mnw
mwioiw~onvowv
Marine Micropaleontology 32 (1997) 95-l 14Glacial-Holocene
calcareous nannofossils and paleoceanography
in
the northern South China Sea
Kuo-Yen Wei *, Tien-Nan Yang, Chi-Yue Huang
Department of Geology, National Taiwan University, Taipei, Taiwan Received
I June 1996; accepted 1 December 1996
Abstract
Last glacial to Holocene paleoceanography of the northern South China Sea was inferred from nannofossil variations and several hydrographic proxies from a piston core, SCS90-36 (17”59.7O’N, 111”29.64’E, water depth 2050 m). The upper part of the sedimentary sequence (dated 15.5 to 1.2 ka) provided a high-resolution record whereas part of the sediments older than 15.5 ka was lost due to erosion. A correspondence analysis of the nannofossil succession suggests that the paleoceanography developed
in four stages. The first stage (26-13.3 ka) has a fairly well-preserved diverse nannoflora
dominated by
Gephyrocapsaand
Florisphaera projkda. The floral composition together with high concentration of ketones (C,,) and organic carbon indicates high surface-water fertility. The second stage, the deglacial period (13.3-10.7 ka), had an increased surface-water turbidity and a stronger influence of Pacific open-ocean waters as evidenced by the decrease of Florisphaera profundaand increase of
Emiliania huxleyi,respectively. A preservation peak of calcareous
microfossils centered at 12 ka correlates to the global preservation event of Termination I. The third stage, early Holocene
(10.74.4 ka), is marked by a gradual increase of F:
profunda and small placolith taxa at the expense of E. huxleyi. The floral composition indicates that conditions were more oligotrophic compared to the pre-Holocene. The preservation of nannofossils became progressively worse, indicating a rise of the nannofossil lysocline. In sediments deposited at 5.5 and 4 ka, nannofossil preservation improves, probably reflecting a local cooling event. During the last stage, from 4.4 to 1.2 ka, E. huxleyi, Umbilicosphaeraand large
Reticulofenestraincreased their relative abundance to replace small placoliths.
Further stratification of the surface water column may have been responsible for this floral succession.
Keywords: Pleistocene; Holocene; South China Sea; nannofossil; paleoceanography
1. Introduction
The extensive development
of glaciers in polar ar-
eas during the last glacial maximum
(LGM) resulted
in a sea-level
drop by 120 m (Fairbanks,
1989).
Most straits that bound the South China Sea (SCS)
became
too shallow
to allow exchange
of waters
*Corresponding author. Tel.: +886-2-3691143; fax: +866-2- 3636095; e-mail: [email protected]between the South China Sea and the open oceans.
The Bashi Strait, located between Taiwan and Lu-
zon Island (Fig. l), with a sill depth of 2500 m,
was the only passageway
to the West Philippine
Sea
during the last glacial. The exposure of continental
shelves, especially
in the southern part, also caused
a l/5 reduction
of the surface area of the South
China Sea (Wang, 1990). The lower sea level might
also have caused the Kuroshio to shift offshore, east
of its current position
(Ujiie et al., 1991; Ahagon
0377~8398/97/$17.00 C 1997 Elsevier Science B.V. All rights reserved.96 K.-Y; Wei et d/Marine Micropaleontology 32 (1997) 95-114
llZ@ 114O 116' 11e 120@
TAIWAN STRAI
ll? 114O 116' 1w 120'
Fig. 1. Locations of Core SCS90-36 studied and Core RC26-16 mentioned in the text.
et al., 1993) putting the South China Sea under a
stronger influence
of the Continent
Coastal Current
(Wang and Wang, 1990). In the northwestern
Pacific,
the Kuroshio Front advanced
southward
during the
last glacial (Moore et al., 1980; Thompson,
1981).
The Kuroshio Front then moved northward at about
13-10 ka (Chinzei
et al., 1987), synchronous
with
the post-glacial
warming of the North Atlantic Ocean
(Duplessy et al., 1981). There were, however, two in-
tervening retreats of the Kuroshio Front to the south
during the post-glacial
period; the first was at 1 l-10
ka, corresponding
to the Younger Dryas Event while
the second took place after 5.5 ka. Both events were
associated with brief cooling (Chinzei et al., 1987).
The winter sea surface temperatures
(SSTs) in the
northern part of the South China Sea were estimated
to be4-6”C
lower than today on average (at RC26- 16
by Wei et al., 1996; at SCS 90-36 by Huang et al.,
1997; and to the south, near the Mindoro
Island,
by Miao et al., 1994). The summer SSTs were es-
timated to be quite similar to today’s or 2°C lower
(Wei et al., 1996; Miao et al., 1994; Huang et al.,
1997). The larger seasonal temperature
contrast has
been attributed to reduced surface water exchanges
across the straits and to a stronger winter monsoon
(Miao et al., 1994). This stronger winter monsoon
during the glacial might also have resulted in better
mixing
of the surface water and so have induced
higher biological
production
(Huang et al., 1997).
A subsequent
drop of surface productivity
during
the transition
from the glacial to the postglacial
has
also been documented
in other western Pacific low-
latitude areas and in the SCS (Herguera and Berger,
1991; Herguera, 1992; Thunell et al., 1992). Both the
mixing rate and nutrient
concentration
of the deep
waters were lowered during the glacial-postglacial
transition (Herguera and Bergen 1994).
Huang et al. (1997) documented
a rich set of
last glacial to Holocene
paleoceanographic
proxies
and interpretations
from the piston core SCS90-36
located on the northern
slope of the South China
Sea (Fig. 1). Here we document a quantitative
study
of the calcareous
nannofossil
assemblages
from the
same core. We attempt to integrate the nannofossil
K.-E Wei et al. /Marine Micropaleontology 32 (1997) 95-114 97
evidence with the other available data to further illus-
trate the deglacial warming and climatic fluctuations
during the Holocene
in the northern
South China
Sea.
2.
Samples and procedures
The piston core SCS 90-36 was recovered from
17”59.70’N, 111”29.64’E, at a water depth of 2050 m
on the southern slope of the Xisha Trough (Fig. 1).
The sequence
consists
of fine silty clay without
any detectable
turbidite
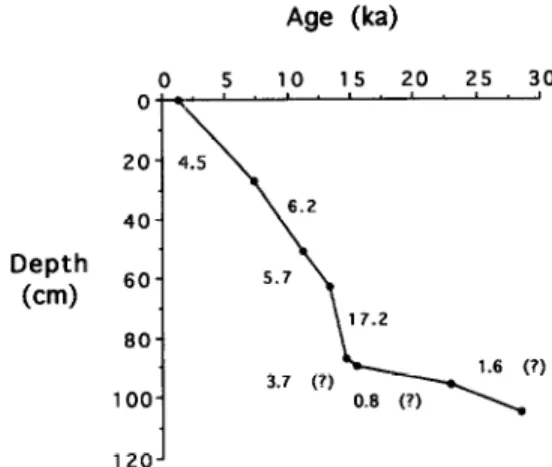
layers. The chronology
of
the sequence
was constrained
by eight radiocarbon
datings
of planktic
foraminifera
using accelerator
mass spectrometry
(AMS)
(Fig. 2; Huang
et al.,
1997). The radiocarbon
age of the bottom of the
studied sequence is about 26 kyr. The extremely low
sedimentation
rates (~2 cm/kyr) in the lowermost
part (Fig. 2), however, have led us to suspect that
most of the sediments
deposited
prior to 15.5 ka
were lost due to erosion.
Samples
were taken every 3 cm from the top
103 cm of the core, corresponding
to a temporal
resolution
of -500
yr per sample for the Holocene
and -200
yr for the deglacial period. A total of 32
samples were analyzed. At least 700 coccoliths were
identified and tallied per sample under a Zeiss Pho-
tomicroscope
at a magnification
of 1250x.
Several
samples were examined
under a scanning
electron
microscope
to facilitate
taxonomic
identifications.
Age W
0 5 10 15 20 25 30Depth
6 o(cm)
a0
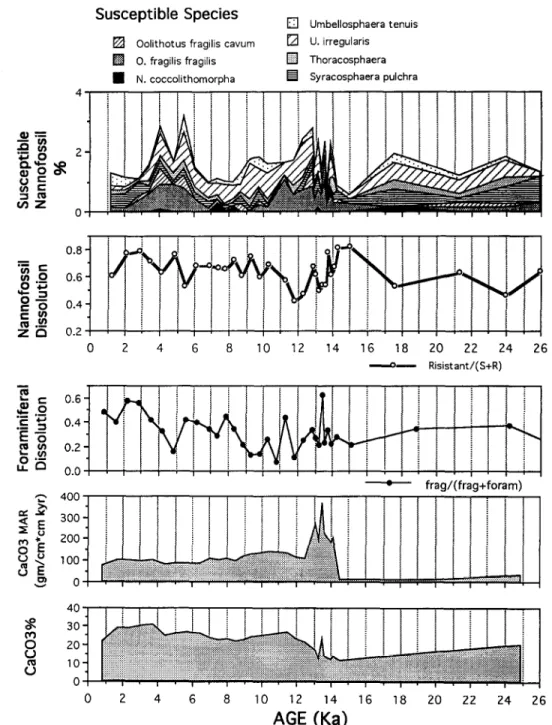
17.2 12oJFig. 2. Depth-age diagram of Core SCS90-36, showing sedi- mentation rates estimated from eight AMS 14C dates of planktic foraminifera.
Table 1
List of tallied nannofossils
Calcidiscus leptoporus a (Murray and Blackman) Loebhch and Tappan, 1978
Calcidiscus macintyrei b (Bukry and Bramlete) Loeblich and Tappan, 1978
Calciosolenia murrayi Gran, 1912 Ceratolithus cristatus a Kamptner, 1950
Coronocyclus nitescens b (Kamptner) Bramlette and Sullivan, 1961
Crenalithus sp. Chen, 1978 Cricolithus jonesi Cohen, 1965 Dictyococcites perplexa Bums, 1975
Dictyococcites productus (Kamptner) Backman, 1980 Discoaster spp. b
Emiliania huxleyi (Lohmann) Hay and Mohler, 1967 Florisphaera ptojiatda profunda Okada and Honjo, 1973 Florisphaera profunda elongata Okada and McIntyre, 1979 Gephyrocapsa caribbeanica Boudreaux and Hay, 1967 Gephyrocapsa oceanica Kamptner, 1943, see Matsuoka and
Okada, 1989
Helicosphaera carteri carteri a (Wallich) Kamptner, 1954 Helicosphaera carteri hyalina a Gaarder, 1970
Helicosphaera carteri wallichii a (Lohmann) Okada and McIntyre, 1977
Neosphaera coccolithomorpha ’ Lecal-Schlauder, 195 1 OolithotusfragilisC (Lohmann) Okada and McIntyre, 1977 Pontosphaera discopora Schiller, 1925
Pontosphaera japonica (Takayama) Bums, 1973 Pontosphaera multipora (Kamptner) Roth, 1970 Pseudoemiliania lacunosa b (Kamptner) Gartner, 1969 Reticulofenestra haqii Backman, 1978
Reticulofenestra minuta Roth, 1970
Reticulofenestra minutulu (Gartner) Haq and Berggren, 1978 Rhabdosphaera clavigera a Murray and Blackman 1898 Rhabdosphaera longistylis a Schiller, 1925
Syracosphaera histrica ’ Kamptner, 1941 Syracosphaera lamina ’ Lecal-Schlauder, 195 1 Syracosphaera pulchra ’ Lohmann, 1902 Syracosphaera spp. ’
Thoracosphaera spp. ’
Umbellosphaera irregularis ’ Paasche, 1955 Umbellosphaera tenuis ’ (Kamptner) Paasche, 1955 Umbilicosphaeru sibogae a (Weber-van Bosse) Gaarder, 1970 a Dissolution-resistant.
b Reworked species. ’ Dissolution-susceptible.
The nannofossil
taxa tallied are listed in Table 1.
Taxonomic
diagnoses
and nomenclatural
notes on
some selected species are given in Appendix A.
To monitor
the preservation
state of the nanno-
fossil assemblages,
a dissolution
index, defined as
the ratio of resistant
species to resistant
+ suscep-
98 K.-E Wei et al. /Marine Micropaleontology 32 (1997) 95-114
tible species, was calculated.
We adopted Roth and
Coulboums’ ranking (Roth and Coulboum,
1982) of
the north tropical Pacific nannofossil
dissolution
sus-
ceptibility
to assign the various species into the two
categories
(Table 1). Only minor and rare species
were included in the calculation
in order to avoid be-
ing overwhelmed
by dominant
species and hopefully
to reduce artifacts introduced
by factors other than
dissolution.
A rich set of paleoceanographic
proxies, including
weight percentage
of carbonate
and organic carbon,
relative abundances
of planktic foraminifera,
carbon
and oxygen isotopic ratios of planktic
and benthic
foraminifera,
U,k index of long-chain
ketones
as
well as abundance of Cs7 ketanes, were measured by
Huang et al. (1997).
3.
Multivariate analysis
To recognize
the down-core
nannofossil
varia-
tion pattern,
we applied
a multivariate
ordination
technique,
detrended
correspondence
analysis
(D-
CA) (Hill and Gauch, 1980), to the nannofloral
count
data. This method allows taxa and samples to be
ordinated simultaneously
and therefore shows the re-
lationship between them. The DCA is a modification
of regular correspondence
analysis to eliminate
the
‘arch effect’ that normally
distorts the projection
of
data points on the first axis. This modified method
can adjust the scores so that the data points are more
linear along the first axis and more evenly spread
out. The DCA assumes that the samples come from
a gradient in which different variables
(taxa) char-
acterize different parts of the gradient. In applying
this method, we explicitly seek an ordination
pattern
along the chronological
axis. In other words, we are
trying to identify taxa which show most age-depen-
dent variation and therefore characterize
the various
stages in the paleoceanographic
development.
All reworked
taxa and taxa with relative abun-
dances less than 1% were excluded. To reduce skew-
ness and to improve normality
of data distribution,
the original percentage
data were natural log trans-
formed prior to analysis.
The transformation
also
moderately
weights the minor and rare taxa relative
to the dominant
taxa so that variations
in the minor
taxa will not be overwhelmed
by the dominant ones.
4. Results and interpretations
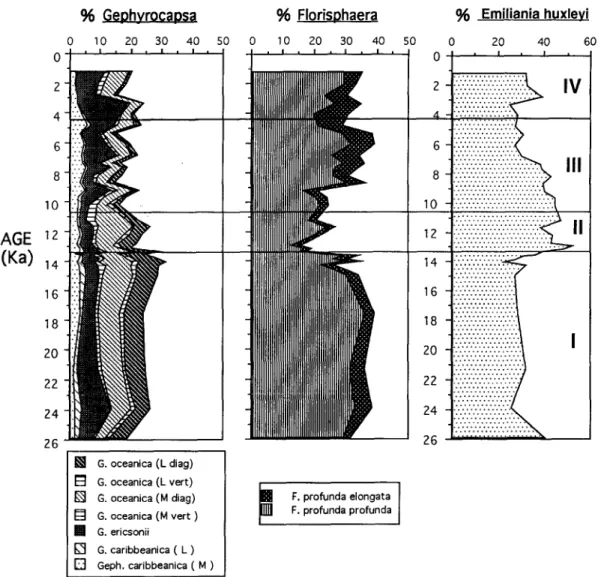
Calcareous
nannofossils
are
fairly
common
throughout
the studied section and display a diverse
assemblage
with good preservation.
Between 28 and
39 species/subspecies
were recorded
per sample.
Representative
species are illustrated
in Plates I-IV.
The nannofossil
assemblages
were dominated
by
three taxa:
Gephyrocapsa spp., Emiliania huxleyi
and a deep dwelling species,
Florisphaera profunda
(Fig. 3). The latter two are considered
to be pelagic
species,
increasing
their relative
abundances
with
water depth, whereas
Gephyrocapsa has a prefer-
ence for neritic environments
in the marginal seas of
the western Pacific (Chen and Shieh, 1982; Okada,
1983, 1992; Cheng, 1992; Chen and Huang, 1995).
The compositions
of the studied samples fall gen-
erally in the bathypelagic
province
of open sea in
the western
Pacific as defined
by Okada (1983).
The occurrence
of
Syracosphaera, Rhabdosphaera,
Oolithotus, Ceratolithus and Umbellosphaera, al-
though rare in abundance,
is typical for the bathy-
pelagic environments
in the western Pacific marginal
seas (Chen and Shieh, 1982; Okada, 1983; Wang and
Samtleben,
1983; Cheng, 1992).
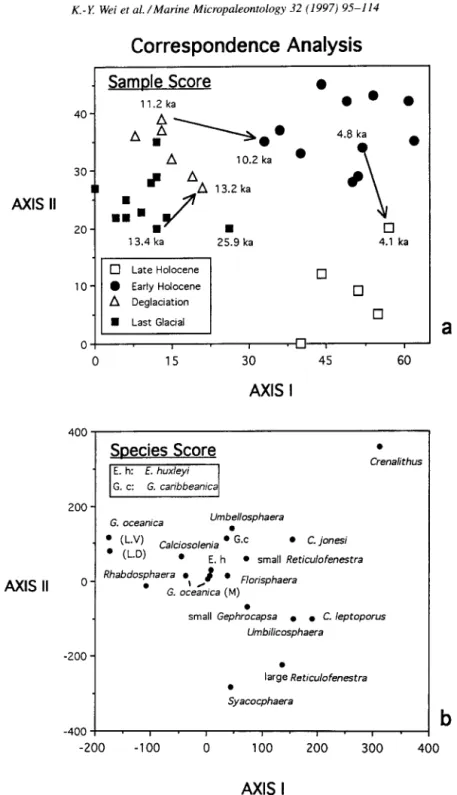
The correspondence
analysis yielded a quite inter-
pretable pattern (Fig. 4). The first two axes account
for 44 and 11% of the data variation,
respectively.
Samples exhibit a well-phased
chronological
ordina-
tion. The floral succession
can be subdivided
into
four stages: Stage I, dated 25.9-13.3
ka, represented
by the glacial assemblage;
Stage II, from 13.3 to 10.7
ka, marked by the deglacial flora; Stage III, 10.74.4
ka, the early Holocene;
and Stage IV, 4.4-1.2
ka,
the late Holocene.
The glacial and deglacial
sam-
ples are clustered
and distributed
on the left half
of the first axis, with a progressive
upward migra-
tion along the second axis. In contrast, the Holocene
samples occupy the right half of the first axis and
show a downward
transition
along the second axis.
The boundaries
of stages are characterized
by major
shifts in the data trajectories
(Fig. 4a). The charac-
teristic nannofossils
of each stage can be found in
the corresponding
positions on the projection
of the
species scores on the two eigen-axes
(Fig. 4b).
The glacial stage (Stage I, 25.9-13.3
ka) is char-
acterized
by the dominance
of
Gephyrocapsa and
K.-I: Wei et al. /Marine Micropaleontology 32 (1997) 95-114 99 96
Gephvrocapsa
96
FlorisDhaera
% Emiliania huxlevi
0 10 20304050 0 1020304050 0 20 40 60 10 ;zz; l2 14 16 18 20 22 24 26
id
G.
oceanica (L diag) n G. oceanica (L vert) @ G. oceanica (M diag) E3 G. oceanica (M vert ) G. ericsonii m G. caribbeanica ( L ) E] Geph. caribbeanica ( M ) - ... ... ... ... 1 6 - ::;:::::::;::i ... ... ... ...Fig. 3. Downcore variation of the relative abundances of three dominant nannofossil taxa. Horizontal lines mark the boundaries of four stages recognized from the correspondence analysis shown in Fig. 4.
Florisphaeru (Fig. 3). The diverse flora and more
frequent
appearance
of dissolution-susceptible
taxa
(such as
Umbellosphaera, Syracosphaera and Cal-
ciosoknia murruyi) relative to other stages suggests
that the nannofossil
preservation
in the glacial in-
terval is generally better. Since the intermediate
and
deep waters of the SCS during the glacials were only
exchanged
with Pacific Intermediate
waters through
the Bashi Strait, the bottom water chemistry
at the
studied site is believed
to be mainly
governed
by
that of the Pacific Intermediate
waters. The good
preservation
of carbonate during glacials was due to
reduced production
of North Atlantic
Deep Waters
(NADW)
and probably
the formation
of nutrient-
depleted
Pacific intermediate
waters
at that time
(Thunell et al., 1992).
Gephyrocapsa tends to dominate in highly fertile
waters. Winter (1982) found that the distribution
of
G. oceanica was closely related to the concentration
of phosphate in the Gulf of Elat (‘Aqaba), Red Sea.
The abundance
of G.
oceanica in the northern South
China Sea also shows a decreasing
trend with con-
centration
of phosphate from south to north (Cheng,
1992). The predominance
of large
Gephyrocapsa in
the nannoflora
(Fig. 6), in association
with the high
concentration
of organic carbon and ketones (CY)
100 K.-Y Wei et al. /Marine Micropaleontology 32 (1997) 95-114
Correspondence
Analysis
1
Sample
Score
11.2
ka0
0.
0
40A ;\,’
4.8 ka 0A
10.2 ka a 30??
‘A
13.2ka AXIS II ?? ??W . 20 ?? 13.4 ka ??Last Glacial ??0
25.9
ka 4.1 ka 0 0 0 ” I ‘Y I0
1;
3;
45 60AXIS I
SDecies Score
-V]
0 Crenalithus 200 - G. oceanica Umbellosphaera ..
??(L.V) Caldosolenia ??G’c ?? C. jonesi ’ U-.D) ?? E. h ??small ReticulofenestraAXIS
II
O
Rhabdosphaera
??3
??Fjorjsphaera
.
G. oje&a (M)a
.
small Gephrocapsa . ??C. leptoporus Umbilicosphaera -200 - . large Reticulofenestra . Syacocphaera
b
-400 I ’ I ’ I I ., . -200 -100 0 100 200 300 400AXIS
I
Fig. 4. Scores of samples (upper panel) and species (lower panel) on the plane of the first two eigen-axes resulting from detrended correspondence analysis.
(Fig. 7; Huang et al., 1997) suggests the existence
of a less maritime
environment
with high fertility
conditions
during the last glacial. The surface-water
productivity
in the SCS was estimated
to be two
times higher during the last glacial maximum
than
in the Holocene (Thunell et al., 1992). Furthermore,
K.-l! Wei et al. /Marine Micropaleontology 32 (1997) 95-114 101
the high frequency
of
Rhabdosphaera spp. (Fig. 6),
a characteristic
cold-subtropical
species (Winter et
al., 1994), is also indicative
of colder conditions
in
comparison
to the post-glacial
stages II-IV.
The increase
in
Emiliania huxleyi during the
deglacial
interval
{(Stage II, 13.3-10.7
ka, Fig. 3)
indicates
an increasing
influence
of the open-ocean
waters due to the concomitant
rise of global sea level
(Fairbanks,
1989). At the same time, in the north-
western
Pacific the Kuroshio
Front moved north-
ward (Chinzei
et al., 1987), synchronous
with the
post-glacial
warming
of the North Atlantic
Ocean
(Duplessy et al., 1981).
The major drop of
Florisphaera between 13.3
and 9 ka (Fig. 3) may either indicate a rise of the
nutricline,
or an increase
of turbidity
of the sur-
face waters. Among the various coccolithophorids,
Florisphaera is distinctive
in living in the lower
euphotic
zone with low light and high nutrients.
Its habitat is restricted to waters below 100 m and
warmer than 10°C (Okada and Honjo, 1973; Honjo
and Okada,
1974; Okada
and McIntyre,
1979).
Molfino
and McIntyre
(1990a,b)
proposed
a con-
ceptual model that when the nutricline
is deeper the
growth of
Florisphaera would be enhanced relative
to surface dwelling
coccolithophorids.
The relative
abundance
of
Florisphaera therefore is indicative of
fluctuation
of the nutricline
in the euphotic zone; the
higher the relative abundance,
the deeper the nutri-
cline. On the other hand, Ahagon et al. (1993) argued
that this model is only applicable
to the equatorial
areas where the nutricline
depth is mainly governed
by the equatorial
divergence.
Ahagon et al. (1993)
instead documented
that in the marginal seas around
the Japanese islands, there was a close relationship
between
the F:
profunda abundance
and seawater
transparency
(Secchi depth). Given the proximity
of
the northern South China Sea to the continent and the
relatively
high sedimentation
rates of the sequence,
we follow Ahagon et al. (1993) in suggesting that the
decrease of the relative abundance
of
Florisphaera
profunda is an indication of increasing turbidity of
the surface waters rather than a shallowing
of the
nutricline.
Increasing
turbidity
is probably
caused
by increased lateral transportation
of fine sediments
carried by the newly established
Continent
Coastal
Current
flowing
through
Taiwan
Strait and conti-
nental shelves off the Pearl River (Zhujiang)
mouth
(Pinxian
Wang, pers. commun.,
1995). The rise of
the sea level due to the global meltwater discharges
during the deglacial period (13.0-l 1.5 and 10.0-9.0
ka, Fairbanks,
1989) would have caused the Taiwan
Strait submerged
and become a passageway
through
which the Continent
Coastal Current flew southward
and flushed previously
deposited
sediments
from
continental
shelves. The fine particles were probably
transported
by contour currents, forming nepheloid
layers with high turbidity.
On the other hand, the timing (13-9 ka) of the
drop of the relative abundance
of E
profunda is con-
sistent with that recorded in the equatorial
Atlantic
(Molfino
and McIntyre,
1990b). If this reduction
of E
profunda abundance is a global phenomenon,
occurring
not only in the Atlantic
equatorial
diver-
gence but also in the western Pacific marginal
seas,
then it might suggest either a global shallowing
of
nutricline
depth, or a global increase of cloudiness
(resulting
in less penetration
of sunlight).
Such a
global mechanism,
however, is difficult to imagine.
The intervals
dated between
12.5 and 11.5 ka
showed the best nannofossil
preservation,
contain-
ing many
Umbellosphaera and Oolithotus fragilis
(Figs. 5 and 6). Planktic
foraminifera
also showed
relatively
good preservation
in the same interval
(Fig. 5; Huang et al., 1997). The dissolution
inten-
sity of the bottom water during the deglacial
time
is inferred to be the lowest throughout
the past 15.5
kyr (Fig. 5), synchronous
with the global deglacial
preservation
spike of Termination
I (Berger, 1977).
Boyle (1988) suggested that during the deglaciation
carbonate preservation
was enhanced. During Termi-
nation I the circulation
of the intermediate
waters of
the NSCS already appears closely linked with that
of the Pacific intermediate
waters, whose corrosive-
ness, in turn, was governed by the relative strength
of North Atlantic
Deep Water and Antarctic
Bot-
tom Water. The occurrence of this preservation
spike
agrees with the ‘nutrient
redistribution
model’ of
Boyle (1988).
Another
significant
change during the deglacial
period is shown by
Gephyrocapsa. The total abun-
dance of
Gephyrocapsa (Fig. 3) continuously
de-
creased from the glacial level through the deglacial
to the late Holocene. Furthermore,
within the
Gephy-
rocapsa complex, the dominance of the large forms
of G.
oceanica was first displaced by the medium-
102 K.-Z Wei et al. /Marine Micropaleontology 32 (1997) 95-114
Susceptible Species
? ?
Umbellosphaera tenuis? ?
Oolithotus fragllis cavum? ?
U. irregularis? ?
0. fragilis fragllis? ?
Thoracosphaera??
N. coccolithomorpha H Syracosphaera pulchra0 2 4 6 8 10 12 14 16 18 20 22 24 26
-- Risistant/(S+R)
- frag/(fr&+foram)
0 2 4 6 8 10 12 14 16 18 20 22 24 26
AGE (Ka)
Fig. 5. Summary of microfossil indicators of carbonate dissolution in comparison with down-core variations in %CaCO, and CaCOj mass accumulation rate.
sized G. oceanica
during the deglacial
stage, and
later during the Holocene
by small forms, collec-
tively designated
as small Gephyrocupsa
(cf. Mat-
suoka and Okada, 1989). This diminution
trend is
also mirrored by the progressive
increase
of small
placolith
forms in the upper section
of the core.
These small placoliths
include
Cricolithus jonesi,
Crenalithus
sp., Dictyococcites,
as well as small
Reticulofenestra
forms such as R. minuta (Fig. 4b
and Fig. 6).
104 K.-Y Wei et al. /Marine Micropaleontology 32 (1997) 95-114
AGE
W)
tP80
G. sacculifer
6a
16 18 24 26uk&
SSTs
OC
22 ,,
23 24 25 26 ; I. I , I. I , IKWng4
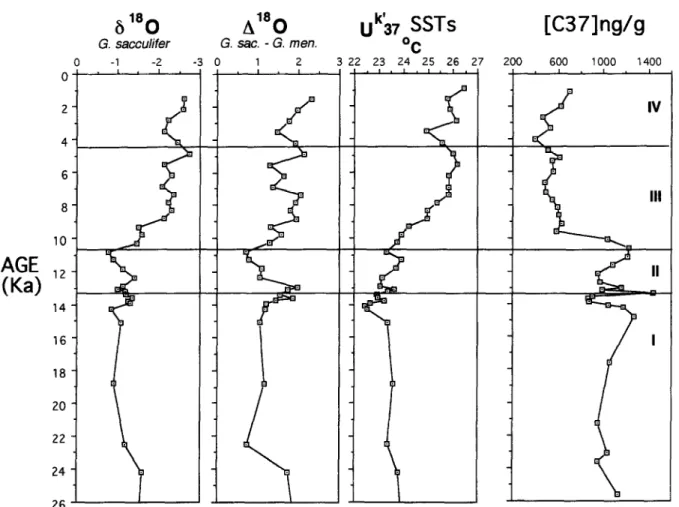
200 600 1000 1400 I . I , I . I IFig. 7. Time-series of 6’8O of planktic foraminifera Globigerinoides sacculifer, 6”O between surface-dwelling (G. sacculifer) and subsurface-dwelling (Globoroculia manardii) species, sea-surface paleotemperature derived from unsaturation ratio of long-chain methyl alkenones ((I$), and ketone (C37) concentration. Data are from Huang et al., 1997.
The early Holocene
(10.7-4.4
ka) assemblage
is
dominated
by
Florisphaera, second by E. huxleyi
and third by
Gephyrocupsa (Fig. 3). Apparently,
the post-deglaciation
condition
has been more mar-
itime with less terrigenous
influence
compared
to
the previous
periods. Small placoliths
became rel-
atively abundant.
These small forms are probably
opportunistic
elements of the flora.
Umbellosphaera,
which inhabits tropical oligotrophic
pelagic surface
waters (McIntyre et al., 1970; Young, 1994), also be-
came relatively more abundant. Such a floral compo-
sition indicates that it was a warmer, more maritime,
well-stratified
oligotrophic
environment
during the
early Holocene.
This interpretation
is supported by
the increase
in 6180 gradient
between
the surface
water (represented
by the planktic foraminifera
Glo-
bigerinoides sacculijkr) and the subsurface water
(50-200
m, represented
by
Globorotalia menardii)
(Fig. 7; Huang et al., 1997). This larger gradient
is indicative
of stronger
stratification
of the water
column. Strong monsoon winds might intermittently
deepen the mixed layer and cause nutrient injection
into the euphotic
zone, inducing
blooming
of op-
portunistic
species such as small placoliths
and
E.
huxleyi.
At about 5.5 and 4.0 ka the preservation
of cal-
careous nannofossils
slightly improved. The dissolu-
tion index of planktic foraminifera
shows also a pe-
riod of better preservation
centered at 5 ka (Fig. 5).
This short period predated slightly the episodic cool-
ing of 1 to 2°C between 4.5 and 3.0 ka (Fig. 7).
K.-E Wei et al./Marine Micropaleontology 32 (1997) 95-114 105
iths decreased during the last 4000 yr while that
of larger forms such as CuZcidiscus Zeptoporus,
R.
minutula
and
R. haqii
increased (Figs. 4 and 6). The
vertical 6180 gradient also increased (Fig. 7), sig-
nifying a more stratified surface-water column. The
frequency and/or intensity of seasonal mixing might
have become lower, which in turn would have re-
duced the yield of :small placoliths in the flora. By 3
ka, sea surface temperatures had become similar to
today’s (Fig. 7).
Throughout the core, the variations of nannofos-
sil preservation bear no obvious relationship to the
variations in CaCOs content or CaC03 accumulation
rate (Fig. 5). This suggests that although the preser-
vation of nannofossils may have been governed by
the general corrosiveness of the intermediate waters
of the SCS, the influence is not necessarily reflected
in the accumulation rates of CaCOs or %CaCOs.
The depth of this site (2050 m) is slightly above
the present nannofossil lysocline (2300 m, Chen and
Shieh, 1982). The nannofossil assemblage therefore
is considered to be less sensitive to the glacial deep-
ening of the lysocline and carbonate compensation
depth (CCD, at 3000 m at present and 4000 m during
the LGM; Rottman, 1979; Thunell et al., 1992) than
other deeper sites. Owing to this site’s proximity to
the continent and riverine sediment input sources of
the Pearl River (Zhujiang) and the Hanjiang River,
the influx of terrestrial sediments is considered to
be the primary controlling factor of sedimentation
of this site, while the bottom water chemistry is
secondary.
5. Summary and conclusions
(1) A quantitative study was performed on last
glacial to Holocene (25.9-1.2 ka) nannofossils from
a piston core collected from the northern South
China Sea. Integrated with other proxies, the time-
progressive change in the nannoflora reveals the
paleoceanographic evolution of the area since the
last glacial.
(2) A detrended correspondence analysis of the
nannofloral data suggests that the floral succession
can be subdivided into four stages, corresponding
to the major paleoceanographic developments in this
area: Stage I (25.9-13.3 ka), glacial stage; Stage II
(13.3-10.7 ka), deglacial stage; Stage III (10.74.4
ka), early Holocene and Stage IV (4.4-1.2 ka), late
Holocene.
(3) The glacial stage is characterized by the domi-
nance of
Gephyrocapsa
and
Florisphaera,
indicating
strong continental influence with high fertility. Good
preservation of nannofossil and high concentration
of ketone and organic carbon are caused by both
high biological productivity and less corrosiveness
of the bottom waters.
(4) The increase of
E. huxleyi
in the deglacial
interval suggests an increasing influence of the open-
ocean, corresponding to the global sea-level rise. The
decrease of a deep-dwelling taxa,
Florisphaera pro-
fundu,
is interpreted as responding to the increased
turbidity in surface waters due to the increased in-
put of fine, flushed sediments during the deglacial
period by the Continental Coastal Current. A preser-
vation spike of nannofossils centered at 12 ka is also
identified.
(5) A time-transgressive
diminution trend of
Gephyrocupsa
existed through the glacial-Holocene,
parallel to the decreasing trend of the relative abun-
dance of
Gephyrocupsa.
This trend is accompanied
by the increase of other small placoliths, espe-
cially during the Holocene. This might suggest that
the NSCS became more maritime and oligotrophic
through time, while the surface waters became more
stratified as indicated by the increase of the vertical
6180 gradient. These small placoliths were oppor-
tunistic elements generated during blooming seasons
when seasonal mixing of the surface waters took
place.
(6) Two episodic cooling events were identified
in the post-glacial stages, one at about 11-10 ka,
the other at 5-3 ka. Both are characterized by bet-
ter preservation of nannofossil assemblages and a
drop in sea-surface paleotemperature of l-2°C as
indicated by the U,k index.
Acknowledgements
We would like to thank Tom Marchitto, Pinxian
Wang, Jeremy Young and an anonymous reviewer
for improving the manuscript with their comments
and criticisms. This paper is a contribution of the
ROC PAGES project. Research funding was pro-
vided by Grant No. NSC84-261 l-M-002~OO4GP of
the National Science Council, ROC.
106 K.-E Wei et al. /Marine Micropaleontology 32 (1997) 95-114
IO
20
K.-E Wei et al. /Marine Micropaleontology 32 (1997) 95-114 107
Appendix A. Thxonomic remarks on selected
taxa
Small elliptical placoliths
Small placoliths belonging to Reticulofenestra, Dictyococ- cites, Gephytocapsa, Emiliania and Crenalithus ate often hard to adequately differentiate under the light microscope due to their small sizes. For the sake of stability and reproducibility of taxonomic identification, we mainly followed strategies outlined
Plate I
Electron micrographs of nannofossils from Core 90-36. Scale bar =3pm.
1. Emiliania huxleyi (Lohmann) Hay and Mohler, proximal view. Subdepth = 17 cm.
2. Emiliania huxleyi (Lohmann) Hay and Mohler, distal view. Subdepth = 17 cm.
3. Reticulofenestra minutula (Gartner) Haq and Berggren, proxi- mal view. Subdepth = 62 cm.
4. Reticulofenestra minuta Roth, proximal view. Subdepth = 62 cm.
5. Cricolithus jonesi Cohen, distal view. Subdepth = 98 cm. 6. Umbellosphaera irregularis Paasche, proximal view. Subdepth = 62 cm.
7. Umbellosphaera tenuis (Kamptner) Paasche, distal view. Sub- depth = 17 cm.
8. Umbilicosphnera sibogae (Weber-van Bosse) Gaarder, distal view. Subdepth = 8 cm.
9. Umbilicosphaera sibogae (Weber-van Bosse) Gaarder, proxi- mal view. Subdepth = 44 cm.
10. Gephyrocapsu oceanica Kamptner (medium-sized, diagonal bar), distal view. Subdepth = 17 cm.
11. Gephyrocupsa oceanica Kamptner (medium-sized, vertical bar), distal view. Subdepth = 17 cm.
12. Calcidiscus leptoporus (Murray and Blackman) Loeblich and Tappan, proximal view. Subdepth = 98 cm.
13. Uolithotus fragilis (Lohmann) Okada and McIntyre, distal view. Subdepth = 17 cm.
14. Oolithotus fragilis (L.ohmann) Okada and McIntyre, proximal view. Subdepth = 98 cm.
15. Syracosphaera pulchra Lohmann, distal view. Subdepth = 98 cm.
16. Syracosphaera lamina Lecal-Schlauder, proximal view. Sub- depth = 17 cm.
17. Syracosphaera lumina Lecal-Schlauder, distal view. Sub- depth = 17 cm.
18. Syrucosphaera sp. distal view. Subdepth = 98 cm. 19. Syracosphaeru mediterrunea Lohmann, distal view. Subdepth = 62 cm.
20. Helicosphneru carteri carteri, (Wallich) Kamptner, distal view. Subdepth = 44 cm.
21. Helicosphaera carteri hyalina, proximal view. Subdepth = 44 cm.
22. Helicosphaera carteri wullichii (Lohmann) Okada and McIn- tyre, proximal view. Subdepth = 44 cm.
by Rahman and Roth (1989), Matsuoka and Okada (1989) and Biekart (1989). The common spirit of these taxonomic schemes is their simplicity and practicality. Differentiation of the vari- ous genera is based on features that are recognizable under the light microscope, such as the overall size, presence/absence of a bridge, relative size of the central opening and extinction pattern under crossed-nicols
Elliptical placoliths showing the typical extinction pattern of Gephymcapsa but without visible bridges were counted as species of Reticulofenestra and Dictyococcites. Specimens with a more or less open central area were assigned to Reticulofenestra, while those with a completely closed central area were identified as Dictyococcites (Matsuoka and Okada, 1989). Two Dictyococ- cites species were recognized based upon size criteria (Backman, 1980; Matsuoka and Okada, 1989): D. productus (smaller than 4 pm) and D. perplexa (larger than 4 pm = D. antarcticus Haq, 1976).
Forms smaller than 4 bm showing a bright inner rim and faint birefringence along the outer margin were considered to be Emiliunia huxleyi. The slits between the ‘T-shaped’ elements on the distal shield are often visible under parallel nicols. However, small etched specimens of Reticulofenestru and Dictyococcites, as well as Gephyrocapsa with its bridge totally dissolved, may possibly be mis-identified as E. huxleyi. Forms similar to E. huxleyi in size, but showing stronger birefringence and a similar extinction pattern to that of Reticulofenestra, are considered to be Crenalithus sp. (Chen, 1978). For some authors, however, Crenalithus is considered to be a synonym of Reticulofenestra (Backman, 1980) or Dictyococcites (Biekart, 1989) (discussed later).
Forms close in size to R. minutula but showing a large central opening surrounded by a narrow rim of strong birefrin- gence were designated as Cricolithus jonesi (Cohen, 196.5; Chen, 1978, 1979). However, dissolved small syracosphaerids might be mistaken for this species under the light microscope.
Crenalithus and Reticulofenestra
Roth (1973) erected the genus Crenalithus for the small el- liptical placolith with the type species originally described as Coccolithus domnicoides Black and Barnes (1961). This genus is characterized by its small size, serrate margin and non-imbri- cated elements in the distal shield. From observation of recent coccolithophorids, Okada and McIntyre (1979) adopted the con- cept of this genus and erected new species and subspecies, C. parvulus parvulus, C. pan&us tecticentrum, C. punctatus and C. sessilis. They considered these living species to resemble the extinct C. doronicoides. Nishida (1979) and Steinmetz (1991) also reported living Crenalithus species. The proximal side of the central area of these living Crenalithus species is covered by either a solid plate or reticulate grid.
In this study, we classified those specimens which resemble R. minuta but with a larger central opening as Crenalithus sp.
(sensu Chen, 1978). However, these Crenalithus forms should
considered as small Reticulofenestra if one follows Backman’s argument (Backman, 1980). These forms are also possibly some dissolved forms of Dictyococcites productus.
108 K.-Y Wei et al. /Marine Micropaleontology 32 (1997) 95-114
K.-I: Wei et al. /Marine Micropaleontology 32 (1997) 95-114 109 cussed above, instead, we tried merely to differentiate as many
forms as possible to explore the paleoenvironmental significance of these small placoliths. Therefore, we tentatively retain the species concept of Crenalithus sp. proposed by Chen (1978) to encompass all the small elliptical placoliths (t3 pm) that have a large central opening and show a thin rim resembling the extinc- tion pattern of Reticulofenestra under crossed polarized light. We observed that such forms do exist in our samples and they could be consistently differentiated from other reported Reticulofenes- tra and Dictyococcites species, although the true identities of these Crenalithus awaits further detailed study.
Crenalithus sp. Chen (F’late III, 16)
Basionym: Chen, 1978, p. 139, pl. 4, figs. l-2.
Remarks: Small elliptical placolith (13 ,um) with non-imbri- cated or slightly overlapping elements. Under scanning electron microscope, it shows a smooth surface with little relief on the distal side. The elements are relatively wide and show a serrate margin. Central area is open. Under crossed polarized light, it
Plate II
Electron micrographs of nannofossils from Core 90-36. Scale bar =3pm.
I. Culciosolenia murrayi Gran. Subdepth = 17 cm.
2. Florisphaera profunda elongata (the larger one) and Florisphaera profunda profunau (the smaller one). Subdepth = 17 cm.
3. Discosphaera tubiferu (Murray and Blackman) Ostenfeld. Subdepth = 98 cm.
4. Discoaster sp. reworked. Subdepth = 62 cm.
5. Rhabodosphaera clavigera Murray and Blackman. Subdepth = 44 cm.
6. Rhabdosphnera clavigera Murray and Blackman. Subdepth = 62 cm.
7. Neosphaera coccolithomorpha Lecal-Schlauder, distal view. Subdepth = 17 cm.
8. Pontosphaeru multipora (Kamptner) Roth, distal view. Sub- depth = I7 cm.
9. Pontosphaera discopora Schiller. Subdepth = 98 cm. 10. Pontosphaera japonrca (Takayama) Nishida, proximal view. Subdepth = 98 cm.
I
I.
Ceratolithus crisratus Kamptner. Subdepth = 62 cm. 12. Coccosphere of Gephyrocapsa oceanica Kamptner. Subdepth = 98 cm.13. Thoracosphaera heimii (Lohmann) Kamptner. Subdepth = 62 cm.
14. Scyphosphaera sp. Subdepth = 98 cm. Backman (1980) considered Crenalithus to be a junior synonym of the genus Reticulofenestra. He rejected the specific epithet doronicoides and identified three species of small Reticulofenestru for the late Neogene: R. mint&da (3-5 wrn with a relatively large central opening), R. haqii (3-5 pm with a relatively smaller central opening) and R. minuta (~3 pm). We did not differentiate R. parvula (~2 pm in size = Crenalithus parvulus Okada and
McIntyre, 1979, sensu Biekart, 1989) from R. minutu.
shows a bright rim with an extinction pattern resembling that of Reticulofenestra. It differs from Dictyococcites productus and Reticulofenestra minuta by its open central area and narrow rim. It is distinguished from Gephyrocapsa by lacking a crossbar in the central opening.
Cricolithus jonesi Cohen (Plate I, 5; Plate III, 10)
Basionym: Cricolithus jonesi Cohen, 1965, p, 16, pl. 2, figs. J. K; pl. 16, figs. a-c.
References: Chen, 1978, p. 140, pl. 5, figs. 1-3; Chen, 1979, fig. 5; Chen and Shieh, 1982, pl. 1, figs. 5-8.
Gephyrocapsa
The proliferation of taxa assigned to the genus Gephyro- capsa and the associated variable concepts of species have caused a lot of controversies among specialists (Rahman and Roth, 1989; Matsuoka and Okada, 1989; Biekart, 1989). One of the problems associated with this group is the small size of its various forms such that the morphological characteris- tics of many species/subspecies are only recognizable under the scanning electron microscope. To facilitate a stable taxonomic practice under the light microscope, we have adopted the size criteria of Matsuoka and Okada (1989) to classify the Gephyro- capsa into three size classes: large forms (maximum diameter >5 pm), medium forms (between 5 and 2.5 pm) and small forms (~2.5 wm). Medium and large Gephyrocapsa were classified as G. caribbeanica or G. oceanica. The former has a smaller cen- tral area spanned by a robust bridge, whereas G. oceanicu has a relatively larger central opening. Specimens of G. oceanica were further assigned into two varieties: one with a vertically oriented bridge against the vertical axis and the other with a diagonally oriented bridge. The large G. oceanica with a diagonal bridge [abbreviated as G. oceanica (large, diagonal)] is similar to G. lumina Bukry (1973) while the large G. oceanica (vertical) is close to G. omega Bukry (1973). All small Gephyrocupsa forms were lumped as small Gephyrocapsa without further splitting. Some of these small Gephyrocapsa specimens, however, can be easily identified as G. ericsonii.
Genus Helicosphaera Kamptner, 1954
Some authors have proposed that Helicosphaeru wallichii and H. hyalina should be regarded as varieties of H. carteri (Theodoridis, 1984, Okada, 1992). We followed the decision made in the Florence Meeting of INA to include these forms as intraspecific varieties of H. carteri (Jordan and Young, 1990). Three varieties have been recognized as shown in Plates I and IV.
Reticulofenestra minuta Roth (Plate I, 4)
Basionym: Reticulofenestra minuta Roth, 1970, p. 850, pl. 5, figs. 3, 4.
Synonyms: Dictyococcites minutus (Haq) Haq, Lohmann and Wise, 1976, p. 759. Gephyrocapsa doronicoides (Black and Barnes) var. 4 Pujos, 1985, p. 563, pl. 2, fig. 1.
References: Biekart, 1989, pl. 1, fig. 4a, b; pl. 6, fig. 6a-c; pi. 7, fig. 2a, b.
110 K.-Y Wei et al. /Marine Micropaleontology 32 (1997) 95-114
I
la
lb
50
5b
17
18
6
7
0
19a
19b
220
22b
Plate III23a
23b
24a
4b
16
Optical micrographs of nannofossils from Core 90-36. Scale bar = 5 pm.
1. Gephyrocupsa oceanica Kamptner (large-sized, vertical bar). Subdepth = 62 cm. (a) Transmitted light. (b) Cross-polarized light. 2. Gephyrocapsa oceanica Kamptner (large-sized, diagonal bar). Subdepth = 62 cm. (a) Transmitted light. (b) Cross-polarized light, 3. Gephymcupsa oceanica Kamptner (medium-sized, vertical bar). Subdepth = 62 cm. (a) Transmitted light. (b) Cross-polarized light. 4. Gephyrocapsa oceanica Kamptner (medium-sized, diagonal bar). Subdepth = 62 cm. (a) Transmitted light. (b) Cross-polarized light. 5. Gephyrocapsa caribbennica Bordeaux and Hay. Subdepth = 62 cm. (a) Transmitted light, (b) Cross-polarized light,
6. Small Gephyrocapsa sp. Subdepth = 62 cm. Cross-polarized light.
7. Small Gephyrocapsa spp. (the small one = Gephyrocapsa ericsonii McIntyre and Be). Subdepth = 62 cm. Cross-polarized light, 8. Emiliania huxleyi (Lohmann) Hay and Mohler. Subdepth = 62 cm. Cross-polarized light.
9. Emiliania huxleyi (Lohmann) Hay and Mohler. Subdepth = 62 cm. Cross-polarized light. 10. Cricolithus jonesi Cohen. Subdepth = 62 cm. Cross-polarized light.
11. Reticulofenestru minutula (Gartner) Haq and Berggren. Subdepth = 62 cm. (a) Transmitted light, (b) Cross-polarized light. 12. Reticulofenestra haqii Backman. Subdepth = 62 cm. Cross-polarized light.
13. Reticulofenestra minuta Roth. Subdepth = 62 cm. Cross-polarized light. 14. Dictyococcites perplexu Burns. Subdepth = 62 cm. Cross-polarized light.
K.-Y: Wei et al. /Marine Micropaleontology 32 (I 997) 95-114 111 Syracosphaera
Gaarder and Heimtdal (1977) provided a detailed review of the systematic paleontology of this genus and lucid descriptions of species. We basically adopted their taxonomic scheme except that we followed Jordan and Young (1990) and Jordan and Kleijne (1994) in retaining all Coronosphaera species in the genus Syracosphaera.
and Paleoclimatic History of Late Neogene Sediments of the Northern Florida Continental Shelf. Ph.D. Diss., Texas A&M Univ., College Station, TX (unpubl.).
Chen, M.-P., 1979. Late Pleistocene calcareous nannoplankton in southern Okinawa Trough. Acta Oceanogr. Taiwan. 10, 95-l 18.
References
Chen, M.-P., Huang, C-K., 1995. The distribution of calcareous nannofossils and grain size in the surface sediments of the East China Sea and their relationship to the current pattern. TAO 6 (l), 129-150.
Ahagon, N., Tanaka, Y., Ujiie, H., 1993. Florisphaera profunda, a possible nannoplankton indicator of late Quaternary changes in sea-water turbidity at the northwestern margin of Pacific. Mar. Micropaleontol. 22, 255-273.
Backman, J., 1980. Miocene-Pliocene nannofossils and sedimen- tation rates in the Hatton-Rockall Basin, N.E. Atlantic Ocean. Stockholm Contrib. Geol. 36 (1), 91 pp.
Berger, W., 1977. Deep sea carbonate and the deglaciation preservation spike in pteropods and foraminifera. Nature 269, 301-304.
Chen, M.-I?, Shieh, K.-S., 1982. Recent nannofossil assemblages in sediments from Sunda Shelf to abyssal plain, South China Sea. Proc. Nat. Sci. Count. (Taiwan, ROC) A 6, 250-285. Cheng, X., 1992. Calcareous nannofossils in surface sediments
of the central and northern parts of the South China Sea. J. Micropalaeontol. 11, 167-176.
Biekart, J.W., 1989. The distribution of calcareous nannoplank- ton in late Quaternary sediments collected by the Snellius II Expedition in some southeast Indonesian basins. Proc. K. Ned. Akad. Wet. Ser. B 92 (2), 77-141.
Black, M., Bames, B., 1961. Coccoliths and discoasters from the floor of the South Atlantic Ocean. J. R. Microsc. Sot. 80 (2). 137-147.
Chinzei, K., Fujioka, K., Kitazato, H., et al., 1987. Postglacial environmental change of the Pacific Ocean off the coast of central Japan. Mar. Micropaleontol. 11, 273-291.
Cohen, C.L.D., 1965. Coccoliths and discoasters from Adriatic bottom sediments. Leidse Geol. Med. 35, 14.
Duplessy, J.C., Delibrias, G., Turon, J.L., Pujol, C., Dupart, J., 1981. Deglacial warming of the northeastern Atlantic Ocean: Correlation with the paleoclimatic evolution of the European continent. Palaeogeogr., Palaeoclimatol., Palaeoecol. 35, 121-
144.
Boyle, E.A., 1988. The role of vertical chemical fractionation in controlling Late Quatemary atmospheric carbon dioxide. J. Geophys. Res. 93, 15701-15714.
Bukry, D., 1973. Coccolith stratigraphy, eastern equatorial Pa- cific, Leg 16, Deep Sea Drilling Project. Init. Rep. DSDP 16, 653-711.
Fairbanks, R.A., 1989. A 17,000-year glacio-eustatic sea level record: Influence of glacial melting rates on the Younger Dryas event and deep sea circulation. Nature 342, 637-642. Gaarder, K.R., Heimdal, B.R., 1977. A revision of the
genus Syrucosphaeru Lohmann (Coccolithineae). ‘Meteor’ Forsch.-Ergeb. D 24, 54-71.
Chen, M.-P., 1978. Calcareous Nannoplankton Biostratigraphy
Plate III (continued)
15. Dictyococcites productus (Kamptner) Backman. Subdepth = 62 cm. Cross-polarized light.
16. Crenalithus sp. Subdepth = 62 cm. Cross-polarized light. 17. Syracosphaera hisrrica Kamptner. Subdepth = 62 cm. Cross- polarized light.
18. Syracosphaera pulchra Lohmann. Subdepth = 26 cm. Cross- polarized light.
19. Syracosphaera sp. Subdepth = 62 cm. (a) Transmitted light. (b) Cross-polarized light.
20. Syracosphaera sp. Subdepth = 23 cm. Cross-polarized light. 21. Syracosphaera lamina Lecal-Schlauder. Subdepth = 26 cm. Cross-polarized light.
22. Discosphaera tubqera (Murray and Blackman) Ostenfeld. Subdepth = 62 cm. (a) Transmitted light. (b) Cross-polarized light.
Herguera, J.C., 1992. Deep-sea benthic foraminiferal and bio- genie opal: Glacial to postglacial productivity changes in the western equatorial Pacific. Mar. Micropaleontol. 19, 79-98. Herguera, J.C., Berger, W.H., 1991. Paleoproductivity from ben-
thic foraminifera abundance: Glacial to postglacial change in the west-equatorial Pacific. Geology 19, 1173-l 176. Herguera, J.C., Berger, W.H., 1994. Glacial to postglacial drop
in productivity in the western equatorial Pacific: Mixing rate vs. nutrient concentrations. Geology 22, 629-632.
Hill, M.O., Gauch, H.G.Jr., 1980. Detrended correspondence analysis: An improved ordination technique. Vegetatio 42, 47-58.
Honjo, S., Okada, H., 1974. Community structure of coccol- ithophores in the photic layer of the mid-Pacific. Micropaleon- tology 20, 209-230.
Huang, C.-Y., Wu, S.-F., Zhao, M., Chen, M.-T., Wang, C.-H., Tu, X., Yuan, P.B., 1997. Last glacial to interglacial surface ocean variability in the South China Sea: A high-resolution record of sea-surface temperature, productivity, and South- east Asia monsoon. Mar. Micropaleontol. 32 (l-2) (this vol- ume).
23. Rhabodosphaera clavigeru Murray and Blackman. Subdepth Jordan, R.W., Kleijne, A., 1994. A classification system for liv- = 62 cm. (a) Transmitted light. (b) Cross-polarized light. ing coccolithophores. In: Winter, A., Siesser, W.G. (Ed%), 24. Rhabodosphaera clavigera Murray and Blackman. Subdepth Coccolithophores. Cambridge Univ. Press, Cambridge, pp. = 23 cm. (a) Transmitted light. (b) Cross-polarized light. 83-105.
112 K.-Y. Wei et al. /Marine Micropaleontology 32 (1997) 95-114 ‘: * *, +‘ * ;
4a
6a
?b8a
8b
12a
12%
l3a
3b
14a
14b
Plate IVt5a
15b
I6
l7a
l7b
Optical micrographs of nannofossils from Core 90-36. Scale bar = 5 pm.
1. Helicosphaera carter-i hyalina Gaarder. Subdepth = 23 cm. (a) Transmitted light. (b) Cross-polarized light.
2. Helicosphaera carteri wallichii (Lohmann) Okada and McIntyre. Subdepth = 62 cm. (a) ‘Transmitted light. (b) Cross-polarized light. 3. Helicosphaera carteri carteri, (Wallich) Kamptner. Subdepth = 26 cm. (a) Transmitted light. (b) Cross-polarized light.