Pretreatment Quality of Life As a Predictor of
Distant Metastasis and Survival for Patients With
Nasopharyngeal Carcinoma
Fu-Min Fang, Wen-Ling Tsai, Chih-Yen Chien, Hui-Chun Chen, Hsuan-Chih Hsu, Tai-Lin Huang,
Tsair-Fwu Lee, Hsuan-Ying Huang, and Chien-Hung Lee
From the Chang Gung Memorial Hospital-Kaohsiung Medical Center, Chang Gung University College of Medicine, Taiwan; and the Kaohsiung Medical University, Kaohsiung, Taiwan. Submitted February 17, 2010; accepted July 2, 2010; published online ahead of print at www.jco.org on August 16, 2010.
Supported by Grant No. NSC 90- 2320-B-182A-006- from the National Science Council, Taiwan; and Grants No. CMRPG860501 and CMRPG860502 from the Chang Gung Memorial Hospi-tal, Taiwan.
Presented in part at the first Congress of Asian Society of Head and Neck Oncology, September 18-19, 2009, Taipei, Taiwan.
Authors’ disclosures of potential con-flicts of interest and author contribu-tions are found at the end of this article.
Corresponding author: Fu-Min Fang, MD, PhD, Department of Radiation Oncology, Chang Gung Memorial Hospital-Kaohsiung Medical Center, 123 Ta-Pei Rd, Niao Sung Hsian, Kaohsiung Hsien, Taiwan; e-mail: fang2569@adm .cgmh.org.tw.
© 2010 by American Society of Clinical Oncology
0732-183X/10/2828-4384/$20.00 DOI: 10.1200/JCO.2010.28.8324
A
B
S
T
R
A
C
T
Purpose
The purpose of this study was to examine the prognostic value of pretreatment quality of life (QoL)
data on locoregional control (LRC), distant metastasis-free survival (DMFS), and overall survival
(OS) in patients with nasopharyngeal carcinoma (NPC).
Patients and Methods
A total of 347 new patients with NPC, who were curatively treated by conformal radiotherapy from
March 2003 to December 2007, were recruited. The Taiwan Chinese version of the European
Organization for Research and Treatment of Cancer Quality of Life Questionnaire C30 was
completed before treatment. Multivariate Cox’s proportional hazards models were used to analyze
the impact of clinical and QoL variables on the treatment results.
Results
The 5-year LRC, DMFS, and OS rates were 72.9%, 79.1%, and 68.4%, respectively. After
adjusting the clinical variables, 10 QoL variables were observed to be significantly (P
⬍ .05) related
to OS, and four QoL variables were related to DMFS. No QoL variable was predictive of LRC.
Among the QoL variables that significantly predicted OS and DMFS, physical functioning was the
most powerful predictor. A 10-point increase in the physical functioning score was associated with
a 23% (95% CI, 12% to 34%) reduction in the likelihood of death and a 22% (95% CI, 9% to 36%)
reduction in the likelihood of distant metastasis.
Conclusion
Our findings indicate that pretreatment QoL variables, especially physical functioning, provide
easily available prognostic value for distant metastasis and survival in patients with NPC.
J Clin Oncol 28:4384-4389. © 2010 by American Society of Clinical Oncology
INTRODUCTION
Nasopharyngeal carcinoma (NPC) is an endemic
head and neck epithelial malignancy.
Approxi-mately 70% of patients with NPC present with stage
III or IV disease at the initial diagnosis.
1Although
advances in diagnostic imaging, radiotherapeutic
techniques, and chemotherapy regimens have
achieved improved locoregional control (LRC),
19% to 29% of patients will have distant metastasis
with controlled locoregional disease.
2-4A variety of
strategies adopting a combination of chemotherapy
and radiotherapy (RT) have been investigated in
patients with advanced disease, but most trials failed
to observe an effect of reducing the incidence of distant
metastases.
4,5To date, distant metastasis is still the
major cause of mortality in patients with NPC.
Patient self-assessment of quality of life (QoL)
has been acknowledged as an important treatment
end point and has been used in clinical oncology
trials to compare different treatment strategies.
Evi-dence is now accumulating to indicate that QoL may
also have prognostic significance for survival of
pa-tients with cancer. Pretreatment QoL has been
shown to be predictive of treatment outcome in
terms of LRC and/or overall survival (OS) in
pa-tients with advanced lung cancer,
6,7breast cancer,
8,9esophageal cancer,
10and head and neck cancer
(HNC)
11-16beyond the established factors such as
stage, grade, or performance status. Nevertheless,
thus far, no systematic study has specifically
ad-dressed the prognostic role of pretreatment QoL in
NPC patients with special attention not only to
sur-vival but also to distant metastases.
If pretreatment QoL is predictive of survival or
distant metastasis among patients with NPC, the
QoL instrument may become a practical and
nonin-vasive means of identifying those patients at high
J
OURNAL OF
C
LINICAL
O
NCOLOGY
O R I G I N A L
R E P O R T
V O L U M E 2 8 䡠 N U M B E R 2 8 䡠 O C T O B E R 1 2 0 1 0
4384 © 2010 by American Society of Clinical Oncology
Downloaded from jco.ascopubs.org on July 29, 2014. For personal use only. No other uses without permission.
Copyright © 2010 American Society of Clinical Oncology. All rights reserved.
risk who need tailored medical support before treatment or those who
must be observed more closely after treatment. We have used the
Taiwan Chinese version of the European Organization for Research
and Treatment of Cancer Quality of Life Questionnaire-Core 30
(EORTC QLQ-C30) questionnaire as an effective tool to assess the
outcomes of patients with NPC.
17,18Hence, the purpose of this study
was to examine the relationships between pretreatment EORTC
QLQ-C30 data and LRC, distant metastasis-free survival (DMFS), and OS
among patients with NPC after controlling for the clinical variables.
PATIENTS AND METHODS
Patients
This was a longitudinal investigation. From March 2003 to December
2007, 441 newly diagnosed patients with NPC were treated at Chang Gung
Memorial Hospital–Kaohsiung Medical Center. There were 347 patients
re-cruited in the study. Exclusion criteria included distant metastasis at the initial
diagnosis (n
⫽21),inabilitytocompletetheprescribedtreatmentcourseofRT
(n
⫽ 18), lost to follow-up after treatment (n ⫽ 22), with previous or
synchro-nous malignancies (n
⫽ 9), and unable to understand (n ⫽ 10) or unwilling to
complete (n
⫽ 14) the Taiwan Chinese version of the EORTC QLQ-C30. The
local institutional research board approved this study, and written
in-formed consent was obtained from each participant. The pretreatment
evaluation included a complete history, physical examination, and
com-prehensive imaging studies, including chest x-ray, computed tomography,
or magnetic resonance image scans of the head and neck, a bone scan, and
an abdominal sonogram.
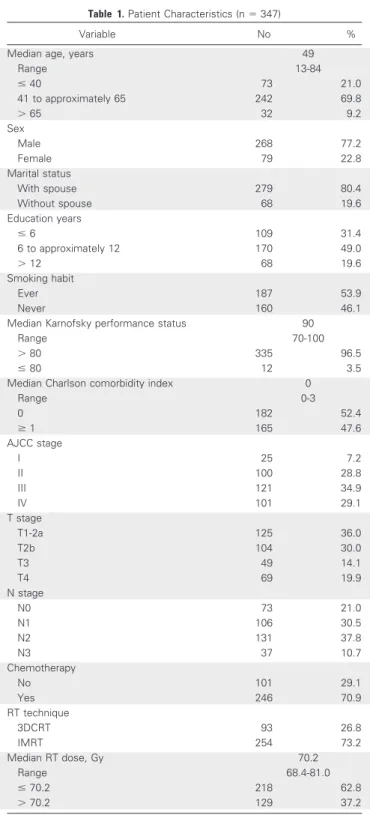
The patient clinical characteristics are presented in Table 1. The median
age was 49 years (range, 13 to 84 years). Most patients were male (77.2%),
married (80.4%), and educated for fewer than 12 years (80.4%).
Approxi-mately one half of the patients had a habit of smoking (53.9%) or self-reported
one or more comorbid conditions (47.6%) based on the Charlson
Comorbid-ity Index.
19The distributions of the clinical stages according to the American
Joint of Cancer Committee (AJCC) staging system published in 2002 were I, 25
(7.2%); II, 100 (28.8%); III, 121 (34.9%); and IV, 101 (29.1%). All patients
were irradiated for the entire treatment course using a conformal technique; 93
patients (26.8%) received three-dimensional conformal RT, and 254 (73.2%)
patients received intensity-modulated RT. The technical details of the NPC
three-dimensional conformal RT/intensity-modulated RT have been
de-scribed previously.
17,18The prescribed dose ranged from 68.4 to 81.0 Gy
depending on the tumor stage, with a daily fraction of 1.8 Gy and 5
fractions per week. Two hundred forty-six patients (70.9%) received
com-bination chemotherapy because of their advanced stage. The prescribed
chemotherapy regimen was 80 to 100 mg/m
2cisplatin intravenous bolus
on day 1 and 800 to 1,000 mg/m
2/24 hours continuous intravenous
infu-sion of fluorouracil on days 1 to 4 of each cycle. One hundred sixty patients
(65.0%) received at least three cycles of chemotherapy before, during,
and/or after the RT course.
Instruments
A variety of QoL instruments have been used as an outcome predictor,
including the EORTC QLQ-C30,
6-8,10-13Short-Form 36 (SF-36),
14,16Func-tional Assessment of Cancer Therapy–Head and Neck,
15or Physical
Self-Efficacy Scale.
20The EORTC QLQ-C30 was chosen for this study because it is
one of the most widely implemented questionnaires, with more than 15 years
of research invested to develop an integrated, modular approach, as well as
utilization in international clinical trials, and the Taiwan Chinese version,
which was obtained from the Quality of Life Unit, EORTC Data Center in
Brussels, Belgium,
21,22was available and easily completed by our patients.
Moreover, it is one of the most common QoL instruments used to predict
survival in patients with cancer. Patients completed the questionnaire at their
first visit to the oncology clinic. The EORTC QLQ-C30 questionnaire
incor-porates a range of QoL issues relevant to a broad range of patients with cancer.
It has been validated for many types of cancer including NPC.
23It contains five
functional scales (physical, role, cognitive, emotional, and social), three
symp-tom scales (fatigue, pain, and nausea/vomiting), a global QoL scale, and six
single-items (dyspnea, insomnia, appetite loss, constipation, diarrhea, and
financial difficulties). All scales and items range in score from 0 to 100. A high
score for a functional or global QoL scale represents a relatively high/healthy
level of functional or global QoL, whereas a high score for a symptom scale or
item represents a high level of symptoms or problems.
Table 1. Patient Characteristics (n⫽ 347)
Variable No %
Median age, years 49
Range 13-84 ⱕ 40 73 21.0 41 to approximately 65 242 69.8 ⬎ 65 32 9.2 Sex Male 268 77.2 Female 79 22.8 Marital status With spouse 279 80.4 Without spouse 68 19.6 Education years ⱕ 6 109 31.4 6 to approximately 12 170 49.0 ⬎ 12 68 19.6 Smoking habit Ever 187 53.9 Never 160 46.1
Median Karnofsky performance status 90
Range 70-100
⬎ 80 335 96.5
ⱕ 80 12 3.5
Median Charlson comorbidity index 0
Range 0-3 0 182 52.4 ⱖ 1 165 47.6 AJCC stage I 25 7.2 II 100 28.8 III 121 34.9 IV 101 29.1 T stage T1-2a 125 36.0 T2b 104 30.0 T3 49 14.1 T4 69 19.9 N stage N0 73 21.0 N1 106 30.5 N2 131 37.8 N3 37 10.7 Chemotherapy No 101 29.1 Yes 246 70.9 RT technique 3DCRT 93 26.8 IMRT 254 73.2 Median RT dose, Gy 70.2 Range 68.4-81.0 ⱕ 70.2 218 62.8 ⬎ 70.2 129 37.2
Abbreviations: AJCC, American Joint of Cancer Committee; RT, radiotherapy; 3DCRT, three-dimensional conformal RT; IMRT, intensity-modulated RT.
QoL Predict Distant Metastasis and Survival in NPC
www.jco.org © 2010 by American Society of Clinical Oncology 4385
Downloaded from jco.ascopubs.org on July 29, 2014. For personal use only. No other uses without permission.
Copyright © 2010 American Society of Clinical Oncology. All rights reserved.
REFERENCES
1. Lee AWM, Poon YF, Foo W: Retrospective
analysis of 5037 patients with nasopharyngeal car-cinoma treated during 1976-1985: Overall survival and patterns of failure. Int J Radiat Oncol Biol Phys 23:261-270, 1992
2. Kwong D, Sham J, Choy D: The effect of
loco-regional control on distant metastatic dissemi-nation in carcinoma of the nasopharynx: An analysis of 1301 patients. Int J Radiat Oncol Biol Phys 30:1029-1036, 1994
3. Geara FB, Sanguineti G, Tucker SL, et al:
Carcinoma of the nasopharynx treated by radiother-apy alone: Determinants of distant metastasis and survival. Radiother Oncol 43:53-61, 1997
4. Lee N, Harris J, Garden AS, et al:
Intensity-modulated radiation therapy with or without chem-otherapy for nasopharyngeal carcinoma: Radiation therapy oncology group phase II trial 0225. J Clin Oncol 27:3684-3690, 2009
5. Guigay J: Advances in nasopharyngeal
carci-noma. Curr Opin Oncol 20:264-269, 2008
6. Coates A, Porzsolt F, Osoba D: Quality of life
in oncology practice: Prognostic value of EORTC QLQ-C30 scores in patients with advanced malig-nancy. Eur J Cancer 33:1025-1030, 1997
7. Langendijk H, Aaronson NK, de Jong JMA, et
al: The prognostic impact of quality of life assessed with the EORTC QLQ-C30 in inoperable non-small cell lung carcinoma treated with radiotherapy. Ra-diother Oncol 55:19-25, 2000
8. Efficace F, Therasse P, Piccart MJ, et al:
Health-related quality of life parameters as prognos-tic factors in a nonmetastaprognos-tic breast cancer popula-tion: An international multicenter study. J Clin Oncol 22:3381-3384, 2004
9. Coates A, Gebski V, Signorini D, et al:
Prog-nostic value of quality-of-life scores during chemo-therapy for advanced breast cancer. J Clin Oncol 10:1833-1838, 1992
10. Fang FM, Tsai WL, Chiu HC, et al: Quality of
life as a survival predictor for esophageal squamous
cell carcinoma treated with radiotherapy. Int J Radiat Oncol Biol Phys 58:1394-1404, 2004
11. de Graeff A, de Leeuw JRJ, Ros WJG, et al:
Sociodemographic factors and quality of life as prog-nostic indicators in head and neck cancer. Eur J Cancer 37:332-339, 2001
12. Fang FM, Liu YT, Tang Y, et al: Quality of life
as a predictor for patients with advanced head and neck carcinoma treated with radiation therapy. Can-cer 100:425-432, 2004
13. Meyer F, Fortin A, Michel Gelinas M, et al:
Health-related quality of life as a survival predictor for patients with localized head and neck cancer treated with radiation therapy. J Clin Oncol 27:2970-2976, 2009
14. Karvonen-Gutierrez CA, Ronis DL, Fowler KE,
et al: Quality of life scores predict survival among patients with head and neck cancer. J Clin Oncol 26:2754-2760, 2008
15. Siddiqui F, Pajak TF, Watkins-Bruner D, et al:
Pretreatment quality of life predicts for locoregional control in head and neck cancer patients: A radiation therapy oncology group analysis. Int J Radiat Oncol Biol Phys 70:353-360, 2008
16. Grignon LM, Jameson MJ, Karnell LH, et al:
General health measures and long-term survival in patients with head and neck cancer. Arch Otolaryn-gol Head Neck Surg 133:471-476, 2007
17. Fang FM, Chien CY, Tsai WL, et al: Quality of life
and survival outcome for patients with nasopharyngeal carcinoma receiving three-dimensional conformal radio-therapy vs. intensity-modulated radioradio-therapy: A longitudi-nal study. Int J Radiat Oncol Biol Phys 72:356-364, 2008
18. Fang FM, Tsai WL, Chen HC, et al:
Intensity-modulated or conformal radiotherapy improves quality of life of patients with nasopharyngeal carcinoma: Compar-isons of four radiotherapy techniques. Cancer 109:313-321, 2007
19. Charlson ME, Pompei P, Ales KL, et al: A new
method of classifying prognostic comorbidity in lon-gitudinal studies: Development and validation. J Chron Dis 40:373-383, 1987
20. De Boer MF, Van den Borne B, Pruyn JF, et al:
Psychosocial and physical correlates of survival and
recurrence in patients with head and neck carci-noma: Results of a 6-year longitudinal study. Cancer 83:2567-2579, 1998
21. Aronson NK, Ahmedzai S, Bergman B, et al:
The European Organisation for Research and Treat-ment of Cancer QLQ-C30: A quality-of-life instru-ment for use in international clinical trials in oncology. J Natl Cancer Inst 85:365-376, 1993
22. Fayers P, Aaronson NK, Bjordal K, et al:
EORTC QLQ-C30 Scoring Manual (ed 2). Brussels, Belgium, European Organisation for Research and Treatment of Cancer Data Center, 1999
23. Chie WC, Hong RL, Lai CC, et al: Quality of life
in patients of nasopharyngeal carcinoma: Validation of the Taiwan Chinese version of the EORTC QLQ-C30 and the EORTC QLQ-H&N35. Qual Life Res 12:93-98, 2003
24. Yates JW, Chalmer B, McKegney FP:
Evalua-tion of patients with advanced cancer using the Karnofsky performance status. Cancer 45:2220-2224, 1980
25. Fuller CD, Braden CJ, Thomas CR: Quality of
life: From a tower of barbel towel toward a unified voice. Int J Radiat Oncol Biol Phys 58:1334-1335, 2004
26. Lin JC, Chen KY, Wang WY, et al: Detection of
Epstein-Barr virus DNA the peripheral-blood cells of patients with nasopharyngeal carcinoma: Relation-ship to distant metastasis and survival. J Clin Oncol 19:2607-2615, 2001
27. Leung SF, Zee B, Ma BB, et al: Plasma
Epstein-Barr viral deoxyribonucleic acid quantitation complements tumor-node-metastasis staging prog-nostication in nasopharyngeal carcinoma. J Clin On-col 24:5414-5418, 2006
28. Tse KP, Tsang NM, Chen KD, et al: MCP-1
promoter polymorphism at 2518 is associated with metastasis of nasopharyngeal carcinoma after treat-ment. Clin Cancer Res 13:6320-6326, 2007
29. Watson M, Haviland JS, Greer S, et al:
Influ-ence of psychological response on survival in breast cancer; a population base cohort study. Lancet 354: 1331-1336, 1999
■ ■ ■
QoL Predict Distant Metastasis and Survival in NPC
www.jco.org © 2010 by American Society of Clinical Oncology 4389