Formulation and evaluation of water-in-oil amoxicillin-loaded nanoemulsions using for Helicobacter pylori eradication
Yu-Hsin Lin1#†, Shu-Fen Chiou2†, Chih-Ho Lai3, Shih-Chang Tsai1, Chih-Wei Chou4, Shu-Fen Peng1, Zih-Sian He1
1 Department of Biological Science and Technology, China Medical University, Taichung, Taiwan. 2 Institute of Molecular Medicine, National Tsing Hua University, Hsin Chu, Taiwan.
3 Department of Microbiology, School of Medicine, China Medical University, Taichung, Taiwan, 4 Department of Cosmeceutics, China Medical University, Taichung, Taiwan.
#Correspondence to: Yu-Hsin Lin, PhD Associate Professor
Department of Biological Science and Technology China Medical University,
Taichung, Taiwan, 40402 Fax: 886-4-2207-1507
E-mail: [email protected]
Abstract
Antibiotics have a short residence time and have low concentrations when absorbed through the basolateral membrane in the stomach; this causes a failure to enhance drug concentrations at
Helicobacter pylori infection sites. This study developed a nanocarrier system with the ability to carry amoxicillin to increase its efficacy against H. pylori. We used a water–in–oil emulsification system to prepare a positively charged nanoemulsion particle composed of amoxicillin, chitosan, and heparin. The particle size of the prepared nanoemulsion particle was controlled by the constituted compositions. The morphology of the nanoemulsion particles was spherical. In vitro analysis of amoxicillin released indicated that the nanocarrier system controlled amoxicillin release in the gastrointestinal dissolution medium and amoxicillin-loaded nanoemulsion particles localized to the site of H. pylori infection. Meanwhile, results of in vivo clearance assays indicated that the prepared amoxicillin-loaded nanoemulsion particles had a significantly greater H. pylori clearance effect in the gastric infection mouse model than the amoxicillin solution alone.
1. Introduction
Helicobacter pylori, a Gram-negative, microaerophilic spiral bacterium that colonizes the
mucosa of the human stomach, has been considered to be an important etiological factor in the development of peptic ulcer disease and gastric cancer [1–3].For effective H. pylori eradication, the standard treatment in the case is a combination of drugs, including antibiotics and a proton pump inhibitor [4]. The microorganism mainly produce virulence factors, including vacuolating cytotoxin which causes epithelium cells degradation and colonized deeply within the gastric mucus layer [5,6]. The particulate system for translocation permeability through gastrointestinal mucin was found to be increased as the particle size decreased to nanosize particles [7,8]. Encapsulation of low molecular weight hydrophilic drugs such as antibiotics in these nanocarriers failed because such small and hydrophilic molecules rapidly leaked from their, nanocarriers has resulted in poor encapsulation efficiency and fast release upon dilution [9,10]. To overcome this issue, we established the water-in-oil emulsification technique for preparing nanoemulsion particles with a better encapsulated antibiotic ability. The emulsion technique is a heterogeneous system, that consists of a water phase dispersed in an oil phase in the presence of blends of hydrophilic and hydrophobic surfactants under homogenization to produce fine droplets [11]. The efficiency of blends of hydrophilic and lipophilic surfactants between oil and water phase was primarily determined by a hydrophilic-lipophilic balance (HLB) system. A surfactant with a higher HLB value is usually more hydrophilic, and one with a lower value is more lipophilic. An optimal HLB value of the surfactants was a key factor for formation of emulsion with minimal droplets [12,13].
In our emulsion method, we selected liquid paraffin as the oil phase and two surfactants, sorbitan monolaurate (Span 20) and polyoxyethylene sorbitan monolaurate (Tween 20), to optimize surfactant levels and HLB value of the antibiotic-loaded chitosan/heparin nanoemulsion particles. Bayindir et al. previously developed niosomal formulations using nonionic surfactants (such as Span 20 and Tween 20) to achieve gastrointestinal stability for paclitaxel oral delivery [14]. It has also been reported that Span 20/Tween 20 mixtures increased the stability of the emulsification procedure compared with pure Span or Tween systems [15]. We prepared nanoemulsion particles composed of chitosan, heparin and an antibiotic (amoxicillin), with liquid paraffin and nonionic surfactant mixture (Span 20/Tween 20) as the oil phase. The molecular formula of hydrophilic amoxicillin is C16H19N3O5S-3H2O and the molecular weight (MW) is 419.45. The antibiotic amoxicillin is a semisynthetic antibiotic that binds to penicillin-binding proteins and interferes with bacterial cell wall synthesis, resulting in lysis of replicating bacteria [16,17]. It was reported that the failure of antibiotic therapies is attributable to the poor stability of the drug in the gastric acid and the poor permeation of the antibiotic across the mucus layer. This followed by resecretion into the lumen, where a sufficient amount of the drug diffuses into the bacteria [18,19].
Thus, the hypothesis of our prepare nanoemulsion particles could encapsulate amoxicillin and infiltrate into the mucus layer, subsequently, amoxicillin release from amoxicillin-loaded nanoemulsion particles, then directly acts locally on H. pylori at a bactericidal concentration [Fig.
1(A)]. We examined their physicochemical characteristics using fourier-transformed infrared spectroscopy (FT–IR), transmission electron microscopy (TEM), and dynamic light scattering. We also investigated amoxicillin release characteristics from the prepared nanoemulsion particles and examined the inhibition of H. pylori growth. In addition, the effect of the nanoemulsion particles and their mechanism of interaction with H. pylori were investigated in the human gastric mucosal AGS cell line (human gastric adenocarcinoma cell line) with confocal laser scanning microscopy (CLSM) [20]. We also assessed the in vivo clearance effect of amoxicillin-loaded chitosan/heparin nanoemulsion particles in H. pylori infected mice.
2. Materials and methods
2.1. Materials
Chitosan (MW 50 kDa) with approximately 85% deacetylation was obtained from Koyo Chemical Co. Ltd. (Japan). Heparin (5000 IU/mL, MW 15 kDa, 179 IU/mg) was purchased from Leo Chemical Factory (Ballerup, Denmark). Liquid paraffin was purchased from Wako Pure Chemical Industries, Ltd. (Osaka, Japan). Tween 20, Span 20, amoxicillin, acetic acid, 3-(4,5-dimethyl-thiazol-yl)-2,5-diphenyltetrazolium bromide (MTT), 4',6-diamidino-2-phenylindole (DAPI), fluoresceinamine isomer I (FA), phosphate buffered saline (PBS), sodium acetate, paraformaldehyde, bismuth subnitrate, Hanks’ balanced salt solution (HBSS), and β-cyclodextrin were purchased from Sigma–Aldrich (St Louis, MO). RPMI 1640, fetal bovine serum (FBS), penicillin, streptomycin, and trypsin-ethylene diamine tetraacetic acid (trypsin-EDTA) were from Gibco (Grand Island, NY). N-hydroxy-succinimide (NHS)-functionalized cyanine 3 (Cy3-NHS) was from Amersham Biosciences (Piscataway, NJ). 1,1’-dioctadecyl-3,3,3‘,3’-tetramethylindodicarbocyanine-5,5’-disulfonic acid (DilC18(5)-DS) lipophilic dye was from Molecular Probes (Eugene, USA). All other chemicals and reagents were of analytical grade.
2.2. Preparation of chitosan/heparin nanoemulsion particles by water-in-oil emulsification
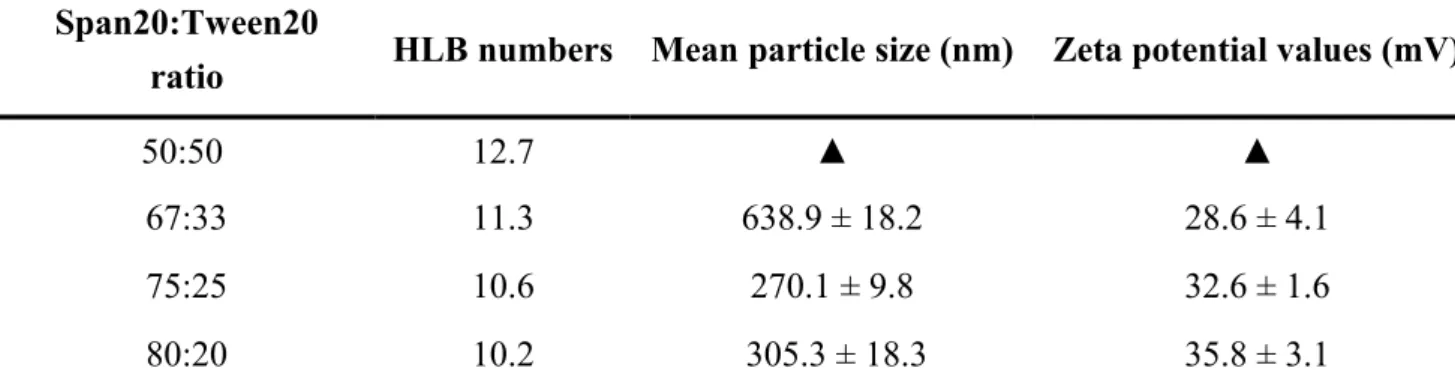
The nanoemulsion particles were prepared by water-in-oil emulsification technique through homogenization using a laboratory-type rotating blade homogenizer (IKA Labortechnik, Staufen, Germany). Table 1 and Table 2 list the conditions for preparation of the nanoemulsion particles, as well as the mean particle sizes and zeta potential values. First, the distinct Span 20:Tween 20 ratio (50:50, 67:33, 75:25, and 80:20) of the Span 20/Tween 20 mixture surfactant (0.4, 0.6, 0.8, 1.0, and 1.2 mL) was added to liquid paraffin (80.0 mL) under continuous mixing. Second, the aqueous heparin (0.2 mg/mL, 4 mL, pH 7.4) was adding to liquid paraffin containing the Span 20/Tween 20 surfactant and homogenization at 15,000 rpm at 4℃ for 2 min. Then, aqueous chitosan (0.6 mg/mL, 4 mL, pH 6.0) was slowly dropped into the resultant heparin emulsion and subject to homogenization at 15,000 rpm at 4℃ for 2 min. The prepared chitosan/heparin nanoemulsion particles were centrifuged twice at 32,000 rpm for 30 min, then the pellets were washed with distilled water and 50% aqueous alcohol to remove traces of paraffin oil and surfactant in this way.
Finally, the pellets were collected and suspended in deionized water for further study.
2.3. Characterization of the prepared nanoemulsion particles
The size distribution and zeta potential value of the nanoemulsion particles at pH 1.2 (0.1 M HCl, simulated gastric medium), pH 6.0, and pH 7.0 (10 mM PBS) simulating the gastric mucosa and the H. pylori survival situation medium were measured using a dynamic light scattering analyzer (Zetasizer ZS90, Malvern Instruments Ltd., UK) [21,22]. The peak variations of the nanoemulsion particles at different pH values were characterized using FT–IR (Shimadzu Scientific Instruments, USA). The morphology of the nanoemulsion particles was visualized under TEM at different pH values. The TEM samples were prepared as follows. The particle suspension was placed onto a 400 mesh copper grid coated with carbon. About 2 min after deposition, the grid was tapped with a filter paper to remove surface water and positively stained with an alkaline bismuth solution.
2.4. Encapsulation efficiency and release profiles of amoxicillin–loaded nanoemulsion particles
To study the release profiles of amoxicillin from amoxicillin-loaded chitosan/heparin nanoemulsion particles, the amoxicillin–loaded nanoemulsion particles were prepared. In brief, amoxicillin (0.2 mg/mL, 0.4 mg/mL, and 0.6 mg/mL, 2 mL, pH 7.4) was mixed with aqueous heparin (0.4 mg/mL, 2 mL, pH 7.4) under continuous stirring for 12 hr at 4℃ and then added to liquid paraffin containing the Span 20/Tween 20 mixture surfactant with homogenization, as described above. Then, the aqueous chitosan (0.6 mg/mL, 4 mL, pH 6.0) was slowly dropped into the resultant heparin/amoxicillin emulsion during homogenization at 15,000 rpm at 4℃ for 2 min. Finally, the amoxicillin-loaded chitosan/heparin nanoemulsion particles were obtained after centrifugation, washed, and then suspended in deionized water. To determine the loading efficiency and loading content, the amoxicillin concentration was assayed by high–performance liquid chromatography (HPLC). The release profiles of amoxicillin from test particles were investigated in simulated dissolution medium (pH 1.2 for 120 min, pH 6.0 for 120 min, and pH 7.0 for 360 min) at 37℃. At set time intervals, samples were removed and centrifuged, and the supernatants were subjected to HPLC. The percentage of cumulative amount of released amoxicillin was determined using a standard calibration curve.
2.5. Viability of AGS cells treated with chitosan/heparin nanoemulsion particles
The AGS cell line (ATCC CRL 1739) was obtained from the American Type Culture Collection (ATCC) and used between passages 40 and 60. The AGS cells were cultured in RPMI 1640 medium containing 10% FBS, penicillin (100 IU/mL), and streptomycin (100 mg/mL), and were kept in an incubator at 37℃, 95% humidity, and 5% CO2 [23]. The cells were harvested for subculture every 3 days with 0.25% trypsin plus 0.05% EDTA solution and were used for the cytotoxicity experiments. The cytotoxicity of the test samples was evaluated in vitro using the MTT
assay. AGS cells were seeded at 5×104 cells/well in 96-well plates and allowed to adhere overnight. The growth medium was replaced with HBSS solution (pH 6.0) that contained various concentrations (0.02-0.20 mg/mL) of chitosan/heparin nanoemulsion particles and incubated for 2 hr. After 2 hr, the test samples were aspirated and the cells were washed twice with 100 µL of PBS. The cells were then incubated in growth medium for an additional 22 hr. The cells were then incubated in growth medium containing 1 mg/mL MTT for an additional 4 hr. Dimethyl sulfoxide (100 µL) was added to each well to ensure solubilization of the formazan crystals. The optical density was read with a Molecular Devices SpectraMax M2e microplate spectrofluorometer
(Sunnyvale, CA) at a wavelength of 570 nm. All experiments were performed six times with eight replicate wells for every sample and control per assay.
2.6. In vitro cellular uptake and CLSM visualization
To track the cellular particles, fluorescent Cy3-chitosan and FA-heparin were prepared according to the procedure described in our previous study [24]. The fluorescent Cy3-chitosan/FA-heparin nanoemulsion particles were prepared according to the procedure described in Section 2.2. The AGS cells were seeded onto 6-cm petri dishes at a density of 5 × 105 cells/cm2 and incubated for 2 days. After incubation, the medium was removed and cells were treated with HBSS solution (pH 6.0) containing Cy3-chitosan/FA-heparin nanoemulsion particles at a concentration of 0.1 mg/mL. After 2 hr, the test samples were aspirated and the cells were then washed three times with PBS before being fixed in 3.7% paraformaldehyde. The cells were washed three times with PBS and stained with DAPI, which specifically bind to the nuclei. The stained cells were examined with excitation at 340, 488, and 543 nm using a CLSM. The images were superimposed using LCS Lite software (version 2.0).
2.7. Evaluating the relationship between H. pylori and amoxicillin-loaded nanoemulsion particles in co-culture with AGS cells
To observe the adhesion of H. pylori to cells, the fluorescent bacteria were labeled with DilC18(5)-DS fluorescently labeled lipophilic dye according to the procedure described in our previous study [25]. Additionally, synthesis of FA-amoxicillin was based on the reaction between the amine group of FA and the carboxylic acid group of amoxicillin. Amoxicillin (60 mg) was dissolved completely in 30 mL of deionized water and 2 mg of FA was dissolved completely in 1 mL of acetonitrile. The FA solution was added gradually to the amoxicillin solution, then 1 mg of 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride was added with continuous stirring at room temperature for 120 min and then freeze dried. To remove the unconjugated FA, the dry FA-amoxicillin sample was precipitated by adding acetonitrile (30 mL), and the precipitate was subjected to repeated cycles of washing and centrifugation until no fluorescence was detected in the supernatant. The resultant FA-amoxicillin was dissolved in deionized water then freeze dried.
To examine the effects on H. pylori of amoxicillin–loaded nanoemulsion particles in co-culture with AGS cells, the AGS cells were seeded on Costar Transwell six-well plates (Corning Costar Corp., NY) at a seeding density of 5 ×105 cells/insert. The AGS cell culture medium was added to both the donor and acceptor compartments. The medium was replaced every 48 hr for the first 6 days and every 24 hr thereafter. The cultures were kept in an incubator and were used for the
H. pylori infection experiments 26–30 days after seeding [26,27]. DiIC18(5)-H. pylori was
incubated with the AGS cells for 2 hr. The medium (pH 6.0) containing the FA-amoxicillin–loaded Cy3-chitosan/heparin nanoemulsion particles or fluorescent FA-amoxicillin solution were then introduced into the donor compartment of the AGS cells for 2 hr at 37℃. After incubation, the test samples were aspirated. The cells were washed twice with PBS and stained with DAPI for 15 min. The stained cells were examined by CLSM with excitation at 340, 488, 543, and 633 nm at 0.2 μm intervals, and 3D images were created using LCS Lite software.
2.8. In vitro and in vivo H. pylori growth inhibition study
We quantified the inhibitory effects of amoxicillin-loaded nanoemulsion particles on growth of H. pylori strains. Briefly, the AGS cells were seeded in 24-well plates at 1×105 cells/well in 500 μL of medium and incubated overnight at 37℃. The following day, H. pylori at a final concentration of 8×108 CFU/mL (colony-forming unit/mL) were added to the AGS cells in the well.
After 2 hr, the medium was removed and cells were treated with HBSS solution (pH 6.0) containing amoxicillin solution and amoxicillin-loaded chitosan/heparin nanoemulsion particles with varying amoxicillin concentrations (0.0 mg/L, 2.0 mg/L, 4.0 mg/L, 8.0 mg/L, 16.0 mg/L) for 2 hr. After 2 hr, the treated AGS cells were washed and incubated for an additional 22 hr. The inhibition of AGS cell growth by H. pylori was analyzed using the trypan blue exclusion [28].
An H. pylori infectious animal model was established according to Qian's method (China Patent, CN 1304729A) with some modifications to determine the ability of amoxicillin or amoxicillin–loaded nanoemulsion particles to clear H. pylori in vivo. Healthy and disease-free 6– week–old male C57BL/6J mice were used for the study. After an overnight fasting, mice were inoculated with an equal amount of bacterial suspension (0.5 mL) by intragastric gavage using a sterile oral feeding needle with each dose containing approximately 1×109 CFU/mL of H. pylori.
The dosing was repeated once daily for 7 consecutive days. After the development of infection after 1 week, the mice were randomly divided into different groups. Each group contained six mice and received different amoxicillin formulations (15 mg/kg in the form of amoxicillin solution or amoxicillin–loaded nanoemulsion particles, and deionized water as a control) once daily for 7 consecutive days. One day after administration of the final dose, the mice were sacrificed and their stomachs removed and subjected to the following tests. Each stomach was homogenized with sterile normal saline (3 mL/stomach) from which serial dilutions were plated on blood agar plates under micro-aerophilic conditions for 5 days at 37°C. The viable bacterial count for each gastric wall was calculated by counting the number of colonies on the agar plates.
2.9. Statistical analysis
Statistical analysis of the differences in the measured properties of the groups was performed with one-way analysis of variance and the determination of confidence intervals, with the statistical package Statistical Analysis System, version 6.08 (SAS Institute Inc., Cary, NC). All data are presented as means and standard deviations, indicated as “mean ± SD”. Differences were considered to be statistically significant when the P values were less than 0.05.
3. Results
3.1. Preparation of nanoemulsion particles
The Chitosan/heparin nanoemulsion particles were prepared by a water–in–oil emulsification method using a mixture of Span 20 and Tween 20 nonionic surfactants in liquid paraffin. We prepared emulsions with various Span 20:Tween 20 ratios, namely: 50:50, 67:33, 75:25, and 80:20 to determine the resulting HLB values. The 75:25 (w/w) Span 20:Tween 20 ratio produced small particle sizes (270.1 ± 9.8 nm), with a small polydispersity index (PDI) 0.19 ± 0.06 and constant HLB number of 10.6; this ratio was used throughout the remainder of the study (Table 1). We investigated the effect of varying the concentration of the Span20-Tween20 surfactant mixture (0.50% wt.%, 0.75% wt.%, 1.00% wt.%, 1.25% wt.%, and 1.50% wt.%) on the formation of chitosan/heparin nanoemulsion particles. As shown in Table 2, as the amount of Span20/Tween20 mixture surfactant incorporated in emulsification phase was increased, the size of the resulting particles decreased appreciably and to give a uniform matrix with a spherical shape [Fig. 1(B)]. From these results, optimal conditions for preparing a fine chitosan/heparin nanoemulsion particles by water–in–oil emulsification were: an aqueous phase (4 mL) containing chitosan (0.6 mg/mL) and heparin (0.2 mg/mL), and an oil phase of paraffin oil (80 mL) containing 1.2 mL of a Span20:Tween20 in a 75:25 ratio. Nanoemulsion particles prepared to this specific composition were used for the remainder of the study.
3.2. Characterization of the prepared nanoemulsion particles at specific pH values
The morphologies of the prepared chitosan/heparin nanoemulsion particles at various pH values were examined by TEM, and pH related chemical changes were followed by FT–IR [Fig. 2]. At pH 6.0 (simulating the environment of the gastric mucosa, and ideal conditions for H. pylori survival situation), the characteristic peaks observed at 1532 cm-1 and 1633 cm-1 were the protonated amino group (–NH3+) on chitosan and the carboxylic ions (–COO–) on heparin, respectively [Fig. 2(A)]. Thus ionized chitosan and heparin formed a polyelectrolyte complex with a spherical matrix structure [Fig. 2(B)]. At pH 1.2 (simulating the pH of gastric acid), the characteristic peak observed at 1239 cm-1 was attributed to the heparin sulfate ions (–SO4–), but some of the –COO– groups on heparin also became protonated (–COOH, 1736 cm-1). Electrostatic interactions between chitosan and heparin were therefore weaker at pH 1.2 than they were at pH
6.0, leading to greater particle sizes (289.1 ± 5.6 nm). At pH 7.0, chitosan ammonium groups were deprotonated [indicated by disappearance of the characteristic peak for –NH3+ on chitosan; Fig. 2(A)], leading to the collapse of the nanoparticles [Fig. 2(B)].
3.3. Encapsulation efficiency and release profile of amoxicillin-loaded nanoemulsion particles
We used chitosan/heparin nanoemulsion particles to encapsulate amoxicillin at various concentrations. It was shown that for an amoxicillin concentration of 0.6 mg/mL, particle size is 296.5 ± 6.3 nm and zeta potential is 29.8 ± 3.1 mV, while the amoxicillin loading efficiency and percentage loading content are 54.3 ± 2.8% and 19.2 ± 1.2%, respectively. Under optimal conditions, these amoxicillin-loaded nanoemulsion particles remained spherical [Fig. 2(B)]. We used the optimal amoxicillin concentration of 0.6 mg/mL for subsequent in vitro studies. Fig. 2(C) shows the in vitro release of amoxicillin in solutions buffered pH 1.2, 6.0, and 7.0. At pH 1.2, the nanoemulsions released 20.5 ± 1.2% amoxicillin over 120 min. At pH 6.0, the structure and morphology of nanoparticle emulsion was stable but there was only a modest release of amoxicillin from amoxicillin-loaded nanoemulsion particles. By contrast, at pH 7.0, the prepared emulsion particles became unstable and collapsed, causing rapid release of the amoxicillin. These findings suggest that chitosan/heparin nanoemulsion particles have potential for sustained drug release.
3.4. Viability and cellular uptake of cells treated with chitosan/heparin nanoemulsion particles
We evaluated the cytotoxicity of various concentrations of chitosan/heparin nanoemulsion particles using AGS cells and MTT assay [Fig. 3(A)]. Cell viability was generally unaffected at concentrations less than 0.14 mg/mL. Thereafter, we used emulsion particles at 0.1 mg/mL to track cellular internalization of particles without causing damage to cultured cells. We used fluorescent nanoemulsion particles [Cy3-chitosan (red)/FA-heparin (green) nanoemulsion particles] to demonstrate cellular uptake. Fig. 3(B) shows the fluorescence intensity of those AGS cells that internalized fluorescent particles. Fluorescent signals appeared in the intercellular spaces and cell cytoplasm [superimposed red (Cy3-chitosan)/green (FA-heparin) spots; i.e., yellow spots, white arrows; Fig. 3(B)] after 2 hr incubation with nanoemulsion particles at pH 6.0.
3.5. Relationship between H. pylori and amoxicillin–loaded nanoemulsion particles
To study the interaction of amoxicillin–loaded nanoemulsion particles with H. pylori attached to the epithelia, we developed a process whereby fluorescent DiIC18 (5)-H. pylori infected monolayers of AGS cells, and the infected cells were then treated with fluorescent FA-amoxicillin alone, or FA-amoxicillin loaded in Cy3-chitosan/heparin nanoemulsion particles. As shown in Fig. 4, the fluorescent images (those after 3D reconstruction) of AGS cell monolayers were observed (the XY plane) and appeared at a deep level (the XZ plane). The AGS–adapted DiIC18 (5)-labeled
H. pylori (purple spot) preferentially targeted cell-cell junctions and was present within the cells.
Cy3-chitosan/heparin nanoemulsion particles (Cy3-chitosan: red spot, FA-amoxicillin: green spot) and observed by CLSM to ascertain whether the nanoparticles and drug were attached in the same way to the intercellular spaces and the cell cytoplasm. The superimposed images (3D reconstruction of the XY and XZ plane), show that administered amoxicillin loaded chitosan/heparin nanoemulsion particles co-localized and interacted at the same location of intercellular spaces and the cell cytoplasm of H. pylori infectious sites (superimposed red spot: Cy3-chitosan, green spot: FA-amoxicillin, and purple spot: DiIC18 (5)-H. pylori appearing as white spots, green arrows; Fig. 4). By contrast, cells incubated with FA-amoxicillin solution alone (green spot), showed less obvious fluorescent signals in intercellular spaces than those seen after incubation with fluorescent FA-amoxicillin loaded in Cy3-chitosan/heparin nanoemulsion particles. The amoxicillin-loaded particles produced an intense fluorescence that emanated from deep within the cells, indicating that the nanoemulsion particles were capable of carrying amoxicillin to the H. pylori infection site. This result suggests that our nanoemulsion particles prepared had a specific interaction with AGS cell monolayers infected with
H. pylori.
3.6. H. pylori growth inhibition study
Fig. 5 shows the influence on gastric mucosal cell viability of H. pylori infection alone and after pretreatment with amoxicillin-loaded chitosan/heparin nanoemulsion particles. The figure shows a significant reduction in AGS cell viability after infection with H. pylori (8 108 CFU/mL) alone; cell viability was 47.6 ± 2.9% that of uninfected controls. We determined the percentage of H. pylori-infected AGS cells after 24 hr treatment using various amoxicillin concentrations (0.0, 2.0, 4.0, 8.0, and 16.0 by mg/L). Cell survival rates seen with amoxicillin solution alone was 52.3 ± 2.4% (amoxicillin concentration of 2.0 mg/L), 57.1 ± 2.2% (4.0 mg/mL), 63.9 ± 3.9% (8.0 mg/mL), and 70.4 ± 3.5% (16.0 mg/L). Meanwhile, the cell survival rates with amoxicillin-loaded chitosan/heparin nanoemulsion particles were 65.1 ± 2.9% (at amoxicillin concentration of 2.0 mg/L), 72.6 ± 3.2% (4.0 mg/mL), 81.4 ± 4.3% (8.0 mg/mL), and 90.5 ± 3.8% (16.0 mg/mL), compared with control (without sample). Therefore, our prepared amoxicillin-loaded chitosan/heparin nanoemulsion particles with a positive surface charge significantly increased the inhibitory effects on H. pylori infected cells compared with amoxicillin solution alone (p < 0.05).
Fig. 6 shows the in vivo clearance data of H. pylori infection after administration of 15 mg/kg amoxicillin alone or amoxicillin-loaded chitosan/heparin nanoemulsion particles for 7 consecutive days. The mean bacterial count of the control group of mice that were given only sterile water was 137.3 ± 15.4 (CFU/stomach). Treatment with 15 mg/kg amoxicillin solution alone gave a mean bacterial count of 45.2 ± 8.7 (CFU/stomach). Meanwhile, treatment with 15 mg/kg amoxicillin in amoxicillin-loaded chitosan/heparin nanoemulsion particles gave a mean bacterial count of 11.3 ± 2.8 (CFU/stomach) with significantly increased the inhibitory effects on H. pylori infected mice compared with amoxicillin solution alone. We used a rapid urease test, also known as the
Campylobacter-like organism test (CLO test), which was developed by Marahall and specifically designed to detect H. pylori. In the presence of H. pylori urease, urea is converted into ammonium hydroxide and is changed the color of the indicator from yellow to red. Fig. 6 shows the mice infected with H. pylori and administered sterile water indicate a positive response as the media changed color from yellow (healthy mice) to red (infected mice). Following treatment with 15 mg/kg amoxicillin the form of amoxicillin solution alone and amoxicillin-loaded chitosan/heparin nanoemulsion particles, the media changed color to a reddish-orange and light orange, respectively. Therefore, we concluded that prepared positive surface charge amoxicillin nanoemulsion particles showed a more complete H. pylori clearance effect than amoxicillin alone.
4. Discussion
Chitosan-based nanocarriers produced by gelation are efficient drug delivery systems for hydrophilic substances such as insulin and nucleic acids [29,30]. However, the gelation approach to preparing nanocarriers to encapsulate hydrophilic amoxicillin introduces problems associated with drug loading efficiency and drug content. We investigated use of water-in-oil emulsification with various surfactants, in particular, Span 20 and Tween 20 at various ratios for the preparation of chitosan/heparin nanoemulsion particles to encapsulate amoxicillin, so improving drug entrapment efficiency and providing a slow drug release rate. Nanoemulsion particle sizes are affected by interactions between surfactants that arise from surfactant mixtures with opposing HLB geometries [14,31]. Tween 20 is a hydrophilic surfactant with an HLB of 16.7, while Span 20 is a hydrophobic surfactant with an HLB of 8.6 [32,33]. Additionally, the mixed surfactant HLBmix value could be calculated as follows: HLBmix = HLBT × T% + HLBS × S%, where HLBT and HLBS are the HLB values of Tween 20 (16.7) and Span 20 (8.6) respectively, T% and S% are the respective weight percentages of Tween 20 and Span 20 present in the surfactant mixtures [34]. We found that a ratio of 75:25 (w/w) provided an HLB value of 10.6, and gave a particle size of 270.1 ± 9.8 nm, with positive zeta potential, 32.6 ± 1.6 mV. Surfactant concentrations in the range 0.75-1.50 by wt.% formed emulsion particles in the size range of 270-930 nm and the mean sizes of the prepared nanoemulsion particles decreased with increasing surfactant concentration (Table 2). Izquierdo et
al. performed similar studies with nanoemulsions and found that the observed decrease in droplet
size with increasing surfactant concentration was due to an increase in the interfacial area and decrease in interfacial tension [35].
H. pylori produce the enzyme urease, which hydrolyzes urea to ammonia and carbon dioxide;
these products in turn neutralize stomach acid and raise pH to allow successful colonization of the gastric environment by H. pylori [36]. Chitosan is a weak base, and the amino group on chitosan has a pKa value of about 6.5. Heparin is a polydispersed, highly sulfated polysaccharide, and the sulfate monoesters and sulfamido groups have pKa values ranging from 0.5 to 1.5, whereas the carboxylate group has a pKa value between 2 to 4. Chitosan and heparin ionize in the pH range 1.2-6.0, and may form polyelectrolyte complexes with a spherical matrix structure. When this matrix
encapsulates amoxicillin, the amoxicillin–loaded nanoemulsion particles release only a small quantity of the drug. At pH 7.0, the nanoemulsion particles became unstable and broke apart, allowing rapid release of the amoxicillin [Fig. 2(B) and Fig. 2(C)]. This occurs because at pH 7.0, chitosan is deprotonated, causing collapse of the nanoemulsion particles [Fig. 2(A)]. Thus, nanoparticles with pH-responsive characteristics that are stable at pH 1.2-6.0 can protect drugs from gastric acid and control amoxicillin release into an H. pylori infection environment.
H. pylori infection is considered a primary risk factor for the development of gastric ulcer and
gastric cancer [37]. When H. pylori colonizes the human gastric mucus layer and adheres to surface epithelial cells, it produces a vacuolating cytotoxin with the ability to modulate the integrity of the epithelium by increasing tight junction permeability to small ions and molecules. H. pylori may preferentially adhere not only to the gastric epithelium surface, but also close to the tight junctions of gastric mucous cells [38,39]. Therefore, the ideal antibiotic dose for H. pylori eradication must not only localize in the stomach, but must also interact locally with the bacterium. In our study of the relationship between amoxicillin-loaded chitosan/heparin nanoemulsion particles and H. pylori (Fig. 4), we used CLSM to show that prepared fluorescent FA-amoxicillin loaded in Cy3-chitosan/heparin nanoemulsion particles (Cy3-chitosan: red spot, FA-amoxicillin: green spot) co-localized and interacted locally at sites of H. pylori infection.
Amoxicillin is a semisynthetic, orally absorbed, broad-spectrum antibiotic, which has been extensively used to provide resistance against H. pylori cultures [40,41]. However, amoxicillin treatment results in incomplete eradication of H. pylori, likely due to the short residence time of the drug in the stomach which prevents effective antimicrobial concentrations being achieved in the gastric mucous layer or epithelial cell surfaces where H. pylori exists [42,43]. To improve the efficacy of anti-H. pylori agents, antibiotics need to localize at the H. pylori infection site in the gastric epithelium. Chitosan, a polycationic, nontoxic, and mucoadhesive polymer is safe, allows prolonged interaction between the delivered drug and the membrane epithelia, and facilitates efficient drug diffusion into the mucus and epithelium layer [44,45]. Fig. 5 shows the amoxicillin– loaded chitosan/heparin nanoemulsion particles with various amoxicillin concentrations significantly increased AGS cell growth compared with amoxicillin solution alone (p < 0.05). Furthermore, clearance of H. pylori in vivo was observed with doses of 15 mg/kg amoxicillin in amoxicillin–loaded chitosan/heparin nanoemulsion particles. The clearance rate showed a more complete H. pylori clearance effect than with amoxicillin alone (Fig. 6).
5. Conclusions
We prepared chitosan/heparin nanoemulsion particles using a water–in–oil emulsification system for delivery of amoxicillin to treat H. pylori infection. The nanoemulsion particles were stable at pH 1.2. In vitro drug release analysis of the nanoemulsion particles indicated that the system could control amoxicillin release in a simulated gastrointestinal dissolution medium. The amoxicillin-loaded chitosan/heparin nanoemulsion particles could localize at intercellular spaces or
in the cell cytoplasm, the site of H. pylori infection, and significantly increased H. pylori growth inhibition compared with amoxicillin alone. Results of in vivo clearance assays indicated that amoxicillin-loaded chitosan/heparin nanoemulsion particles had a more complete H. pylori clearance effect on induced gastric H. pylori infection mice compared with amoxicillin alone.
Acknowledgements
This work was supported by grants from the National Science Council (NSC100-2628-E-039-003-MY3) and the China Medical University (CMU-100-S-23). The Leica TSC SP2 confocal Spectral microscopy experiment and Malvern ZS90 Zetasizer Nano apparatus supported by the Medical Research Core Facilities center, Office of Research & Development, China Medical University were gratefully acknowledged.
References
[1] Vattem DA, Lin YT, Ghaedian R, Shetty K. Cranberry synergies for dietary management of Helicobacter pylori infections. Process Biochem 2005;46:1583-92.
[2] Peterson WL. Helicobacter pylori and peptic ulcer disease. N Engl J Med 1991;324:1043-8. [3] Hejazi R, Amiji M. Stomach-specific anti-H. pylori therapy. I: preparation and characterization
of tetracyline-loaded chitosan microspheres. Int J Pharm 2002;235:87-94.
[4] van der Hulst RW, Keller JJ, Rauws EA, Tytgat GN. Treatment of Helicobacter pylori infection: a review of the world literature. Helicobacter 1996;1:6-19.
[5] van Amsterdam K, van Vliet AH, Kusters JG, van der Ende A. Of microbe and man: determinants of Helicobacter pylori-related diseases. FEMS Microbiol Rev 2006;30:131-56. [6] Cover TL, Blaser MJ. Purification and characterization of the vacuolating toxin from
Helicobacter pylori. J Biol Chem 1992;267:10570-5.
[7] Khanvilkar K, Donovan MD, Flanagan DR. Drug transfer through mucus. Adv Drug Deliv Rev 2001;48:173-93.
[8] Gonçalves C, Pereira P, Gama M. Self-assembled hydrogel nanoparticles for drug delivery applications. Materials 2010;3:1420-60.
[9] Hillaireau H, Le Doan T, Appel M, Couvreur P. Hybrid polymer nanocapsules enhance in vitro delivery of azidothymidine-triphosphate to macrophages. J Control Release 2006;116:346-52. [10] Hillaireau H, Le Doan T, Besnard M, Chacun H, Janin J, Couvreur P. Encapsulation of
antiviral nucleotide analogues azidothymidine-triphosphate and cidofovir in poly(iso-butylcyanoacrylate) nanocapsules. Int J Pharm 2006;324:37-42.
[11] Motohiro S, Yohei K, Yukitaka K, Shuji A, Ryuichi M. Effect of the hydrophilic surfactants on the preparation and encapsulation efficiency in course and fine W/O/W type emulsions. Colloids Surf A Physicochem Eng Asp 2004;238:83-90.
[12] Jina X, Ugine TA, Chena J, Streetta AD. Method for determining the best hydrophilic-lipophilic balance (HLB) number for a compatible non-ionic surfactant in formulation development for aerial conidia of metarhizium anisopliae (Hypocreales: Clavicipitaceae). Biocontrol Sci Technol 2009;19:341-7.
[13] Boyd J, Parkinson C, Sherman P. Factors affecting emulsion stability and the HLB concept. J Colloid Interface Sci 1972;41:359-70.
[14] Bayindir ZS, Yuksel N. Characterization of niosomes prepared with various nonionic surfactants for paclitaxel oral delivery. J Pharm Sci 2010;99:2049-60.
[15] Sajjadi S, Zerfa M, Brooks BW. Phase inversion in p-xylene/water emulsions with the non-ionic surfactant pair sorbitan monolaurate/polyoxyethylene sorbitan monolaurate (Span 20/Tween 20). Colloids Surf A Physicochem Eng Asp 2003;218:241-54.
[16] Shah S, Qaqish R, Patel V, Amiji M. Evaluation of the factors influencing stomach-specific delivery of antibacterial agents for Helicobacter pylori infection. J Pharm Pharmacol 1999;51: 667-72.
[17] Kim JM, Kim JS, Jung HC, Kim N, Kim YJ, Song IS. Distribution of antibiotic MICs for Helicobacter pylori strains over a 16-year period in patients from Seoul, South Korea. Antimicrob Agents Chemother 2004;48:4843-7.
[18] Endo H , Yoshida H, Ohmi N, Ohta K, Higuchi S. Localization of [14C]amoxicillin in rat gastric tissue when administered with lansoprazole and clarithromycin. J Antimicrob Chemother 2001;48:923–26.
[19] Endo H, Yoshida H, Ohmi N, Ohta K, Higuchi S, Suga T. Localization of [14C]clarithromycin in rat gastric tissue when administered with lansoprazole and amoxicillin. J Antimicrob Chemother 2002;50:285–88.
[20] Hall AJ, Tripp M, Howell T, Darland G, Bland JS, Babish JG. Gastric mucosal cell model for estimating relative gastrointestinal toxicity of non-steroidal anti-inflammatory drugs. Prostaglandins Leukot Essent Fatty Acids 2006;75:9-17.
[21] Li Y, Wang WP, Wang HY, Cho CH. Intragastric administration of heparin enhances gastric ulcer healing through a nitric oxide-dependent mechanism in rats. Eur J Pharmacol 2000;399:205-14.
[22] Shu XZ, Zhu KJ, Song W. Novel pH-sensitive citrate cross-linked chitosan film for drug controlled release. Int J Pharm 2001;212:19-28.
[23] Sheng H, Shah PK, Audus KL. Sucralfate effects on mucus synthesis and secretion by human gastric epithelium in vitro. Int J Pharm 1996;131:159-69.
[24] Lin YH, Chang CH, Wu YS, Hsu YM, Chiu SF, Chen YJ. Development of pH-responsive chitosan/heparin nanoparticles for stomach-specific anti-Helicobacter pylori therapy. Biomaterials 2009;30:3332-42.
[25] Chang CH, Huang WY, Lai CH, Hsu YM, Yao YH, Chen TY, Wu JY, Peng SF, Lin YH. Development of novel nanoparticles shelled with heparin for berberine delivery to treat Helicobacter pylori. Acta Biomater 2011;7:593-603.
[26] Kim JS, Kim JM, Jung HC, Song IS. Expression of cyclooxygenase-2 in human neutrophils activated by Helicobacter pylori water-soluble proteins: possible involvement of NF-kappaB and MAP kinase signaling pathway. Dig Dis Sci 2001;46:2277-84.
[27] Hennig EE, Godlewski MM, Butruk E, Ostrowski J. Helicobacter pylori VacA cytotoxin interacts with fibronectin and alters HeLa cell adhesion and cytoskeletal organization in vitro. FEMS Immunol Med Microbiol 2005;44:143-50.
[28] Sieveking D, Leach ST, Mitchell HM, Day AS. Role of serum factors in epithelial cell responses to Helicobacter pylori infection in vitro. J Gastroenterol Hepatol 2005;20:1610-5. [29] Ma Z, Lim TM, Lim LY. Pharmacological activity of peroral chitosan-insulin nanoparticles in
diabetic rats. Int J Pharm 2005;293:271-80.
[30] Liu W, Sun S, Cao Z, Zhang X, Yao K, Lu WW, Luk KD. An investigation on the physicochemical properties of chitosan/DNA polyelectrolyte complexes. Biomaterials 2005;26:2705-11.
[31] Leong TS, Wooster TJ, Kentish SE, Ashokkumar M. Minimising oil droplet size using ultrasonic emulsification. Ultrason Sonochem 2009;16:721-7.
[32] Tsai HA, Huang DH, Ruaan RC, Lai JY. Mechanical properties of asymmetric polysulfone membranes containing surfactant as additives. Ind Eng Chem Res 2001;40:5917-22.
[33] Barakat NS, Yassin AE. In vitro characterization of carbamazepine-loaded precifac lipospheres. Drug Deliv 2006;13:95-104.
[34] Kunieda H, Shinoda K. Evaluation of the hydrophile-lipophile balance (HLB) of nonionic surfactants. I. Multisurfactant systems. J Colloid Interface Sci 1985;107:107-21.
[35] Izquierdo P, Esquena J, Tadros ThF, Dederen C, Garcia MJ, Azemar N, Solans C. Formation and stability of nano-emulsions prepared using the phase inversion temperature method. Langmuir 2002;18:26-30.
[36] Scott DR, Weeks D, Hong C, Postius S, Melchers K, Sachs G. The role of internal urease in acid resistance of Helicobacter pylori. Gastroenterology 1998;114:58-70.
[37] Beil W, Kilian P. EPs 7630, an extract from Pelargonium sidoides roots inhibits adherence of
Helicobacter pylori to gastric epithelial cells. Phytomedicine 2007;14:5-8.
[38] Bardonnet PL, Faivre V, Pugh WJ, Piffaretti JC, Falson F. Gastroretentive dosage forms: overview and special case of Helicobacter pylori. J Control Release 2006;111:1-18.
[39] Pelicic V, Reyrat JM, Sartori L, Pagliaccia C, Rappuoli R, Telford JL, Montecucco C, Papini E. Helicobacter pylori VacA cytotoxin associated with the bacteria increases epithelial permeability independently of its vacuolating activity. Microbiology 1999;145:2043-50.
[40] Patel JK, Patel MM. Stomach specific anti-Helicobacter pylori therapy: preparation and evaluation of amoxicillin-loaded chitosan mucoadhesive microspheres. Curr Drug Deliv 2007;4: 41-50.
[41] Lai CH, Kuo CH, Chen PY, Poon SK, Chang CS, Wang WC. Association of antibiotic resistance and higher internalization activity in resistant Helicobacter pylori isolates. J Antimicrob Chemother 2006;57:466-71.
[42] Cooreman MP, Krausgrill P, Hengels KJ. Local gastric and serum amoxicillin concentrations after different oral application forms. Antimicrob Agents Chemother 1993;37:1506-9.
[43] Atherton JC, Cockayne A, Balsitis M, Kirk GE, Hawkey CJ, Spiller RC. Detection of the intragastric sites at which Helicobacter pylori evades treatment with amoxicillin and cimetidine. Gut 1995;36:670-4.
[44] Thanou M, Verhoef JC, Junginger HE. Chitosan and its derivatives as intestinal absorption enhancers. Adv Drug Deliv Rev 2001;50:S91-101.
[45] Ko JA, Lim HJ, Park HJ. Effect of microencapsulated precipitants of Lactobacillus casei ATCC 393 on Helicobacter pylori eradication . Process Biochem 2011;46:631-5.
Tables
Table 1. Mean particle sizes and zeta potential values for chitosan/heparin nanoemulsion particles with different ratios of Span 20/Tween 20 mixture surfactant (1.50 wt%) and HLB values in water– in–oil emulsification system (n = 5). HLB: hydrophilic-lipophilic balance.
Span20:Tween20
ratio HLB numbers Mean particle size (nm) Zeta potential values (mV)
50:50 12.7 ▲ ▲
67:33 11.3 638.9 ± 18.2 28.6 ± 4.1
75:25 10.6 270.1 ± 9.8 32.6 ± 1.6
80:20 10.2 305.3 ± 18.3 35.8 ± 3.1
▲ Precipitation of aggregates was observed.
Table 2. Mean particle sizes and zeta potential values for chitosan/heparin nanoemulsion particles in with different concentrations of Span 20/Tween 20 mixture surfactant in the water–in–oil emulsification system (n = 5).
Span 20/Tween 20 mixture
surfactant concentration (wt%) Mean particle size (nm) Zeta potential values (mV)
0.50% ▲ ▲
0.75% 932.6 ± 35.9 38.9 ± 9.8
1.00% 728.8 ± 5.2 36.6 ± 2.5
1.25% 324.2 ± 4.6 34.6 ± 3.6
1.50% 270.1 ± 9.8 32.6 ± 1.6
Figure Captions
Fig. 1. (A) A representation for prepared amoxicillin loaded in chitosan/heparin nanoemulsion particles and the strategy for eradicating H. pylori using the nanoemulsion particles. (B) TEM micrographs of the nanoemulsion nanoparticles at different Span 20/Tween 20 mixture surfactant compositions.
Fig. 2. (A) FT-IR spectra of chitosan/heparin nanoemulsion particles in specific pH environments. (B) TEM micrographs of the prepared nanoemulsion nanoparticles in specific pH environments. (C) In vitro release profiles of amoxicillin from amoxicillin-loaded nanoemulsion particles at different pH values at 37℃ (n = 5).
Fig. 3. (A) Cell viability after treatment with different concentrations of chitosan/heparin nanoemulsion particles (n = 6). (B) Confocal images of AGS cells incubated with internalized Cy3-chitosan/FA-heparin nanoemulsion particles for 120 min.
Fig. 4. Fluorescent images (after 3D reconstruction) of AGS cell monolayers infected with fluorescent H. pylori (DilC18(5)-labeled H. pylori) and incubated with FA-amoxicillin solution alone or FA-amoxicillin-loaded fluorescent Cy3-chitosan/heparin nanoemulsion particles for 2 hr.
Fig. 5. Changes in cell growth after AGS cells were pre-infected with H. pyloi for 2 hr and then incubated with amoxicillin solution alone or amoxicillin-loaded chitosan/heparin nanoemulsion particles.
Fig. 6. Effects of amoxicillin solution alone and amoxicillin-loaded chitosan/heparin nanoemulsion particles in a H. pylori-induced gastric infection mouse model