T h e
n e w e n g l a n d j o u r n a l
o fm e d i c i n e
n engl j med 355;3 www.nejm.org july 20, 2006
322
percent of lung volume) among those with a higher amount of potentially recruitable lung, but these volumes included 183±482 and 135±434 ml of gas, respectively, and only 14±32 and 11±35 g of tissue, respectively, accounting for 1±3 per- cent and 1±2 percent of the total lung tissue.
Luciano Gattinoni, M.D.
Pietro Caironi, M.D.
Fondazione Istituto di Ricovero e Cura a Carattera Scientifico 20122 Milan, Italy
[email protected]
V. Marco Ranieri, M.D.
Azienda Ospedaliera San Giovanni Battista-Molinette 10126 Turin, Italy
Borges JB, Okamoto VN, Matos GF, et al. Reversibility of lung collapse and hypoxemia in early acute respiratory distress syndrome. Am J Respir Crit Care Med (in press).
Brower RG, Morris A, MacIntyre N, et al. Effects of recruit- ment maneuvers in patients with acute lung injury and acute respiratory distress syndrome ventilated with high positive end- expiratory pressure. Crit Care Med 2003;31:2592-7. [Erratum, Crit Care Med 2004;32:907.]
D’Angelo E, Calderini E, Robatto FM, Puccio P, Milic-Emili J.
Lung and chest wall mechanics in patients with acquired immu- nodeficiency syndrome and severe Pneumocystis carinii pneu- monia. Eur Respir J 1997;10:2343-50.
Quintel M, Pelosi P, Caironi P, et al. An increase of abdomi- nal pressure increases pulmonary edema in oleic acid-induced lung injury. Am J Respir Crit Care Med 2004;169:534-41.
The National Heart, Lung, and Blood Institute Acute Respi- ratory Distress Syndrome (ARDS) Clinical Trials Network. Com- parison of two fluid-management strategies in acute lung injury.
N Engl J Med 2006;354:2564-75
1.2.
3.
4.
5.
Hepatitis B Virus with Primary Resistance to Adefovir
To the Editor: Schildgen et al. (April 27 issue) 1 describe the results of tenofovir treatment in three patients infected with adefovir-resistant vari- ant hepatitis B virus (HBV) that had a valine at position 233 of the reverse-transcriptase domain instead of isoleucine (rtI233V). We recently pub- lished the results of two trials in which entecavir
demonstrated superiority over lamivudine in pa- tients with chronic hepatitis B that was positive for antibody against hepatitis B e antigen (HBeAg) 2 and in patients with HBeAg-negative chronic hep- atitis B.
3Four of the patients with HBeAg-positive chron- ic hepatitis B and four of those with HBeAg-
HBV DNA (copies/ml)
10
1110
1210
1010
910
710
610
410
310
210 10
810
50
0 24 36 48
Week
Patient 1, HBeAg-positive Patient 2, HBeAg-positive Patient 3, HBeAg-positive Patient 4, HBeAg-positive Patient 5, HBeAg-negative Patient 6, HBeAg-negative Patient 7, HBeAg-negative Patient 8, HBeAg-negative 10
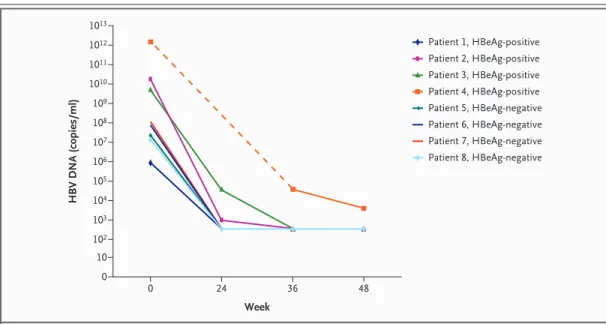
13Figure 1. Decrease in HBV DNA Levels during 48 Weeks of Entecavir Therapy in Eight Patients with an rtI233V Mutation in HBV Conferring Primary Resistance to Adefovir.
The limit of detection of the polymerase-chain-reaction assay was 300 copies per milliliter. Data on the HBV DNA burden at 24 weeks were not available for Patient 4.
Copyright © 2006 Massachusetts Medical Society. All rights reserved.
Downloaded from www.nejm.org at NATIONAL TAIWAN UNIV MEDICAL LIBRARY on May 9, 2007 .
correspondence
n engl j med 355;3 www.nejm.org july 20, 2006 323
negative chronic hepatitis B were infected with a strain that had the rtI233V mutation and received entecavir therapy. After 48 weeks, all six patients with paired liver-biopsy specimens that could be evaluated had histologic improvement, and two of the four HBeAg-positive patients had serocon- version.
Seven of the eight patients had undetectable viral loads (<300 copies of HBV DNA per milliliter) by week 36, and the mean decrease from base- line was 6.75 log (on a base-10 scale) copies per milliliter in the HBeAg-positive group and 5.11 log copies per milliliter in the HBeAg-negative group (Fig. 1). We tested the in vitro susceptibil- ity to entecavir of one HBV isolate with the rtI233V (and V191I) mutation from our viral archive and found a median effective concentration of 1.4 nM, as compared with 4 nM for wild-type virus.
Ting-Tsung Chang, M.D.
National Cheng Kung University Hospital Tainan 704, Taiwan
[email protected]
Ching-Lung Lai, M.D.
Queen Mary Hospital Hong Kong, China
Dr. Lai reports having received research fees, advisory fees, and lecture fees from Idenix Pharmaceuticals, and research and lecture fees from Lg Life Sciences.
Schildgen O, Sirma H, Funk A, et al. Variant of hepatitis B virus with primary resistance to adefovir. N Engl J Med 2006;
354:1807-12.
Chang TT, Gish RG, de Man R, et al. A comparison of enteca- vir and lamivudine for HBeAg-positive chronic hepatitis B. N Engl J Med 2006;354:1001-10.
Lai CL, Shouval D, Lok AS, et al. Entecavir versus lamivudine for patients with HBeAg-negative chronic hepatitis B. N Engl J Med 2006;354:1011-20. [Erratum, N Engl J Med 2006;354:1863.]
The authors reply: Chang and Lai describe eight patients infected with an HBV variant carry- ing the novel rtI233V mutation we recently iden- tified in three German patients. It causes primary resistance to adefovir, and it was also recently described in one Australian patient in whom adef- ovir therapy failed. 1
1.
2.
3.
Chang and Lai report that their patients were very efficiently treated with entecavir. We greatly appreciate their rapid comments for two reasons.
First, their data, in agreement with ours, indicate that the rtI233V mutation occurs in approximate- ly 2 percent of all patients with chronic hepatitis B (8 of 434 patients). Thus, our finding was not an isolated observation. It remains surprising that the mutation was not detected in earlier studies of adefovir. 2,3
Second, the observations by Chang and Lai show entecavir to be a highly efficient therapy op- tion for patients with the rtI233V mutation who have no response to adefovir, since tenofovir, which is what we used in our study, is still not licensed for HBV therapy. Since the receipt of lamivudine therapy for more than 12 weeks was an exclusion criterion in their studies, it is very likely that their patients were sensitive to lamiv- udine. Thus, it remains unknown how useful ente- cavir would be in patients with lamivudine-resis- tant rtI233V mutations. In our study, two patients with a response to tenofovir were resistant to lamivudine.
Oliver Schildgen, Ph.D.
Institute for Medical Microbiology, Immunology, and Parasitology
D-53105 Bonn, Germany
Hüseyin Sirma, M.D.
Heinrich-Pette-Institute D-20251 Hamburg, Germany
Wolfram Gerlich, Ph.D.
Institute for Medical Virology D-35392 Giessen, Germany
[email protected]
Bartholomeusz A, Kuiper M, Angus P, et al. Molecular analy- sis of HBV polymerase mutations associated with dual adefovir and lamivudine resistance. J Hepatol 2006;44:Suppl 2:S179. ab- stract.
Hadziyannis SJ, Tassopoulos NC, Heathcote EJ, et al. Long- term therapy with adefovir dipivoxil for HBeAg-negative chronic hepatitis B. N Engl J Med 2005;352:2673-81.
Marcellin P, Chang TT, Lim SG, et al. Adefovir dipivoxil for the treatment of hepatitis B e antigen–positive chronic hepatitis B.
N Engl J Med 2003;348:808-16.
1.
2.
3.