Modi
fiable determinants of hearing impairment in adults
Weihai Zhan
a,⁎
, Karen J. Cruickshanks
a,b, Barbara E.K. Klein
b, Ronald Klein
b, Guan-Hua Huang
c,
James S. Pankow
d, Ronald E. Gangnon
e,f, Theodore S. Tweed
ga
Department of Population Health Sciences, University of Wisconsin School of Medicine and Public Health, Madison, WI 53726, USA
b
Department of Ophthalmology and Visual Sciences, University of Wisconsin School of Medicine and Public Health, Madison, WI 53726, USA
c
Institute of Statistics, National Chiao Tung University, Hsinchu 300, Taiwan
d
Division of Epidemiology Community Health, University of Minnesota, Minneapolis, MN 55454, USA
e
Department of Population Health Sciences, University of Wisconsin, Madison, WI 53726, USA
fDepartment of Biostatistics and Medical Informatics, University of Wisconsin, Madison, WI 53726, USA g
Department of Communication Disorders, University of Wisconsin, Madison, WI 53726, USA
a b s t r a c t
a r t i c l e i n f o
Available online 17 August 2011 Keywords:
Hearing impairment Hearing loss Birth cohort effect Education
Objective. To identify factors contributing to the declining prevalence of hearing impairment in more recent generations.
Methods. We used data on hearing thresholds and potential risk factors of hearing impairment collected from studies in Beaver Dam, Wisconsin, the Epidemiology of Hearing Loss Study (1993–1995, n=3753; 1998–2000, n=2800 and 2003–2005, n=2395), the concurrent Beaver Dam Eye Study on the same cohort, and a subgroup (n = 2173) of the Beaver Dam Offspring Study (2005–2008).
Results. Educational attainment significantly reduced the odds ratio (OR) of the birth cohort effect on hearing impairment from 0.90 to 0.93, while a history of ear infection had a reverse effect on the decreasing trend (significantly changing the OR from 0.93 to 0.94). Occupational noise exposure, smoking, and a history of cardiovascular disease, while associated with hearing impairment, did not attenuate the cohort effect. The cohort effect remained significant after known risk factors were adjusted (OR=0.93; 95% confidence interval, 0.89–0.97).
Conclusion. These data provide strong evidence that environmental, lifestyle, or other modifiable factors contribute to the etiology of hearing impairment and add support to the idea that hearing impairment in adults may be prevented or delayed.
© 2011 Elsevier Inc. All rights reserved.
Introduction
Rapid change in disease prevalence over time is a vital indicator that a disorder has environmental or other modifiable risk factors. Understanding these changes may provide important insights into ways to improve population health. Previously, Zhan et al. analyzed the trend of hearing impairment in older adults and found that people born in more recent years were less likely to have hearing impairment at a given age than those born in earlier years (Zhan et al., 2010). Over a typical generational span of 20 years, the prevalence of hearing impairment declined by 42% and 23% for men and women, respectively. This birth cohort effect suggested that environmental and modifiable factors may be associated with the development of hearing impairment because human genetic changes are extremely slow and unlikely to happen in less than 20 years.
During the 20th century there were many positive changes in the environment (e.g., cleaner air and water, institution of noise-reduction efforts in the work place) and behavioral factors (e.g., higher education, recent decreased rates of smoking), improvements in health care (e.g., immunizations and antibiotics), and declines in other disorders (e.g., cardiovascular disease (CVD), hypertension, and hypercholesterolemia), that may have beneficial effects on overall health and reduced morbidity, but obesity and sedentary lifestyles have become more common (Flegal et al., 2002; Garte, 2007; Hill and Needham, 2006; Middendorf, 2004).
Noise exposure, smoking, drinking, diabetes, CVD and its risk factors and socioeconomic factors (Agrawal et al., 2008; Cruickshanks et al., 2010; Gates et al., 2000; Helzner et al., 2005) have been associated with hearing impairment. However, which of these factors may be related to improvements in hearing health is unknown. Therefore, the purpose of this paper was to evaluate the associations of modifiable factors reported to be associated with hearing impairment to determine if these factors contributed to the observed decreasing temporal trend in a population-based cohort and a subgroup of their adult offspring.
⁎ Corresponding author at: Department of Psychology, Wesleyan University, 207 High Street, Middletown, CT, 06459-0408, USA. Fax: + 1 860 685 2761.
E-mail address:[email protected](W. Zhan).
0091-7435/$– see front matter © 2011 Elsevier Inc. All rights reserved. doi:10.1016/j.ypmed.2011.08.012
Contents lists available atSciVerse ScienceDirect
Preventive Medicine
Methods Subjects
Methods used in the Epidemiology of Hearing Loss Study (EHLS) and the Beaver Dam Offspring Study (BOSS) have been reported in detail elsewhere (Cruickshanks et al., 1998; Zhan et al., 2010). The EHLS cohort consisted of adults who participated in the population-based Beaver Dam Eye Study (BDES) and were alive as of March 1, 1993. This cohort was examined in 1993–1995 (n=3753), 1998–2000 (n=2800) and 2003–2005 (n=2395). Adult offspring of participants in the EHLS participated in the BOSS (n = 3285). This analysis excluded 1112 BOSS participants younger than 45 years because all EHLS participants were older than 45 years. A signed informed consent was obtained from all study participants. Characteristics of participants and nonparticipants have been described in the previous reports (Nondahl et al., 2006; Zhan et al., 2010). In general, eligible people who did not participate in the EHLS were older, more likely to be male, and had a lower socioeconomic status as indicated by education level than participants; eligible people who did not participate in the BOSS were slightly younger and more likely to be male than participants.
Data collection
Data were collected through the use of examinations, questionnaire interviews and laboratory tests for blood samples. The same standardized hearing examination procedures (including an otoscopic evaluation, screen-ing tympanogram, and pure-tone air- and bone-conduction audiometry) except minor modifications were applied to the three EHLS cycles and the BOSS. Pure-tone air-conduction thresholds were measured for each ear at 0.5, 1, 2, 3, 4, 6 and 8 kHz (American Speech-Language-Hearing Association (AHSA), 1987) in a sound-treated booth using clinical audiometers. Bone-conduction thresholds were measured at 0.5 and 4 kHz at the baseline EHLS, while at each follow-up and BOSS examination, bone-conduction thresholds were measured at 0.5, 2 and 4 kHz. Masking procedures were used when necessary. At each examination, hearing impairment was defined as a pure-tone average of air-conduction thresholds at 0.5, 1, 2 and 4 kHz greater than 25 dB HL (hearing level) in either ear.
The questionnaire was administrated as an interview, focusing on ear and hearing-related medical history, noise exposure (occupational noise expo-sure, military service and leisure-time noise exposure), hearing perception, socioeconomic status including educational attainment, lifestyle factors, general medical history and medication use. Some variables for the EHLS participants were provided by the concurrent BDES on the same cohort (Klein et al., 2006). Birth cohort was defined as the year of birth. Educational attainment was divided into four categories: less than high school, high school, some college, college graduate and above. History of ear infection was based on the question,“Has a doctor ever told you (or your parents) that you had an ear infection?” Smoking status was classified as non-smoker, past smoker and current smoker, and history of CVD was defined as reporting a history of angina, heart attack, or stroke.
Analysis techniques
All statistical analyses were conducted by using SAS software, version 9 (SAS Institute Inc., Cary, North Carolina). The alternating logistic regression (ALR) proposed byCarey et al. (1993)wasfirst applied to determine factors associated with hearing impairment, accounting for correlations from the repeated measurements and familial aggregations. ALR was then used to examine how each significant modifiable factor changed the birth cohort effect on hearing impairment by comparing the odds ratios (ORs) and regression coefficients for the birth cohort effect in the fully-adjusted model to those in the model with one factor removed.
Statistical significance (pb0.05) of the change of the birth cohort effect was tested using the bootstrap approach (Miller, 2004). To account for the correlation within families and within subjects, we resampled families as a whole. A total of 1000 bootstrap data sets were generated and 95% confidence interval (CI) for the change of the regression coefficient was calculated. A 95% CI excluding zero indicated a statistically significant change of the birth cohort effect.
As a sensitivity analysis to reduce heterogeneity in hearing impairment, 751 hearing impairment cases were excluded if they had self-reported onset of hearing impairment atb30 years old, a history of ear surgery, a conductive
hearing impairment without any evidence of decreased hearing sensitivity if the conductive hearing impairment was resolved, or the difference in the PTA between two ears wasN20 dB.
Results
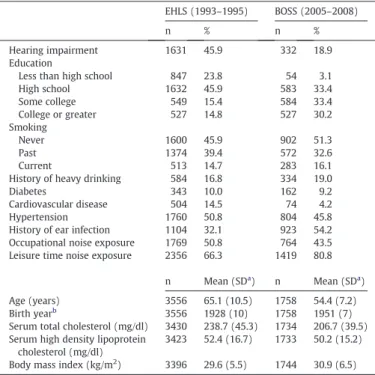
Participants included in these analyses were ages 45–100 years, with birth years between 1902–1946 (EHLS) and 1922–1962 (BOSS). Shown inTable 1are the characteristics of baseline EHLS participants and a subgroup of the BOSS participants. The overall prevalence of hearing impairment at each baseline was 45.9% (EHLS) and 18.9% (BOSS), respectively. Only 30.2% EHLS participants had received education higher than high school, while 63.6% of the BOSS subjects attended college. Occupational noise exposure was common (51% and 43.5% of EHLS and BOSS participants, respectively).
Table 2shows age-adjusted associations between potential risk factors and hearing impairment. Results from sex-specific (stratifying on sex) models were similar to those from sex-adjusted models for most factors. Higher education was significantly associated with lower odds of hearing impairment. History of heavy drinking was significantly associated with higher odds of hearing impairment. Occupational noise exposure was significant only in men while ear infection and CVD were significant only in women. Leisure time noise exposure, serum HDL cholesterol, serum total cholesterol, diabetes, hypertension and body mass index were not associated with hearing impairment.
In a stepwise analysis (model 1,Table 3), age, sex, occupational noise exposure (men only), smoking, CVD (women only) and a history of ear infection (women only) remained significantly associated with the higher prevalence of hearing impairment, and higher education was associated with lower odds of hearing impairment.
The previous study (Zhan et al., 2010) conducted in the same population observed a significant birth cohort effect (adjusting for age
Table 1
Distributions of factors and hearing impairment in the Epidemiology of Hearing Loss Study (EHLS, 1993–1995) and a subgroup of the Beaver Dam Offspring Study (BOSS, 2005–2008), Beaver Dam, Wisconsin.
EHLS (1993–1995) BOSS (2005–2008)
n % n %
Hearing impairment 1631 45.9 332 18.9
Education
Less than high school 847 23.8 54 3.1
High school 1632 45.9 583 33.4 Some college 549 15.4 584 33.4 College or greater 527 14.8 527 30.2 Smoking Never 1600 45.9 902 51.3 Past 1374 39.4 572 32.6 Current 513 14.7 283 16.1
History of heavy drinking 584 16.8 334 19.0
Diabetes 343 10.0 162 9.2
Cardiovascular disease 504 14.5 74 4.2
Hypertension 1760 50.8 804 45.8
History of ear infection 1104 32.1 923 54.2 Occupational noise exposure 1769 50.8 764 43.5 Leisure time noise exposure 2356 66.3 1419 80.8
n Mean (SDa ) n Mean (SDa ) Age (years) 3556 65.1 (10.5) 1758 54.4 (7.2) Birth yearb 3556 1928 (10) 1758 1951 (7) Serum total cholesterol (mg/dl) 3430 238.7 (45.3) 1734 206.7 (39.5) Serum high density lipoprotein
cholesterol (mg/dl)
3423 52.4 (16.7) 1733 50.2 (15.2) Body mass index (kg/m2
) 3396 29.6 (5.5) 1744 30.9 (6.5)
a
Standard deviation.
b
The birth year for EHLS and the subgroup of BOSS participants ranged from 1902– 1946 and 1922–1962, respectively.
and sex) on hearing impairment. Model 2 inTable 3demonstrated that this birth cohort effect (in the unit of 5-year increase) remained (OR: 0.93, 95% CI: 0.89, 0.97) after adjusting for significant risk factors. The effect of a single risk factor on the birth cohort effect was then examined by comparing regression coefficients and odds ratios for the birth cohort effect in the fully-adjusted model to those in the model with one factor removed (Table 4). Educational attainment signi fi-cantly attenuated the odds ratio of the birth cohort effect from 0.90 to 0.93. On the other hand, a history of ear infection significantly strengthened the odds ratio from 0.94 to 0.93. Inclusion of other modifiable risk factors in the model did not significantly change the birth cohort effect on hearing impairment.
The sensitivity analysis conducted in a subset of participants with a reduced heterogeneity in hearing impairment showed that each risk factor, except history of ear infection, had a similar effect on the birth cohort effect as that inTable 4In this subset, the birth cohort effect remained significant (pb0.0001) after controlling for covariates, and a history of ear infection did not significantly change the birth cohort effect.
Discussion
In this study, educational attainment attenuated the temporal trend of declining prevalence of hearing impairment, suggesting that increasing educational attainment over time contributed to this declining trend in more recent generations. The link between educational attainment or other indicators of socioeconomic status and hearing impairment has been observed in other populations (Davis, 1989; Helvik et al., 2009) and the influence of education on hearing impairment is likely to be complex and multi-factorial. Higher socioeconomic status has also been associated with lower risk of atherosclerosis and CVD, asthma and other chronic conditions, yet the
Table 2
Odds ratio (95% confidence interval) between potential modifiable risk factors and hearing impairment controlling for sex in the pooled sample (n=10,159 observations) from the EHLS (1993–1995, 1998–2000, 2003–2005) and BOSS (2005–2008), Beaver Dam, Wisconsin.
Risk factor Age-adjusted, sex-specific association Age- and sex-adjusted associationa
Men Women
Education (per level) 0.67 (0.62–0.74) 0.82 (0.75–0.90) Men: 0.68 (0.62–0.74); Women: 0.82 (0.75–0.90) Occupational noise exposure 1.68 (1.37–2.05) 1.12 (0.95–1.33) Men: 1.67 (1.38–2.04); Women: 1.12 (0.94–1.32)
Leisure time noise exposure 1.21 (0.98–1.49) 0.96 (0.76–1.22) 1.08 (0.93–1.26)
History of ear infection 0.95 (0.79–1.13) 1.38 (1.16–1.64) Men: 0.95 (0.79–1.13); Women: 1.39 (1.17–1.65) Smoking status
Non-smoker 1.00 1.00 1.00
Past smoker 1.31 (1.09–1.58) 1.11 (0.93–1.32) 1.19 (1.05–1.36)
Current smoker 1.70 (1.34–2.17) 1.24 (0.96–1.58) 1.44 (1.21–1.71)
History of heavy drinking 1.18 (1.00–1.39) 1.31 (1.00–1.70) 1.21 (1.05–1.39)
Diabetes 1.03 (0.84–1.26) 1.24 (1.00–1.54) 1.11 (0.96–1.29)
History of cardiovascular disease 1.14 (0.94–1.38) 1.60 (1.28–2.00) Men: 1.16 (0.96–1.39); Women: 1.57 (1.25–1.96)
Hypertension 0.95 (0.84–1.08) 0.98 (0.86–1.12) 0.97 (0.88–1.06)
Serum total cholesterol (per 10 mg/dl increase) 1.02 (1.00–1.03) 1.00 (0.99–1.02) 1.01 (1.00–1.02) Serum high density lipoprotein cholesterol (per 10 mg/dl increase) 1.00 (0.94–1.06) 0.97 (0.93–1.02) 0.99 (0.95–1.02) Body mass index (per 1 kg/m2) 1.01 (1.00–1.03) 1.00 (0.99–1.02) 1.01 (1.00–1.02) a
The interactions between sex and education, sex and occupational noise exposure, sex and history of ear infection, and sex and history of cardiovascular disease were statistically significant.
Table 3
Associations between significant modifiable risk factors and hearing impairment in the multivariate model with/without birth cohort included in the pooled sample (n = 9566 observations) from the EHLS (1993–1995, 1998–2000, 2003–2005) and BOSS (2005– 2008), Beaver Dam, Wisconsin.
OR (95% CI) in model 1a
OR (95% CI) in model 2b
Birth cohort Not included 0.93 (0.89–0.97)
Education (per level)
—Men 0.70 (0.64–0.76) 0.71 (0.65–0.78)
—Women 0.83 (0.75–0.92) 0.85 (0.77–0.95)
Occupational noise exposure
—Men 1.43 (1.16–1.77) 1.42 (1.15–1.77)
—Women 1.03 (0.86–1.23) 1.05 (0.87–1.25)
History of ear infection
—Men 1.04 (0.87–1.24) 1.07 (0.89–1.29) —Women 1.43 (1.20–1.71) 1.49 (1.24–1.79) Smoking status Non-smoker 1.00 1.00 Past smoker 1.15 (1.01–1.32) 1.16 (1.01–1.33) Current smoker 1.34 (1.13–1.60) 1.34 (1.12–1.60) History of CVD —Men 1.06 (0.87–1.28) 1.05 (0.86–1.28) —Women 1.52 (1.21–1.91) 1.51 (1.20–1.90)
Abbreviation: OR, odds ratio; CI, confidence interval.
aModel 1 included age, age2, age3, sex, education, smoking, occupational noise
exposure, history of ear infection, cardiovascular disease (CVD), interaction between sex and education, interaction between sex and occupational noise exposure, interaction between sex and history of ear infection, and interaction between sex and CVD. Regression coefficients (p values) for within subcluster (Alpha1) and between-subcluster (Alpha2) association were 4.77 (pb0.0001) and 0.14 (p=0.18), respectively.
b Model 2 included all terms in model 1, plus birth cohort, which was centered at
year 1937 in the unit of 5-year increase. Regression coefficients (p values) for within subcluster (Alpha1) and between-subcluster (Alpha2) association were 4.77 (pb0.0001) and 0.15 (p=0.18), respectively.
Table 4
Comparisons of odds ratios and regression coefficients for birth cohort (5-year interval) in the model with and without a significant risk factor in the pooled sample (n=9566 observations) from the EHLS (1993–1995, 1998–2000, 2003–2005) and BOSS (2005– 2008), Beaver Dam, Wisconsin.
Odds ratio
Regression coefficient (p-value)
Percent change of regression coefficient (95% confidence interval of the changea)
Full modelb
0.93 −0.075 (0.001) Impact of removing
one factor from the full model Education (per level) 0.90 −0.109 (b0.0001) 45.1% (24.3% to 105.3%) Occupational noise exposure 0.93 −0.076 (0.001) 0.9% (−4.1% to 6.6%) Smoking 0.93 −0.075 (0.001) 0.7% (−5.2% to 5.8%) History of ear infection 0.94 −0.063 (0.004) −16.4% (−36.2% to −3.8%) Cardiovascular disease 0.93 −0.075 (0.001) 0.5% (−7.7% to 3.5%)
a95% confidence interval was obtained according to the bootstrap approach. b The full model include cohort, age, age2, age3, sex, education, smoking,
occupational noise exposure, history of ear infection, cardiovascular disease (CVD), interaction between sex and education, interaction between sex and occupational noise exposure, interaction between sex and history of ear infection, and interaction between sex and CVD. Birth cohort was centered at year 1937 in the unit of 5-year increase.
exact mechanisms for these protective effects remain uncertain (Albert et al., 2006; Shankardass et al., 2007). Explanations for the underlying mechanisms commonly emphasize lifestyle, behavioral and psychological factors and material conditions (Madsen et al., 2010). Individuals with higher education are more likely to have a healthy lifestyle (Hill and Needham, 2006), such as less smoking and noise exposure, which may lead to a lower risk of hearing impairment. However, in the present study, these two factors were controlled for, suggesting that other mechanisms may be involved. People with more education may have better nutrition, access to health care and less workplace stress or may be more resilient to the biological effects of stress and infection (Albert et al., 2006; Cruickshanks et al., 2010). While educational attainment in older adults is difficult to modify, factors related to educational attainment may be modifiable. Thus more research is needed to identify the factors associated with education which may contribute to protective health effects.
Although occupational noise exposure, smoking and CVD were significantly, independently associated with hearing impairment, they did not change the birth cohort effect. There are several possible explanations. First, the prevalence of these factors did not change across the birth cohort, or the change may show a symmetrical curved pattern. For example, some cohorts may have an increasing prevalence of risk factors, while others may have a decreasing prevalence, with the overall impact being no change to the birth cohort effect on hearing impairment. The curve relationship between birth cohort and smoking has been observed in a study conducted among 125,707 American radiologic technologists (Freedman et al., 2002). Second, an imprecise measurement of the factors may underestimate the association between the factor and hearing impairment. For example, the use of hearing protection devices was not taken into account when defining the noise exposure. However, the use of hearing protection devices in the studied population was low (Nondahl et al., 2006) and thus is unlikely to have impacted these results. Third, since smoking and CVD are related to the mortality (Critchley and Capewell, 2003), participants may be less likely to smoke or have CVD than nonparticipants. Consequently, the associ-ation between smoking/CVD and hearing impairment may be diluted. A history of ear infection reduced the magnitude of the decreasing trend of hearing impairment, suggesting that the prevalence of ear infection was increasing. The introduction of antibiotics may have changed the types of ear disorders, with an increase in the prevalence of serous otitis media (Ruben, 2009). It has been reported that ear infections might affect hearing more before the introduction of antibiotics than during the following years (Tambs et al., 2004). An increase in less harmful infections coupled with a decrease in harmful infections may contribute to thefinding. A history of ear infection did not change the birth cohort effect when excluding those observations in which hearing impairment was not consistent with presbycusis, suggesting that the effect of ear infections on the observed birth cohort effect was more likely to be due to conductive problems. It should be noted that the increasing trend of ear infections may be artificial. For example, because of the availability of effective treatment, people whose ear infections occurred after the introduc-tion of antibiotics might be more likely to visit a doctor and recall a history of infection, particularly in case of a mild or moderate infection, than those with ear infections before the introduction of antibiotics.
Strengths and limitations
This study has numerous strengths, including but not limited to, the large population-based study design representing multiple generations of people aged 45 years and older, use of the standardized protocol for audiometric testing and covariate collection across the EHLS and the BOSS, and use of the ALR to adjust for correlations
among repeated measurements within individuals that may be further nested within families.
Nevertheless, there are some limitations to this study. First, most of the potential modifiable risk factors were self-reported which may be subject to measurement errors. Second, risk factors may have differentially affected the relationship between participation and prevalence of hearing impairment in different birth cohorts. The EHLS participants were younger and healthier than non-participants, while the BOSS participants were slightly older than non-participants. We do not know whether the BOSS participants were less healthy than non-participants. Third, the definition of hearing impairment, while often used in clinical and epidemiological studies, does not represent hearing impairment from a single etiological pathway. We chose to replicate ourfindings in a subset analysis excluding those with early onset, a history of ear surgery, a conductive hearing impairment without any evidence of decreased hearing sensitivity if the conductive hearing impairment was resolved, and asymmetric hearing impairments as an attempt to address the problem of heterogeneity. The results were consistent with the overall model suggesting that these findings reflect pathways important in sensorineural hearing impairment.
Conclusion
Educational attainment attenuated the observed birth cohort effect, while a history of ear infection partly countered the decreasing birth cohort effect on hearing impairment. Occupational noise exposure, smoking and a history of CVD were significantly associated with hearing impairment, but these factors did not change the birth cohort effect. The birth cohort effect remained significant after adjusting for known risk factors associated with hearing impairment, suggesting that other unknown factors may contribute to the decreasing trend. Our data provide strong evidence that environmen-tal, lifestyle, or other modifiable factors contribute to the etiology of hearing impairment and add support to the idea that hearing impairment in adults may be prevented or delayed.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Acknowledgments
The project described was supported by R01AG021917 (KJC) from the National Institute on Aging, National Eye Institute, and National Institute on Deafness and Other Communication Disorders. Additional support was from grants R37AG11099 (KJC) from the National Institute on Aging and U10EY06594 (RK) from the National Eye Institute. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute on Aging or the National Institutes of Health.
References
Agrawal, Y., Platz, E.A., Niparko, J.K., 2008. Prevalence of hearing loss and differences by demographic characteristics among US adults: data from the National Health and Nutrition Examination Survey, 1999–2004. Arch. Intern. Med. 168, 1522–1530. Albert, M.A., Glynn, R.J., Buring, J., Ridker, P.M., 2006. Impact of traditional and novel
risk factors on the relationship between socioeconomic status and incident cardiovascular events. Circulation 114, 2619–2626.
American Speech-Language-Hearing Association (AHSA), 1987. Guidelines for Manual Pure-Tone Threshold Audiometry, 20. ASHA.
Carey, V., Zeger, S.L., Diggle, P., 1993. Modeling multivariate binary data with alternating logistic regressions. Biometrika 80, 517–526.
Critchley, J.A., Capewell, S., 2003. Mortality risk reduction associated with smoking cessation in patients with coronary heart disease: a systematic review. JAMA 290, 86–97.
Cruickshanks, K.J., Wiley, T.L., Tweed, T.S., et al., 1998. Prevalence of hearing loss in older adults in Beaver Dam, Wisconsin. The Epidemiology of Hearing Loss Study. Am. J. Epidemiol. 148, 879–886.
Cruickshanks, K.J., Nondahl, D.M., Tweed, T.S., et al., 2010. Education, occupation, noise exposure history and the 10-yr cumulative incidence of hearing impairment in older adults. Hear. Res. 264, 3–9.
Davis, A.C., 1989. The prevalence of hearing impairment and reported hearing disability among adults in Great Britain. Int. J. Epidemiol. 18, 911–917.
Flegal, K.M., Carroll, M.D., Ogden, C.L., Johnson, C.L., 2002. Prevalence and trends in obesity among US adults, 1999–2000. JAMA 288, 1723–1727.
Freedman, D.M., Tarone, R.E., Doody, M.M., et al., 2002. Trends in reproductive, smoking, and other chronic disease risk factors by birth cohort and race in a large occupational study population. Ann. Epidemiol. 12, 363–369.
Garte, S., 2007. Where We Stand, A Surprising Look at the Real State of Our Planet, 1st ed. AMACOM, New York.
Gates, G.A., Schmid, P., Kujawa, S.G., Nam, B., D'Agostino, R., 2000. Longitudinal threshold changes in older men with audiometric notches. Hear. Res. 141, 220–228. Helvik, A.S., Krokstad, S., Tambs, K., 2009. Socioeconomic inequalities in hearing loss in a healthy population sample: the HUNT study. Am. J. Public Health 99, 1376–1378.
Helzner, E.P., Cauley, J.A., Pratt, S.R., et al., 2005. Race and sex differences in age-related hearing loss: the Health, Aging and Body Composition Study. J. Am. Geriatr. Soc. 53, 2119–2127. Hill, T.D., Needham, B.L., 2006. Gender-specific trends in educational attainment and
self-rated health, 1972–2002. Am. J. Public Health 96, 1288–1292.
Klein, R., Klein, B.E., Moss, S.E., Wong, T.Y., 2006. The relationship of retinopathy in persons without diabetes to the 15-year incidence of diabetes and hypertension: Beaver Dam Eye Study. Trans. Am. Ophthalmol. Soc. 104, 98–107.
Madsen, M., Andersen, A.M., Christensen, K., Andersen, P.K., Osler, M., 2010. Does educational status impact adult mortality in Denmark? A twin approach. Am. J. Epidemiol. 172, 225–234.
Middendorf, P.J., 2004. Surveillance of occupational noise exposures using OSHA's Integrated Management Information System. Am. J. Ind. Med. 46, 492–504. Miller, D.P., 2004. Bootstrap 101: obtain robust confidence intervals for any statistic.
SUGI 29, Statistics and Data Analysis Paper 193–29.
Nondahl, D.M., Cruickshanks, K.J., Dalton, D.S., et al., 2006. The use of hearing protection devices by older adults during recreational noise exposure. Noise Health 8, 147–153. Ruben, R.J., 2009. Serous otitis media in the 20th and 21st centuries: evolving views and
treatments. Acta Otolaryngol. 129, 343–347.
Shankardass, K., McConnell, R.S., Milam, J., et al., 2007. The association between contextual socioeconomic factors and prevalent asthma in a cohort of Southern California school children. Soc. Sci. Med. 65, 1792–1806.
Tambs, K., Hoffman, H.J., Engdahl, B., Borchgrevink, H.M., 2004. Hearing loss associated with ear infections in Nord-Trøndelag, Norway. Ear Hear. 25 (4), 388–396. Zhan, W., Cruickshanks, K.J., Klein, B.E., et al., 2010. Generational differences in the