Type of manuscript: Original article
Zopiclone use correlates with increased risk of hip fracture in older people: a case-control study in Taiwan
Shih-Wei Lai MD 1,2, Cheng-Li Lin MS 1,3, Wen-Chi Chen, DM and MS, Kuan-Fu Liao
DM and MS 4,
1College of Medicine, China Medical University and 2Department of Family Medicine, China
Medical University Hospital, Taichung, Taiwan
3Management Office for Health Data, China Medical University Hospital, Taichung, Taiwan 4Graduate Institute of Integrated Medicine, China Medical University and 5Department of
Internal Medicine, Taichung Tzu Chi General Hospital, Taichung, Taiwan
Corresponding author: Kuan-Fu Liao, Department of Internal Medicine, Taichung Tzu Chi General Hospital, No.66, Sec. 1, Fongsing Road, Tanzi District, Taichung City, 427, Taiwan Phone: 886-4-2205-2121; Fax: 886-4-2203-3986
E-mail: [email protected]
Running head: zopiclone and hip fracture Date 2015/May/18
Word count: 267 in abstract, 1694 in text, 2 tables and 22 references 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18
ABSTRACT
Objectives. The aim of this study was to explore whether zopiclone use correlates with increased risk of hip fracture in older people.
Design, Setting, Participants, Measurements. This case-control study was conducted to analyze the claim data from the Taiwan National Health Insurance Program. There were 10996 subjects aged ≥ 65 years with the first episode of hip fracture from 1998 to 2011 as the case group and 9081 randomly selected subjects aged ≥ 65 years without hip fracture matched with sex, comorbidities and index year of diagnosing hip fracture as the control group. Subjects whose last remaining one tablet for zopiclone was within 7 days before the date of diagnosing hip fracture were defined as current use of zopiclone. Subjects whose last remaining one tablet for zopiclone was ≥ 8 days before the date of diagnosing hip fracture were defined as late use of zopiclone. Subjects who never receive 1 prescription for zopiclone were defined as never use of zopiclone. A multivariable unconditional logistic regression model was used to estimate the odds ratio and 95% confidence interval to explore the correlation between zopiclone use and hip fracture.
Results. After adjustment for confounders, the multivariable logistic regression analysis demonstrated that the adjusted odds ratio of hip fracture was 3.56 for subjects with current use of zopiclone (95% confidence interval 2.33, 4.84), when compared with those with never use of zopiclone. The adjusted odds ratio decreased to 1.05 for those with late use of
19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37
zopiclone (95% confidence interval 0.94, 1.18), but without statistical significance.
Conclusions. Current use of zopiclone correlates with increased risk of hip fracture in older people. Clinicians should alert older people about the risk of hip fracture when prescribing zopiclone.
Keywords: hip fracture; older people; zopiclone 38
39 40 41 42
INTRODUCTION
Hip fracture is one of the most common disorders in older population worldwide and
represents a poor prognosis due to its easily complicated outcome including higher mortality and its impact on heavy caregivers’ burden. 1-3 In Nilson et al’s study in Sweden, the
incidence rates of hip fracture decreased from 514 to 363 per100000 persons in men aged 65–79 and decreased from 882 to 581 per100000 persons in women aged 65–79 since 1997 to 2009.4 In Chie et al’s study using the database of the Taiwan National Health Insurance
Program, the estimated incidence rates of hip fracture were 278.9 per 100000 persons in men aged 65–79 and 454.7 per100000 persons in women aged 65–79 during the period of 1996 to 2000.5 The incidence rates of hip fracture seem to be high in western and oriental
countries, but women more predominate.
To date, many risk factors including polypharmacy and some fall-risk-increasing drugs for treatment of insomnia, epilepsy, psychosis, Parkinson's disease, depression and chronic obstructive pulmonary disease have been found to be potentially associated with hip fracture.6-14 In addition, use of non-benzodiazepine hypnotic agents including zolpidem,
eszopiclone, and zaleplon, was found to be associated with increased risk of hip fracture among older people and nursing home residents No formal pharmacoepidemiological study focuses on the relationship between zopiclone use and hip fracture. Zopiclone, also a non-benzodiazepine hypnotic agent, is the second frequently used non-benzodiazepine hypnotic agent after zolpidem in Taiwan.15 Due to the same class of nonbenzodiazepine hypnotic
agents as zolpidem, eszopiclone, and zaleplon, we make a rational hypothesis that zopiclone use could be associated with increased risk of hip fracture in older people. If the association really exists, clinicians should keep in mind about the risk of hip fracture among older people with zopiclone use. Given extensive use of zopiclone in Taiwan, therefore, we conducted a population-based case-control study to explore this issue.
43 44 45 46 47 48 49 50 51 52 53 54 55 56 57 58 59 60 61 62 63 64 65 66 67
METHODS
Design and study population
A population-based case-control study was conducted to use the database from the Taiwan National Health Insurance Program. This program launched in March 1995 and covered over 99% of the total 23 million citizens living in Taiwan.16 The details of the program were well
written in previous papers. This study was approved by the Ethics Review Board of China Medical University and Hospital in Taiwan (CMU-REC-101-012).
Study subjects, other medications and comorbidities
Subjects aged 65 years or older with a first-episode of hip fracture according to International Classification of Diseases 9th Revision Clinical Modification (ICD-9 code 820) during the period of 1998-2011 were included as the case group. The index date for each case was defined as the date of diagnosing hip fracture. For each case of hip fracture, one control subject aged 65 years or older without hip fracture was randomly selected from the same database as the control group. To decreased biased results, subjects who had prescriptions of other non-benzodiazepinehypnotic agents including zolpidem, zaleplon and eszopiclone were excluded from this study. History of prescriptions for benzodiazepines available in Taiwan was included in this study. The number of medication use including oral medicine, topic use medicine and injection medicine was included in this study. Comorbidities potentially related to hip fracture before the index date were selected as follows: alcohol-related disease, cancer, chronic kidney disease, chronic obstructive pulmonary disease, diabetes mellitus, dementia, depression, hyperlipidemia, hypertension, osteoporosis, Parkinson's disease, as well as cardiovascular disease including coronary artery disease, heart failure,
cerebrovascular disease and peripheral atherosclerosis. All comorbidities were diagnosed with ICD-9 codes. Both case and control groups were matched with sex, benzodiazepines use, number of medication use, comorbidities and index year of diagnosing hip fracture. 68 69 70 71 72 73 74 75 76 77 78 79 80 81 82 83 84 85 86 87 88 89 90 91 92 93
Definition of zopiclone exposure
Based on the prescription history, we can measure the last remaining one tablet for zopiclone. Subjects whose last remaining one tablet for zopiclone was within 7 days before the date of diagnosing hip fracture or who still had zopiclone tablets at the date of diagnosing hip fracture were defined as current use of zopiclone. Subjects whose last remaining one tablet for zopiclone was ≥ 8 days before the date of diagnosing hip fracture were defined as late use of zopiclone. Subjects who never receive 1 prescription for zopiclone were defined as never use of zopiclone.
Statistical analysis
The distribution of sex, age, zopiclone use, benzodiazepines use, number of medication use, and comorbidities were compared between the case group and the control group using the Chi-square test and the fisher-exact test for categorized variables and the t-test for continuous variables. Variables found significantly related to hip fracture in the univariable
unconditional logistic regression model were further included in the multivariable
unconditional logistic regression model. The odds ratio (OR) and 95% confidence interval (CI) were estimated to explore the risk of hip fracture associated with zopiclone use. All data processing and statistical analyses were performed with the SAS software version 9.2 (SAS Institute, Inc., Cary, North Carolina, USA). A two-tailed P value of < 0.05 was considered statistically significant.
RESULTS
Characteristics of the study population
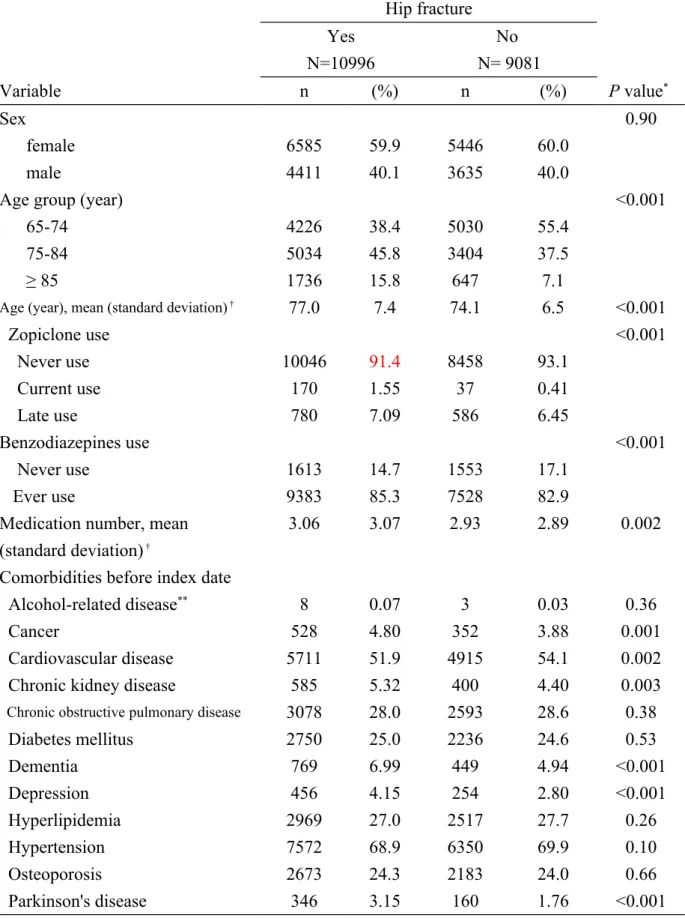
Table 1 presents the distributions of sex, age, zopiclone use, benzodiazepines use, number of medication use, and comorbidities between the case group and the control group. This study consisted of 10996 cases with hip fracture and 9081 controls without hip fracture, with similar distributions of sex and some comorbidities. The mean age (standard deviation) of the 94 95 96 97 98 99 100 101 102 103 104 105 106 107 108 109 110 111 112 113 114 115 116 117 118 119
study subjects was 77.0 ± 7.4 years for the case group and 74.1 ± 6.5 for the control group (t-test, P <0.001). The case group had higher prevalent rates of current use of zopiclone (1.55% vs. 0.41%), late use of zopiclone (7.09% vs. 6.45%), ever use of benzodiazepines (85.3% vs. 82.9%), cancer (4.80% vs. 3.88%), chronic kidney disease (5.32% vs. 4.40%), dementia (6.99% vs. 4.94%), depression (4.15% vs. 2.80%), and Parkinson's disease (3.15% vs. 1.76%) than the control group, with statistical significance. The mean number of medication use (standard deviation) of the study subjects was 3.06 ± 3.07 for the case group and 2.93 ± 2.89 for the control group (t-test, P= 0.002).
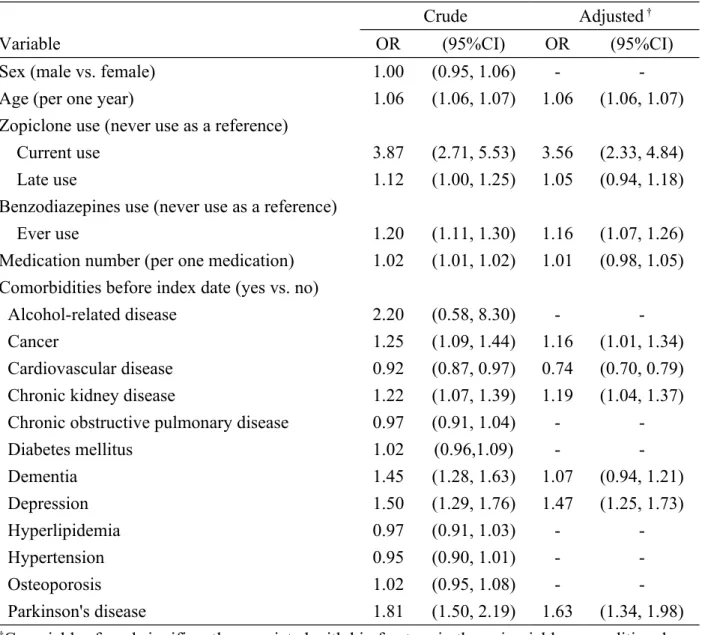
Risk of hip fracture associated with zopiclone use and other factors
Table 2 presents the risk of hip fracture associated with zopiclone use and other factors. After adjustment for potential confounding factors, the multivariable unconditional logistic
regression model presented that the adjusted odds ratio of hip fracture was 3.56 for subjects with current use of zopiclone (95% confidence interval 2.33, 4.84), when compared with those with never use of zopiclone. The adjusted odds ratio decreased to 1.05 for those with late use of zopiclone (95% confidence interval 0.94, 1.18), but without statistical
significance. In addition, age (per one year), benzodiazepines use, cancer, chronic kidney disease, depression, and Parkinson's disease were other factors significantly associated with hip fracture.
DISCUSSION
To the best of our knowledge, no formal pharmacoepidemiological study has evaluated the relationship between zopiclone use and the risk of hip fracture. In this population-based case-control study, we noted that current use of zopiclone was associated with 3.56–fold increased odds of hip fracture. The odds decreased to 1.05 for late use of zopiclone, but without statistical significance. These findings suggest that only people who continued to take 120 121 122 123 124 125 126 127 128 129 130 131 132 133 134 135 136 137 138 139 140 141 142 143
zopiclone would have the risk. The risk would decrease among those stopping use of zopiclone for 8 days or longer. In our previous study, polypharmacy (use 5 or more drugs) correlates with increased risk for hip fracture in older people.10 We made an additional
analysis. As a reference of people with never use of zopiclone and without polypharmacy, the adjusted odds ratio of hip fracture was 6.53 for people with current use of zopiclone and without polypharmacy (95% CI 3.80, 11.2). This means that zopiclone use has a unique effect on risk of hip fracture, independent of polypharmacy.
Zolpidem and zopiclone belong to 'Z' non-benzodiazepine hypnotic agents. Finkle et al reported that the fracture risk associated with zolpidem use exceeds that with alprazolam or lorazepam in older people.18 Wang et al reported that zolpidem use is associated with a
1.95-fold increased risk of hip fracture in older people (95%CI 1.09-3.51).6 The risk of hip fracture
associated with zopiclone use seems to be higher in our study than that associated with zolpidem use in Wang et al’s study (adjusted odds ratio 3.56 v.s. 1.95). The increased risk can be partially explained by the mechanism that zolpidem use is associated with balance
impairment, cognitive impairment, and falls. Although no direct evidence showing the relationship between zopiclone use and falls, similarly, zopiclone use is associated with impaired balance, impaired cognition and impaired psychomotor performance. One or combination of these impairments may more likely lead to falls and even hip fracture in older people with zopiclone use. However, further research is needed to explore the biologic mechanism by which zopiclone use causes hip fracture.
Some important limitations should be discussed in this study. First, due to natural limitation of the claim database used, we did not know whether patients really took zopiclone or not. 144 145 146 147 148 149 150 151 152 153 154 155 156 157 158 159 160 161 162 163 164 165
We used zopiclone prescriptions for instead. Second, theoretically, we should conduct a case-control study which consisted of subjects with zopiclone use only and subjects without using any agent. It is considerably difficult to include this kind of rigorous criteria for study, particularly using the claim database. Third, clinically, some fall-risk-increasing drugs for treatment of epilepsy, psychosis, Parkinson's disease, depression and chronic obstructive pulmonary disease are found to be potentially associated with hip fracture. Therefore, it seems to be relatively difficult to include all concomitant offending drugs for analysis. It is also relatively difficult to differentiate which drug is the most probable key one. The next more rational step is to focus on one potential drug to estimate its relative risk on hip fracture. That is why we only include zopiclone use for analysis. Fourth, because there are not specific ICD-9 codes for falls, gait and balance disorders, we can not include these disorders for analysis.
Some strengths of this study can be documented. This study included a large sample size to increase its statistical power. The study design and results seem to be reasonable. It has an important clinical implication and provides the updated evidence to this issue.
We conclude that current use of zopiclone is associated with an increased risk of hip fracture in older people. Clinicians should alert older people about the risk of hip fracture when prescribing zopiclone. 166 167 168 169 170 171 172 173 174 175 176 177 178 179 180 181 182 183
ACKNOWLEDGMENTS
Conflict of Interest Statement
The authors disclose no conflicts of interest.
Conflict of Interest Disclosures:
Elements of Financial/Personal Conflicts *Author 1 Shih-Wei Lai Author 2 Cheng-Li Lin Author 3 Kuan-Fu Liao Author 4
Yes No Yes No Yes No Yes No
Employment or Affiliation √ √ √ Grants/Funds √ √ √ Honoraria √ √ √ Speaker Forum √ √ √ Consultant √ √ √ Stocks √ √ √ Royalties √ √ √ Expert Testimony √ √ √ Board Member √ √ √ Patents √ √ √ Personal Relationship √ √ √
*Authors can be listed by abbreviations of their names. For “yes” x mark(s): give brief explanation below:
This study is supported in part by Taiwan Ministry of Health and Welfare Clinical Trial and 184 185 186 187 188 189 190 191 192 193
Research Center of Excellence (MOHW104-TDU-B-212-113002), China Medical University Hospital, Academia Sinica Taiwan Biobank, Stroke Biosignature Project (BM104010092), NRPB Stroke Clinical Trial Consortium (MOST 103-2325-B-039 -006), Tseng-Lien Lin Foundation in Taichung in Taiwan, Taiwan Brain Disease Foundation in Taipei in Taiwan, and Katsuzo and Kiyo Aoshima Memorial Funds in Japan. These funding agencies did not influence the study design, data collection and analysis, decision to publish, or preparation of the manuscript..
Author Contributions:
Shih-Wei Lai substantially contributed to the conception of the article. He planned and conducted this study. He initiated the draft of the article and critically revised the article. Cheng-Li Lin conducted the data analysis and critically revised the article.
Kuan-Fu Liao planned and conducted this study. He participated in the data interpretation and also critically revised the article.
Sponsor’s Role: None.
194 195 196 197 198 199 200 201 202 203 204 205 206 207 208 209
REFERENCES
1. Hung LW, Tseng WJ, Huang GS, Lin J. High short-term and long-term excess mortality in geriatric patients after hip fracture: a prospective cohort study in Taiwan. BMC
Musculoskelet Disord 2014;15:151.
2. Lin PC, Lu CM. Hip fracture: family caregivers' burden and related factors for older people in Taiwan. J Clin Nurs 2005;14:719-26.
3. Sterling RS. Gender and race/ethnicity differences in hip fracture incidence, morbidity, mortality, and function. Clin Orthop Relat Res 2011;469:1913-8.
4. Nilson F, Moniruzzaman S, Gustavsson J, Andersson R. Trends in hip fracture incidence rates among the elderly in Sweden 1987-2009. J Public Health (Oxf) 2013;35:125-31.
5. Chie WC, Yang RS, Liu JP, Tsai KS. High incidence rate of hip fracture in Taiwan: estimated from a nationwide health insurance database. Osteoporos Int 2004;15:998-1002. 6. Wang PS, Bohn RL, Glynn RJ, Mogun H, Avorn J. Zolpidem use and hip fractures in older people. J Am Geriatr Soc 2001;49:1685-90.
7. Marks R, Allegrante JP, Ronald MacKenzie C, Lane JM. Hip fractures among the elderly: causes, consequences and control. Ageing Res Rev 2003;2:57-93.
8. French DD, Campbell R, Spehar A, Cunningham F, Foulis P. Outpatient medications and hip fractures in the US: a national veterans study. Drugs Aging 2005;22:877-85.
9. Benetos IS, Babis GC, Zoubos AB, Benetou V, Soucacos PN. Factors affecting the risk of hip fractures. Injury 2007;38:735-44.
10. Lai SW, Liao KF, Liao CC, Muo CH, Liu CS, Sung FC. Polypharmacy correlates with increased risk for hip fracture in the elderly: a population-based study. Medicine (Baltimore) 2010;89:295-9.
11. Lan TY, Hou SM, Chen CY, et al. Risk factors for hip fracture in older adults: a case-control study in Taiwan. Osteoporos Int 2010;21:773-84.
12. Berry SD, Lee Y, Cai S, Dore DD. Nonbenzodiazepine sleep medication use and hip fractures in nursing home residents. JAMA Intern Med 2013;173:754-61.
13. Rossini M, Viapiana O, Adami S, et al. Medication use before and after hip fracture: a population-based cohort and case-control study. Drugs Aging 2014;31:547-53.
14. Lin FY, Chen PC, Liao CH, Hsieh YW, Sung FC. Retrospective population cohort study on hip fracture risk associated with zolpidem medication. Sleep 2014;37:673-9.
15. Hsiao F-Y, Hsieh P-H, Gau C-S. Ten-year trend in prescriptions of z-hypnotics among the elderly: A nationwide, cross-sectional study in Taiwan. Journal of Clinical Gerontology and Geriatrics 2013;4:37-41.
16. Database NHIR. Taiwan. http://nhird.nhri.org.tw/en/Backgroundhtml [cited in 2015 April]. 210 211 212 213 214 215 216 217 218 219 220 221 222 223 224 225 226 227 228 229 230 231 232 233 234 235 236 237 238 239 240 241 242 243 244 245 246
17. Hung SC, Liao KF, Lai SW, Li CI, Chen WC. Risk factors associated with symptomatic cholelithiasis in Taiwan: a population-based study. BMC Gastroenterol 2011;11:111.
18. Finkle WD, Der JS, Greenland S, et al. Risk of fractures requiring hospitalization after an initial prescription for zolpidem, alprazolam, lorazepam, or diazepam in older adults. J Am Geriatr Soc 2011;59:1883-90.
19. Frey DJ, Ortega JD, Wiseman C, Farley CT, Wright KP, Jr. Influence of zolpidem and sleep inertia on balance and cognition during nighttime awakening: a randomized placebo-controlled trial. J Am Geriatr Soc 2011;59:73-81.
20. Kolla BP, Lovely JK, Mansukhani MP, Morgenthaler TI. Zolpidem is independently associated with increased risk of inpatient falls. J Hosp Med 2013;8:1-6.
21. Mets MA, de Vries JM, de Senerpont Domis LM, Volkerts ER, Olivier B, Verster JC. Next-day effects of ramelteon (8 mg), zopiclone (7.5 mg), and placebo on highway driving performance, memory functioning, psychomotor performance, and mood in healthy adult subjects. Sleep 2011;34:1327-34.
22. Gustavsen I, Hjelmeland K, Bernard JP, Morland J. Individual psychomotor impairment in relation to zopiclone and ethanol concentrations in blood--a randomized controlled double-blinded trial. Addiction 2012;107:925-32.
247 248 249 250 251 252 253 254 255 256 257 258 259 260 261 262 263 264 265
Table 1. Characteristics of cases with hip fracture and controls in older people Hip fracture Yes N=10996 No N= 9081 Variable n (%) n (%) P value* Sex 0.90 female 6585 59.9 5446 60.0 male 4411 40.1 3635 40.0
Age group (year) <0.001
65-74 4226 38.4 5030 55.4
75-84 5034 45.8 3404 37.5
≥ 85 1736 15.8 647 7.1
Age (year), mean (standard deviation) † 77.0 7.4 74.1 6.5 <0.001
Zopiclone use <0.001 Never use 10046 91.4 8458 93.1 Current use 170 1.55 37 0.41 Late use 780 7.09 586 6.45 Benzodiazepines use <0.001 Never use 1613 14.7 1553 17.1 Ever use 9383 85.3 7528 82.9
Medication number, mean (standard deviation) †
3.06 3.07 2.93 2.89 0.002
Comorbidities before index date
Alcohol-related disease** 8 0.07 3 0.03 0.36
Cancer 528 4.80 352 3.88 0.001
Cardiovascular disease 5711 51.9 4915 54.1 0.002
Chronic kidney disease 585 5.32 400 4.40 0.003
Chronic obstructive pulmonary disease 3078 28.0 2593 28.6 0.38
Diabetes mellitus 2750 25.0 2236 24.6 0.53 Dementia 769 6.99 449 4.94 <0.001 Depression 456 4.15 254 2.80 <0.001 Hyperlipidemia 2969 27.0 2517 27.7 0.26 Hypertension 7572 68.9 6350 69.9 0.10 Osteoporosis 2673 24.3 2183 24.0 0.66 Parkinson's disease 346 3.15 160 1.76 <0.001
Data are presented as the number of subjects in each group, with percentages given in parentheses.
Table 2. Odds ratio and 95% confidence interval of hip fracture associated with zopiclone use and comorbidities in older people
Crude Adjusted †
Variable OR (95%CI) OR (95%CI)
Sex (male vs. female) 1.00 (0.95, 1.06) -
-Age (per one year) 1.06 (1.06, 1.07) 1.06 (1.06, 1.07)
Zopiclone use (never use as a reference)
Current use 3.87 (2.71, 5.53) 3.56 (2.33, 4.84)
Late use 1.12 (1.00, 1.25) 1.05 (0.94, 1.18)
Benzodiazepines use (never use as a reference)
Ever use 1.20 (1.11, 1.30) 1.16 (1.07, 1.26)
Medication number (per one medication) 1.02 (1.01, 1.02) 1.01 (0.98, 1.05) Comorbidities before index date (yes vs. no)
Alcohol-related disease 2.20 (0.58, 8.30) -
-Cancer 1.25 (1.09, 1.44) 1.16 (1.01, 1.34)
Cardiovascular disease 0.92 (0.87, 0.97) 0.74 (0.70, 0.79)
Chronic kidney disease 1.22 (1.07, 1.39) 1.19 (1.04, 1.37)
Chronic obstructive pulmonary disease 0.97 (0.91, 1.04) -
-Diabetes mellitus 1.02 (0.96,1.09) - -Dementia 1.45 (1.28, 1.63) 1.07 (0.94, 1.21) Depression 1.50 (1.29, 1.76) 1.47 (1.25, 1.73) Hyperlipidemia 0.97 (0.91, 1.03) - -Hypertension 0.95 (0.90, 1.01) - -Osteoporosis 1.02 (0.95, 1.08) - -Parkinson's disease 1.81 (1.50, 2.19) 1.63 (1.34, 1.98)
†Covariables found significantly associated with hip fracture in the univariable unconditional
logistic regression model were further examined by the multivariable unconditional logistic regression model.
Additionally adjusted for age, benzodiazepines use, medication number, cancer, cardiovascular disease, chronic kidney disease, dementia, depression, and Parkinson's disease
267