ARTICLE NO.RC989017
Characterization of Three Endogenous Peptide Inhibitors
for Multiple Metalloproteinases with Fibrinogenolytic
Activity from the Venom of Taiwan Habu
(Trimeresurus mucrosquamatus)
Kai-Fa Huang,* Chin-Chun Hung,* Shih-Hsiung Wu,*
,† and Shyh-Horng Chiou*
,†
,1†Institute of Biochemical Sciences, National Taiwan University, Taipei; and *Institute of Biological Chemistry,
P.O. Box 23-106, Academia Sinica, Taipei, Taiwan
Received May 23, 1998
talloproteinases present in crotalid and viperid venoms.
Three small peptide components were isolated and
q 1998 Academic Presspurified from the venom of Taiwan habu
(Trimere-surus mucrosquamatus), which show specific
activ-ity to inhibit the strong proteolytic activactiv-ity of
multiple metalloproteinases present in the crude
Venoms of the snake families of Viperidae and
Cro-venom. Using multiple chromatographies coupled
talidae cause shock, intravascular clotting, systemic
with successive ultrafiltrations, three inhibitors,
and local hemorrhage, edema and necrosis upon
victim-i.e. pyroglutamate-lysine-tryptophan (pyroGlu-Lys-Trp),
ized prey. It has also been well known that snake
ven-pyroglutamate-asparagine-tryptophan
(pyroGlu-Asn-oms contain complex mixtures of pharmacologically
ac-Trp) and pyroglutamate-glutamine-tryptophan
(pyro-tive peptides and proteins including some potent
pro-Glu-Gln-Trp) were obtained in good yields and high
teolytic enzymes (1-3). The common symptom of
homogeneity. The yields of these peptide fractions
hemorrhage caused by snakebites is usually the result
were estimated to be about 0.65 mg, 0.55 mg and 0.42
of structural destruction of capillary basement
mem-mg from 250 mem-mg total lyophilized crude venom, which
branes via proteolytic degradation by these proteases
corresponded to the approximate concentrations of 8.4
(4). Death is usually the result of the combined effects
mM, 7.3 mM and 5.4 mM respectively in venom
secre-of several components in the venom. In order to avoid
tion. Detailed and unambiguous structural
determina-the auto-digestion of venom gland by its own
degrada-tion was established by amino acid analyses, mass
spectrometry and microsequencing of purified pep-
tive proteases present in gland secretions, snake
ven-tides. Further functional characterization of these
oms of various species have been reported to contain
three tripeptides showed that they could weakly in-
some self-defensive enzyme inhibitors such as serine
hibit three metalloproteinases previously isolated
protease inhibitors (5-7), citrate and small peptides (8),
from the same venom. The inhibitory activities were
that can selectively bind to these proteases thereby
similar among these tripeptides and their IC
50(concen-
partially inhibit and minimize their proteolytic
activi-tration for 50% inhibition) were estimated in a range
ties in crude venoms. Venom proteinases so far
charac-of 0.20-0.95 mM, which is much more effective than
terized have been shown to be a heterologous group of
citrate, another venom protease inhibitor of low mo-
proteins, generally belonging to two major classes of
lecular-weight component. Since these tripeptides are
proteases, i.e. thrombin-like enzymes and
metallopro-the endogenous peptide inhibitors present in metallopro-the lu-
teinases with a wide range of molecular masses (9-11).
men of venom glands, it is conceivable that they may
Early studies have indicated that, a few snake
spe-act as a self-defensive mechanism against the
auto-di-cies of Viperidae and Crotalidae families contained
en-gestive deleterious effect of the strong
metalloprotein-dogenous small peptides present as a form of
pyrogluta-ases in vivo, particularly several zinc-dependent
me-mate(pyroGlu)-containing tripeptides, e.g.
pyroGlu-Asn-Trp and pyroGlu-Gln-Trp (12,13). These small
N-terminally blocked peptides were later found in
venoms of various species of snakes, and shown to be
1Corresponding address: Institute of Biological Chemistry,
Acade-relatively good inhibitors of zinc-dependent
metallopro-mia, P.O. Box 23-106, Taipei, Taiwan. Fax: (886)-2-3635038. E-mail:Vol. 248, No. 3, 1998 BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS
Cleavage of the N-terminal pyroglutamate from peptides and
se-concentration present in various snake venoms of
quence analysis. Cleavage of the N-terminal pyroglutamate from
Crotalidae and Viperidae families (16), has also been
peptides was accomplished using sequencing grade pyroglutamate
identified as a low molecular-weight component in
aminopeptidase (Boehringer Mannheim). About 5-10 mg peptidescrude venom to play a role as an endogenous inhibitor
were dissolved in 45ml digestion buffer (100 mM sodium phosphate, 10 mM EDTA, 5 mM dithiothreitol and 5% glycerol, pH 8.0). Afterof venom enzymes and proteinases (17). Therefore it
addition of 5ml pyroglutamate aminopeptidase (0.25mg/ml) and
incu-would be of interest to compare the inhibitory activities
bation for 18 hours at 47C first and then 4 hours at 25 7C, peptides in
of these small endogenous components in snake
ven-reaction mixture were separated by reverse-phase high performance
oms on the proteinases isolated from the same venom
liquid chromatography. Sequencing of deblocked peptides wascar-in order to provide some car-insights car-into the protracted
ried out by automated Edman degradation with a microsequencingsequenator (Model 477A, Applied Biosystems) as described (21).
issue on how snakes develop the self-defensive
mecha-nism in protecting the venom gland itself from the dam-
Determination of protein and peptide concentration. Proteincon-aging effect of its secreted metalloproteinases. Pre-
centration was determined by dye-binding assays (22) or estimated from the absorbance of the purified protein at 280 nm, using anviously we have identified three metalloproteinases
extinction coefficient (E1%) of 10. Concentrations of
tryptophan-con-with strong fibrinogenolytic activity from the Taiwan
taining tripeptides were measured by absorbance at 280 nm using
habu (Trimeresurus mucrosquamatus), designated as
a molar extinction coefficient of 5600 M01cm01.TM-1, TM-2, and TM-3 (18,19). In this report we have
FTC-caseinolytic activity assay of metalloproteinases and its
inhi-focused on the isolation and characterization of three
bition by peptides and citrate. Caseinolytic activity of
metallopro-pyroglutamate tripeptides and compare their inhibi-
teinases was measured using a fluorescence substrate, FTC-caseintory activities with citrate ion on these three metallo-
as reported previously (23), with or without the addition of smallproteinases. One novel tripeptide, pyroGlu-Lys-Trp,
peptide inhibitors or sodium citrate. Reaction mixtures, containing 5ml enzyme (0.2-0.4mg/ml) and 5ml of the indicated concentrationshas for the first time been identified and shown to be
of inhibitor fractions (or replacing this with the assay buffer, 100 mM
a genuine inhibitor for these metalloproteinases.
Tris.HCl/10 mM CaCl2, pH 8.0, as positive control), was incubated at
room temperature for 15 min, followed by adding 5ml FTC-casein (10 mg/ml in 50 mM Tris.HCl, pH 7.2) and 35ml of the assay buffer
MATERIALS AND METHODS
for a prolonged incubation at 377C for 70 min. Proteolysis was termi-nated by adding 120 ml of 5% TCA and mixing extensively. The
Materials. Lyophilized venom powder was obtained from the
lo-reaction mixture was allowed to stand at room temperature for 1 h cal snake farm. The substrate FTC-casein was purchased from Sigma
and the TCA-insoluble protein was sedimented by centrifugation at Chemical Company (St. Louis, MO). Gel suspensions of TSK
DEAE-13,000 rpm for 2.5 min. A 90-ml aliquot of the supernatant fraction 650(M) anion-exchange resin were purchased from Merck
(Darms-was diluted with 0.5 M Tris buffer, pH 8.5, with vigorous mixing to tadt, Germany).
ensure the entire sample was at the proper pH. Fluorescence was
Isolation and purification of small peptides from crude venom. An measured using an excitation wavelength at 490 nm and an emission
anion-exchange chromatography on an open column (2.5 1 46 cm) wavelength at 522 nm on a Hitachi’s F-4010 fluorescence spectropho-packed with TSK DEAE-650(M) gel suspension was employed to tometer. Percent inhibition for the caseinolytic activity of metallopro-separate small peptide components. Venom powder dissolved in teinases was estimated from the fluorescence yields of assayed sam-0.025 M ammonium bicarbonate, pH 7.8 starting buffer (total 5 ml) ples as compared with that of control.
was applied to the column and then eluted in a linear gradient of 0.025-0.5 M ammonium bicarbonate, followed by 0.5-1.0 M
ammo-nium bicarbonate, pH 8.0 buffer, similar to those described pre-
RESULTS AND DISCUSSION
viously (18). Two fractions rich in small peptides were obtained fromthe above chromatography and ultrafiltered through membranes
(YM-10, Amicon) and the flow-throughs were collected and lyophi-
The venoms of various snakes have been shown to
lized. Reverse-phase chromatography was performed on a Hitachi’spossess very strong and stable proteolytic enzymes
liquid chromatograph using C18column (0.46 1 25 cm, Vydac).Elu-with fibrinolytic or fibrinogenolytic activity, notably in
tion was carried out in a linear gradient of 5-95% acetonitrile in 0.1%the snake families of Crotalidae and Viperidae (24-26).
TFA and the eluates were monitored at either 214 nm or 280 nm.Major fractions eluted from the column were collected and lyophi-
Currently several proteolytic enzymes of snake origin
lized.
have been used clinically as potential antithrombotic
agents (27-30) with the special concern on how to
modu-SDS – polyacrylamide gel electrophoresis and amino acid analyses.The purity of purified polypeptides was checked by SDS-polyacryl-
late and mitigate unwanted toxic effects of local and
amide slab gel electrophoresis (5% stacking/15% resolving gel) assystemic hemorrhage or bleeding caused by these
pro-described (20) with some modifications (i.e., 5% cross-linking N, N*-teinases. Most venom proteases with such hemorrhagic
methylenebisacrylamide in the gel solution). The amino acidcompo-side effects consist mainly of metalloproteinases (31).
sitions were determined with a Beckman 6300 amino acid analyzerusing a single-column system based on a conventional ion-exchange
In this study we have made an effort to look for natural
chromatography system.proteinase inhibitors present in the crude venom of
Taiwan habu and their effects on three strong
hemor-Mass spectrometry analyses. Fast atom bombardment (FAB)
mass spectra were obtained on an Autospec mass spectrometer (Mi-
rhagic metalloproteinases isolated previously from the
cromass, UK) fitted with a cesium ion gun operated at 25 kV. Samplessame venom (18,19). Successful identification and
char-were dissolved in 5% acetic acid for loading onto the probe tip coatedacterization of three pyroglutamate tripeptides could
with monothioglycerol as matrix. Multiple scans at 6 seconds peraccount for the resistance of snake venom components
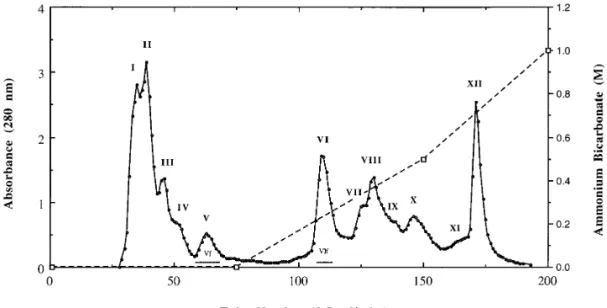
decay were acquired and summed for data processing using the OpusFIG. 1. Isolation of small peptides from the crude venom of Taiwan habu (Trimeresurus mucrosquamatus) on TSK DEAE-650(M) column. About 250 mg of lyophilized venom powder was dissolved in the starting buffer of 0.025 M ammonium bicarbonate, pH 7.8, and applied to the column equilibrated with the same buffer. Elution was carried out in three steps similar to that described in the previous report (18,19) with some modifications. The column eluates (3.5 ml/tube) were monitored for absorbance at 280 nm. Fractions I-XII, as indicated, were collected and ultrafiltered through YM-10 Amicon membrane, the flowthroughs were collected, and lyophilized for further analysis and purification. The bars marked with Vf and VIf represent the flowthrough fractions obtained after ultrafiltrations through membrane.
Isolation and purification of peptides from crude
twelve fractions and ultrafiltered through membranes
with a molecular weight cut-off of 10,000. The
flow-venom of Taiwan habu.
Fractionation of T.
mucros-quamatus venom on a TSK DEAE-650 anion-exchange
throughs from ultrafiltration were collected,
concen-trated and examined for their UV and fluorescence
column (Fig. 1) using ammonium bicarbonate as
elu-tion buffer yielded an eluelu-tion pattern superior to that
emission spectra. Flow-through fractions Vf
and VIf
,
obtained from fractions V and VI in Fig. 1, which were
reported previously (19). It is to be noted that fractions
I-V, eluted at the initial buffer, contained multiple pro-
eluted by the initial buffer and the first-step gradient
respectively, were shown to have a high absorbance at
teolytic enzymes with molecular weights of about
24,000 as judged by SDS-gel electrophoresis (data not
280 nm and an emission maximum of about 358 nm
detected on a Hitachi F-4010 fluorescence
spectropho-shown), corresponding to those of metalloproteinases
reported by our group (19). Fractions VI-X, eluted at
tometer when excited at 295 nm.
High performance liquid chromatographies (HPLC) of
the first-step gradient, possessed also multiple
protein-ases with molecular weights in a range of 27,000 to
these two flow-throughs were further carried out on a
Hitachi liquid chromatograph (Fig. 2) in order to purify
32,000, corresponding to a family of serine proteases
described in the previous report (32). We collected
these fractions in a homogeneous form. HPLC showed
TABLE 1
Amino Acid Compositions, Sequences, and Fluorescence Emission Wavelengths of Small Peptide Fractions
Compositiona Sequence residued
lemic Asx Glx Lys Trp (nm) 2nd 3rd Vf — 44.6% 45.0% n.d.b 358.2 Lys Trp VIf-a 46.8% 50.2% — n.d. 358.0 Asn Trp VIf-b — 95.5% — n.d. 358.0 Gln Trp L-Trp 354.4
aData of amino acid analyses are expressed as mol%, and V
f, VIf-a and VIf-b are fractions collected from reverse-phase HPLC.
bn.d., not determined.
clemi, the emission wavelength with the highest fluorescence yield using excitation at 295 nm. L-Trp is used as the standard for
wavelength comparison.
dThe N-terminal first residue was blocked and the second and third residues were determined by the treatment of peptides with
Vol. 248, No. 3, 1998 BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS
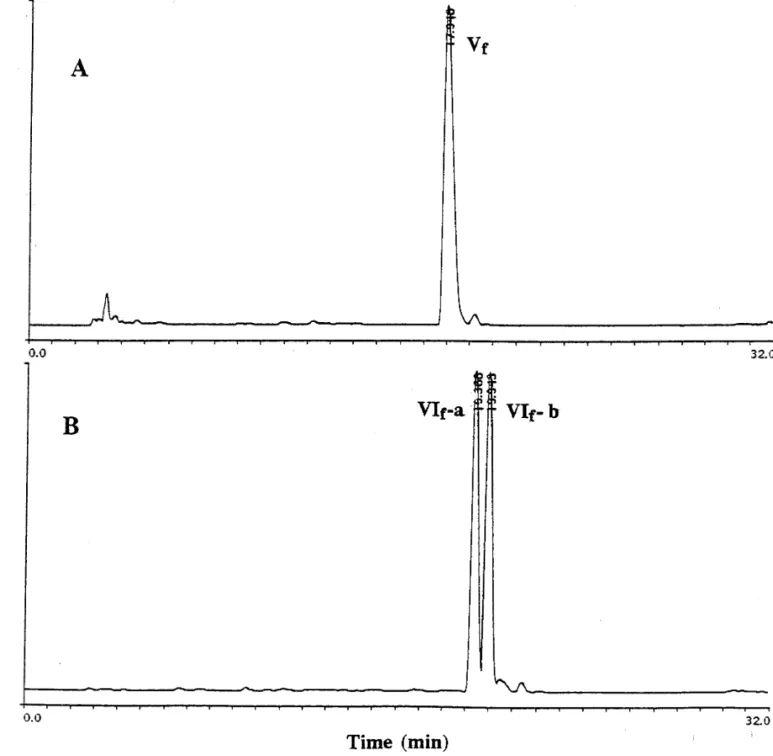
FIG. 2. High-performance liquid chromatographies (HPLC) on a reverse-phase C18column of fractions Vf and VIf obtained from TSK
DEAE-650(M) column after ultrafiltrations. About 50ml (2 mg/ml) of fraction Vf (A) or fraction VIf (B) was injected into a C18column
equilibrated with 5% acetonitrile in 0.1% TFA, and the bound materials were eluted with a linear gradient of 5-95% acetonitrile in 0.1% TFA for 30 min. After HPLC the fraction VIf eluted as two peaks of equal amounts, denoted as VIf-a and VIf-b respectively as indicated in (B), whereas fraction Vf eluted as one major homogeneous peak (A).
that fraction VIf
can be split into two peaks at elution
in peak VIf-a and VIf-b produced molecular ions with
times of 19.4 and 19.9 min with equal yields, denoted as
a mass/charge ratio of about 430 and 444 (Fig. 3),
corre-VIf-a and VIf-b respectively (Fig. 2B). Fraction Vf
was
sponding to those molecular masses of
pyroGlu-Asn-eluted as one major peak (Fig. 2A).
Trp and pyroGlu-Gln-Trp respectively. These two
pyro-glutamate tripeptides are identical to those isolated
previously from snakes of A. halys blomhoffii, B.
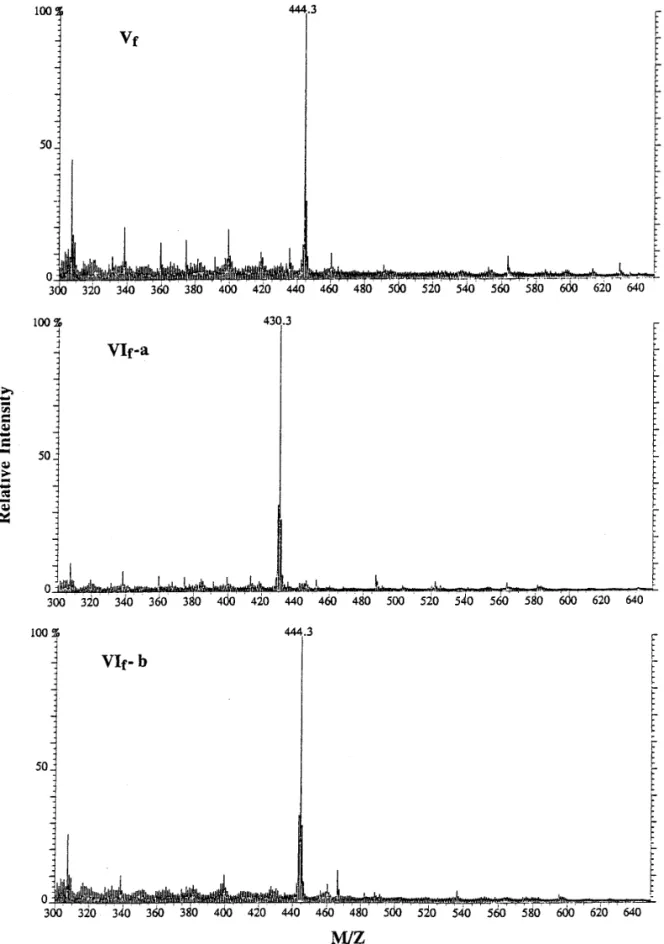
jara-Amino acid compositions and mass spectrometry.
raca, C. adamanteus and T. flavoviridis (12). On the
Amino acid analyses showed that peak Vf
contained
other hand, the molecular ion emitted from the sample
equal amounts of Glx (Glu or Gln) and Lys. Likewise,
of peak Vf
showed a mass/charge ratio of about 444
peak VIf-a contained Glx and Asx (Asp or Asn), however
(Fig. 3), corresponding to a peptide of pyroGlu-Lys-Trp,
only Glx was identified in peak VIf-b (Table 1).
FIG. 3. Mass spectrometry of small peptides in peaks Vf, VIf-a and VIf-b from HPLC. Approximately 1mg of each peptide sample dissolved in 5% acetic acid was analyzed by mass spectrometry (FAB-MS). The X-axis designates the mass/charge ratio, which is representative of the molecular weights of these peptides. The number above the major peak in each spectrum shows the measured molecular weight of major molecular ion peak by FAB-MS.
reported before. It is concluded that these small pep-
judged by the fluorescence spectra and
microsequenc-ing after deblockmicrosequenc-ing of the N-terminal residue (Table
tides are all composed of three amino acids and
Vol. 248, No. 3, 1998 BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS
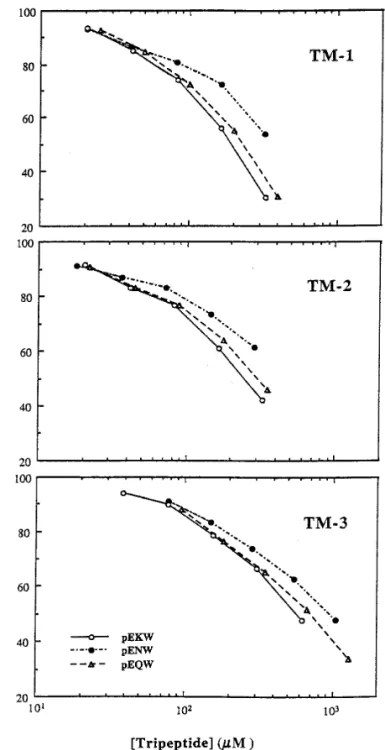
same venom of Taiwan habu, were employed to
in-vestigate the inhibitory activities of these tripeptide
samples using a fluorescence substrate, fluorescein
thiocarbamoyl (FTC)-casein. In the absence of
tri-peptides, FTC-casein was almost completely
hy-drolyzed after incubation with metalloproteinases at
37
7C for 2 h, whereas the FTC-caseinolytic activity
of metalloproteinases was dose-dependently
inhib-ited by increasing the tripeptide inhibitors. As
shown in Fig. 4, there are no much activity difference
among three pyroglutamate tripeptides to inhibit
each metalloproteinase within the concentration
range used in this study. The IC50
(concentration for
50% inhibition) of these tripeptides for TM-1, TM-2
and TM-3 were estimated to be 0.20 mM, 0.25 mM
and 0.58 mM for pyroGlu-Lys-Trp, respectively; 0.37
mM, 0.46 mM and 0.95 mM for pyroGlu-Asn-Trp,
respectively; and 0.24 mM, 0.31 mM and 0.71 mM
for pyroGlu-Gln-Trp, respectively. These IC50
values
are generally weaker than most conventional
prote-ase inhibitors of larger sizes such as trypsin or
chy-motrypsin inhibitors. However they still exhibit the
high specificity for these venom metalloproteinases
and not the thrombin-like serine proteases present
in the same venom (32, data not shown). Moreover
when comparing IC50
of these tripeptides with that
of citrate ion (17), another endogenous proteinase
inhibitor present in snake venoms, these tripeptides
are actually 100-fold more effective than citrate. It
is also noteworthy that TM-1 and TM-2 appear to
be slightly more susceptible to tripeptide inhibitors
than TM-3, which corroborates our previous
conclu-sion that TM-1 and TM-2 are more closely related
to each other than to TM-3, and both of them are
distinguishable from TM-3 in their partial protein
sequences (19).
In conclusion we have isolated and characterized
three endogenous tripeptides which show specific
in-hibition against the strong metalloproteinases
pres-ent in the same venom. Characterization of these
FIG. 4. Dose-dependent inhibition of T. mucrosquamatus
metal-pyroglutamate tripeptides may provide a molecular
loproteinase activities by three pyroglutamate tripeptides. Theper-basis to account for the resistance of snake venom
centage inhibition of FTC-caseinolytic activity (1 mg/ml) bytripep-components to these metalloproteinases in vivo.
Fur-tides on TM-1, TM-2 and TM-3 metalloproteinases isolated from T.mucrosquamatus venom (18,19), was measured and estimated ac-
ther elucidation of the mechanistic aspects of these
cording to that as described in Materials and Methods. Incubation
tripeptides in relation to the inhibition of venom
me-of FTC-casein substrates with each metalloproteinase isme-oform in thetalloproteinases could open avenues for exploring
absence of tripeptides was used as control for 100% FTC-caseinolyticthe potential of using these peptide inhibitors as
activity and each data point was the average of two experiments.modulators in alleviating the toxic side effect of
bleeding or hemorrhage associated with several
venom protease-based antithrombotic agents.
VIf-b and Vf
from 250 mg crude venom were estimated
to be about 0.65 mg, 0.55 mg and 0.42 mg, respectively,
REFERENCES
using a molar extinction coefficient for tryptophan of
5600 M
01cm
01at 280 nm.
1. Jimenez-Porras, J. M. (1968) Ann. Rev. Pharmacol. 8, 299 – 318.
Inhibition of metalloproteinase activities by tripep-
2. Markland, F. S. Jr. (1988) in Hemostasis and Animal Venomstide inhibitors.
Three metalloproteinase isoforms,
(Pirkle, H., and Markland, F. S., Eds.), pp. 149 – 172, Dekker,New York.
TM-1, TM-2 and TM-3 isolated previously from the
3. Tu, A. T. (1982) in Rattlesnake Venoms: Their Actions and Treat- 18. Huang, K.-F., Hung, C.-C., and Chiou, S.-H. (1993) Biochem.
Mol. Biol. International 31, 1041 – 1050.
ment (Tu, A. T., Ed.), pp. 247 – 312, Dekker, New York.
4. Ownby, C. L., Bjarnason, J., and Tu, A. T. (1978) Am. J. Pathol. 19. Huang, K.-F., Hung, C.-C., Pan, F.-M., Chow, L.-P., Tsugita, A., 93, 201 – 218. and Chiou, S.-H. (1995) Biochem. Biophys. Res. Commun. 216,
223 – 233. 5. Hokama, Y., Iwanaga, S., Tatsuki, T., and Suzuki, T. (1976) J.
Biochem. 79, 559 – 578. 20. Laemmli, U. K. (1970) Nature 227, 680 – 685.
6. Strydom, D. J. (1977) Biochim. Biophys. Acta 491, 361 – 369. 21. Chiou, S.-H., Hung, C.-C., and Huang, K.-F. (1992) Biochem.
Biophys. Res. Commun. 187, 389 – 396.
7. Ritonja, A., Meloun, B., and Gubensek, F. (1983) Biochim.
Bio-phys. Acta 746, 138 – 145. 22. Bradford, M. (1976) Anal. Biochem. 72, 248 – 254.
8. Bieber, A. L. (1979) in Handbook of Experimental Pharmacology 23. Twining, S. S. (1984) Anal. Biochem. 143, 30 – 34. (Lee, C. Y., Ed.), Vol. 52, pp. 295 – 306, Springer, Berlin.
24. Ouyang, C. (1957) J. Formosan Med. Assoc. 56, 435 – 448. 9. Pirkle, H., and Stocker, K. (1991) Thromb. Haemostas. 65, 444 –
25. Meaume, J. (1966) Toxicon 4, 25 – 58. 450.
26. Jimenez-Porras, J. M. (1970) Clin. Toxicol. 3, 389 – 431. 10. Markland, F. S. (1991) Thromb. Haemostas. 65, 438 – 443.
27. Esnouf, M. P., and Tunnah, G. W. (1967) Brit. J. Haematol. 13, 11. Kini, R. M., and Evans, H. J. (1992) Toxicon 30, 265 – 293.
581 – 590. 12. Kato, H., Iwanaga, S., and Suzuki, T. (1966) Experientia 22, 49 –
28. Bell, W. R., Pitney, W. R., and Goodwin, J. F. (1968) Lancet i, 50.
490 – 493. 13. Lo, T. B. (1972) J. Chin. Biochem. Soc. 1, 39 – 46.
29. Cercek, B., Lew, A. S., Hod, H., Jano, J., Lewis, B., Reddy, 14. Robeva, A., Politi, V., Shannon, J. D., Bjarnason, J. B., and Fox,
K. N. N., and Ganz, W. (1987) Thromb. Res. 47, 417 – 426. J. W. (1991) Biomed. Biochim. Acta 50, 769 – 773.
30. Pollak, V. E., Glas-Greenwalt, P., Olinger, C. P., Wadhwa, N. K., 15. Francis, B., and Kaiser, I. I. (1993) Toxicon 31, 889 – 899.
and Myre, S. A. (1990) Am. J. Med. Sci. 299, 319 – 325. 16. Freitas, M. A., Geno, P. W., Sumner, L. W., Cooke, M. E.,
Hudi-31. Bjarnason, J. B., and Fox, J. W. (1994) Pharmacol. Ther. 62, burg, S., Ownby, C. L., Kaiser, I. I., and Odell, G. V. (1992)
Tox-325 – 372.
icon 30, 461 – 464.
17. Francis, B., Seebart, C., and Kaiser, I. I. (1992) Toxicon 30, 32. Hung, C.-C., Huang, K.-F., and Chiou, S.-H. (1994) Biochem.
Biophys. Res. Commun. 205, 1707 – 1715.