRD patients receiving long-term dialysis. In stage 4 CKD patients with empyema, aerobic Gram-negative organisms were the predom-inant pathogens; K pneumoniae was the most fre-quently isolated sole pathogen. However, in ESRD patients receiving maintenance dialysis, aerobic Gram-positive organisms were the predominant pathogens. S aureus and Enterococcus spp were the most frequently isolated sole pathogens.
Over the past 20 years, aerobic Gram-positive
organisms have been found to be the most common isolates in acute empyema,10 –12particularly S aureus
and S pneumoniae, which together accounted for approximately 70% of aerobic Gram-positive iso-lates. However, as recently reported,13 there has
been a significant increase in Gram-negative infec-tion. In our study, aerobic Gram-negative bacteria were the major pathogens isolated from the empy-emas of stage 4 CKD patients. Most stage 4 CKD patients (96%) with culture-positive empyema had underlying diseases; most patients were relatively immunocompromised hosts with diabetes mellitus (39%), malignancy (15%), or liver cirrhosis (18%). It is well known that immunocompromised patients are prone to pleural involvement with fungal or aerobic GNB infections.12,14 The markedly high rate of
Gram-negative bacterial infection in the empyemas of the stage 4 CKD patients may be associated with the high incidence of underlying disease and poor renal function. However, ESRD patients receiving long-term dialysis had a higher rate of bacteremia and aerobic Gram-positive empyemas despite a sim-ilar incidence of underlying disease and a simsim-ilar frequency of central venous catheter implants (Table 1, 4). However, a higher incidence of Hickman insertion for maintenance dialysis was noted in ESRD patients. These factors may increase the frequency of these patients being exposed to poten-tially infectious factors during the normal course of dialysis therapy or being exposed to a catheter infection15–17; this may explain the bacteriologic
find-ings in the empyemas of ESRD patients. In the present study, S aureus and Enterococcus spp, which
Table 5—Incidence of Bacteremia, Pneumonia, Inadequate Antibiotic Treatment, and Mortality of Patients in CKD
Stage 4 and ESRD Receiving Hemodialysis and PD*
Patient Characteristics Bacteremia Pulmonary Infiltration Inadequate Antibiotic Use Infection-Related Mortality Stage 4 CKD group (n⫽ 67) Aerobic GNB (n⫽ 42) 9 (21) 25 (60) 16 (38) 28 (67) Aerobic GPC (n⫽ 16) 8 (50) 12 (75) 3 (19) 7 (44) Aerobic Gram-positive bacilli (n⫽ 1) 0 (0) 1 (100) 0 (0) 1 (100) Anaerobes (n⫽ 1) 0 (0) 0 (0) 0 (0) 0 (0) Tuberculosis (n⫽ 2) 0 (0) 2 (100) 2 (100) 0 (0) Mixed (n⫽ 5) 0 (0) 4 (80) 3 (100) 4 (80) Hemodialysis group (n⫽ 29) Aerobic GNB (n⫽ 9) 2 (22) 6 (67) 3 (33) 4 (44) Aerobic GPC (n⫽ 16) 12 (75) 5 (31) 9 (56) 6 (38) Anaerobes (n⫽ 1) 0 (0) 1 (100) 0 (0) 0 (0) Mixed (n⫽ 3) 0 (0) 3 (100) 1 (33) 1 (33) PD group (n⫽ 6) Aerobic GNB (n⫽ 0) 0 (0) 0 (0) 0 (0) 0 (0) Aerobic GPC (n⫽ 4) 1 (25) 2 (50) 2 (50) 1 (25) Anaerobes (n⫽ 0) 0 (0) 0 (0) 0 (0) 0 (0) Mixed (n⫽ 2) 0 (0) 2 (100) 2 (50) 1 (50)
are normal skin and intestinal flora, were the most frequently isolated sole pathogens in ESRD patients receiving hemodialysis and PD.
Compared to the recently published Multicenter Intrapleural Sepsis Trial 1,18,19 there were clear
differences between the general population and CKD patients (with or without dialysis) in the patho-gens isolated in those with empyemas (Fig 1). In the present study, aerobic Gram-negative organisms rather than aerobic Gram-positive organisms, and particularly K pneumoniae rather than Streptococcus spp, were the main causative microorganisms of empyema in stage 4 CKD patients. Theoretically, in relatively immunocompromised hosts, renal func-tional impairment or underlying disease may favor these pathogens. However, among hospital-acquired infections, ORSA was still the most common patho-gen, not only in the general population, but also in patients with chronic kidney disease. These results may imply that, when treating patients with renal functional impairment, different antibiotics should be used empirically in hospital- and community-acquired empyemas.
In our study, 61% of stage 4 CKD patients and 67% of ESRD patients receiving hemodialysis had pneumonia at the time that an aerobic GNB empy-ema was diagnosed (Table 4). It is thought that the infectious process occurring in the pleural cavity may be due to bacteria from the lung parenchyma sec-ondarily invading the pleural space. A similar situa-tion was also noted in the stage 4 CKD group, in which empyema was caused by aerobic Gram-posi-tive cocci (GPC). However, only 31% of hemodialy-sis patients with aerobic GPC empyemas had pneu-monia; however, 75% of hemodialysis patients had bacteremia. Therefore, the majority of GPC empy-emas were related to the presence of bacteremia in dialysis patients due to hematogenous seeding of an extrapulmonary focus, likely related to dialysis cath-eter implants and/or the dialysis process.
A higher mortality rate was noted among stage 4 CKD patients than among ESRD patients. This may be due to the fact that GNB were the predominant pathogens in the stage 4 CKD 4 group; GNB are more toxic than GPC, which were the major patho-gen in the ESRD group. Hence, patients with S aureus empyema receiving long-term hemodialysis have a favorable prognosis, particularly compared to CKD patients not receiving dialysis.
In our series, the successful rate of fibrinolytic therapy in these patients with poor catheter drainage was 78% (seven of nine patients). Our finding con-firms that fibrinolytic therapy is effective in the treatment of poor catheter drainage in patients with CPPEs or empyemas, which was comparable to these previous studies.20,21
Previous studies22–24 noted an important
associa-tion between inadequate antimicrobial treatment for bacteremia and hospital mortality. In the present study, among the stage 4 CKD group with aerobic Gram-negative empyema, 16 patients initially re-ceived inadequate antimicrobial treatment, and their mortality rate was 81% (13 of 16 patients); the other 27 patients initially received adequate antimicrobial treatment, and their mortality rate was 56% (15 of 27 patients). Among the stage 4 CKD group with aerobic Gram-positive empyema, 2 patients initially received inadequate antimicrobial treatment (mor-tality rate, 50%; 1 of 2 patients); the other 14 patients initially received adequate antimicrobial treatment (mortality rate, 42%; 6 of 14 patients). Similar results were observed in the ESRD patients receiving he-modialysis. In the aerobic Gram-negative group, three patients initially received inadequate antimi-crobial treatment (mortality rate, 67%; two of three patients); the other six patients initially received adequate antimicrobial treatment (mortality rate, 33%; two of six patients). In the aerobic Gram-positive group, nine patients initially received inad-equate antimicrobial treatment (mortality rate, 33%; three patients); the other seven patients initially received adequate antimicrobial treatment (mortal-ity rate, 43%; three of seven patients). Due to the lower toxicity of Gram-positive organisms, inade-quate antimicrobial treatment did not contribute to hospital mortality. However, inadequate antimicro-bial treatment was an important factor in patients with aerobic Gram-negative empyemas, both in the stage 4 CKD group and the ESRD group.
The present study had several limitations. First, all of the patients were recruited from our hospital.The prevalence of etiologies may differ in other geo-graphic regions. Second, this was a retrospective study. Also, the sample size of the ESRD group receiving maintenance PD therapy was small, even though an average of 225 ESRD patients per month have been receiving PD in our hospital in recent years. A large prospective, randomized trial is re-quired to confirm our results with respect to the ESRD group receiving maintenance PD therapy who have empyema develop.
In conclusion, empyema has a high mortality in stage 4 CKD and ESRD patients. The causative pathogens and outcomes of empyemas differ be-tween stage 4 CKD patients and ESRD patients; it appears that the dialysis catheter or the dialysis process itself are responsible for the differences. Clinicians treating patients with renal functional impairment who have empyema develop must take into consideration the dialysis status when empiri-cally choosing antibiotics while awaiting culture
re-sults. Thus, these findings are clinically applicable in the treatment of patients with chronic renal failure and empyema.
References
1 Pesanti EL. Immunologic defects and vaccination in patients with chronic renal failure. Infect Dis Clin North Am 2001; 15:813– 832
2 Sarnak MJ, Jaber BL. Pulmonary infectious mortality among patients with end-stage renal disease. Chest 2001; 120:1883– 1887
3 Berman SJ, Johnson EW, Nakatsu C, et al. Burden of infection in patients with end-stage renal disease requiring long-term dialysis. Clin Infect Dis 2004; 39:1747–1753 4 Richard EB, Christopher JS. Pleural empyema. Clin Infect
Dis 1996; 22:747–764
5 K/DOQI clinical practice guidelines for chronic kidney dis-ease: evaluation, classification, and stratification. Am J Kidney Dis 2002; 39:S1–S266
6 Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron 1976; 16:31– 41
7 Light RW. A new classification of parapneumonic effusions and empyema. Chest 1995; 108:299 –301
8 Soo Hoo GW, Wen YE, Nguyen TV, et al. Impact of clinical guidelines in the management of severe hospital-acquired pneumonia. Chest 2005; 128:2778 –2787
9 Mermel L, Farr BM, Sherertz RJ, et al. Guidelines for the management of intravascular catheter-related infections. Clin Infect Dis 2001; 32:1249 –1272
10 Brook I, Frazier EH. Aerobic and anaerobic microbiology of empyema: a retrospective review in two military hospitals. Chest 1993; 103:1502–1507
11 Varkey B, Rose HD, Kutty CP, et al. Empyema thoracis during a ten-year period: analysis of 72 cases and comparison to a previous study (1952–1967). Arch Intern Med 1981; 141:1771–1776
12 Smith JA, Mullerworth MH, Westlake GW, et al. Empyema thoracis: a 14-year experience in a teaching center. Ann Thorac Surg 1991; 51:39 – 42
13 Albrecht SJ, Fishman NO, Kitchen J, et al. Reemergence of Gram-negative health care-associated bloodstream infections. Arch Intern Med 2006; 166:1289 –1294
14 Vianna NJ. Nontuberculous bacterial empyema in patients with and without underlying disease. JAMA 1971; 215:69 –75 15 Kessler M, Hoen B, Mayeux D, et al. Bacteremia in patients on chronic hemodialysis: a multicenter prospective survey. Nephron 1993; 64:95–100
16 Hoen B, Kessler M, Hestin D, et al. Risk factors for bacterial infections in chronic hemodialysis adult patients: a multicen-tre prospective survey. Nephrol Dial Transplant 1995; 10: 377–381
17 Hoen B, Paul-Dauphin ADH, Kessler M. EPIBACDIAL: a multicenter prospective study of risk factors for bacteremia in chronic hemodialysis patients. J Am Soc Nephrol 1998; 9:869 – 876
18 Maskell NA, Davies CWH, Nunn AJ, et al. UK controlled trial of intrapleural streptokinase for pleural infection. N Engl J Med 2005; 352:865– 874
19 Maskell NA, Batt S, Emma LH, et al: the bacteriology of pleural infection by genetic and standard methods and its mortality significance. Am J Respir Crit Care Med 2006; 174:817– 823
20 Bouros D, Schiza S, Tzanakis N, et al. Intrapleural urokinase versus normal saline in the treatment of complicated parap-neumonic effusions and empyema: a randomized, double-blind study. Am J Respir Crit Care Med 1999; 159:37– 42 21 Bouros D, Schiza S, Tzanakis N, et al. Intrapleural urokinase
in the treatment of complicated parapneumonic pleural effusions and empyema. Eur Respir J 1996; 9:1656 –1659 22 Ibrahim EH, Sherman G, Ward S, et al. The influence of
inadequate antimicrobial treatment of bloodstream infections on patient outcomes in the ICU setting. Chest 2000; 118: 146 –155
23 Leibovici L, Shraga I, Drucker M, et al. The benefit of appropriate empirical antibiotic treatment in patients with bloodstream infection. J Intern Med 1998; 244:379 –386 24 Kollef MH, Sherman G, Ward S, et al. Inadequate
antimi-crobial treatment of infections: a risk factor for hospital mortality among critically ill patients. Chest 1999; 115:462– 474
DOI 10.1378/chest.07-0005
2007;132; 532-539
Chest
Shih, Te-Chun Hsia and Chih-Yen Tu
Chia-Hung Chen, Wu-Huei Hsu, Hung-Jen Chen, Wei Chen, Chuen-Ming
*
Patients With Chronic and End-Stage Renal Disease
Different Bacteriology and Prognosis of Thoracic Empyemas Between
January 12, 2012
This information is current as of
http://chestjournal.chestpubs.org/content/132/2/532.full.html Updated Information and services can be found at:
Updated Information & Services
http://chestjournal.chestpubs.org/content/132/2/532.full.html#ref-list-1 This article cites 24 articles, 17 of which can be accessed free at: References
http://www.chestpubs.org/site/misc/reprints.xhtml found online at:
Information about reproducing this article in parts (figures, tables) or in its entirety can be Permissions & Licensing
http://www.chestpubs.org/site/misc/reprints.xhtml Information about ordering reprints can be found online: Reprints
"Services" link to the right of the online article.
Receive free e-mail alerts when new articles cite this article. To sign up, select the Citation Alerts
PowerPoint slide format. See any online figure for directions.
articles can be downloaded for teaching purposes in
CHEST
Figures that appear in