Vol. 27: 215-225, 1996 DISEASES OF AQUATIC ORGANISMS
Dis Aquat Org Published December 12
White spot syndrome baculovirus (WSBV)
detected in cultured and captured shrimp, crabs
and other arthropods
Chu-Fang Lol, Ching-Hui HO', Shao-En pengl, Chau-Huei Chenl, Hui-Chen ~ s u ' ,
Ya-Lin ~ h i u ' ,
Chen-Fang chang2, Kuan-Fu ~ i u ~ ,
Mao-Sen Su2, Chung-Hsiung wang3,
Guang-Hsiung
KOU'~''Department of Zoology, National Taiwan University, Taipei, Taiwan, ROC
'Tung Kang Marine Laboratory, Taiwan Fisheries Research Institute, Tung Kang, Ping Tung, Taiwan, ROC 'Department of Plant Pathology and Entomology, National Taiwan University, Taipei, Taiwan, ROC
ABSTRACT: Whlte spot syndrome baculovirus (WSBV) has been found across ddferent shrimp species and in different Asian countries. The detection of WSBV in shrimp with white spot syndrome has already been achieved by means of l-step polymerase chain reaction (PCR). In an attempt to establish a more sensitive assay, we evaluated the effect of 2-step amplification with nested primers on the sensitivity of WSBV diagnostic PCR. The sensitivity of the 2-step amplification was 10" to 10"imes higher than that of l-step amplification. Using both techniques, we successfully detected WSBV DNA in cultured and captured shrimp, crabs and other arthropods. Cultured Penaeus monodon (black tiger shrimp),
P.
japonicus (kuruma shrimp), P penicjllatus (red tail shrimp), and Metapenaeus ensis (sand shrimp) displaying white spot syn- drome were collected from farms at different localities. One-step amplification of the DNA extracted from these shrimps consistently yielded an expected 1447 bp PCR product. Some of the tested specimens of cul- tured Scylla serrata (mud crab) that exhibited white spot syndrome were positive in l-step WSBV diag- nostic PCR, while others were positive only in 2-step WSBV diagnostic PCR. Use of the 2-step amplifica- tion protocol also detected a WSBV-specific DNA fragment in Macrobrachium rosenbergii (the giant freshwater prawn) exhibiting white spot syndrome. We also confirmed that WSBV exists in wild-caught shrimp ( P monodon, f! japonicus,P
semisulcatus and P penicillatus) and crabs (Charybdisferiatus. Por- tunuspelagicus and P. sanguinolentus) collected from the natural environment in coastal waters around southern Taiwan. Detection of WSBV in non-cultured arthropods collected from WSBV-affected shrimp farms revealed that copepods, the pest crab Hehce tndens, small pest Palaemonidae prawn and the larvae of an Ephydridae insect were reservoir hosts of WSBV. The relatedness between WSBV and Thailand's systemc ectodermal and mesodermal baculovirus (SEMBV) is d~scussed in this paper.KEY WORDS: White spot syndrome
. Shrimp viral disease . PCR
. Cultured and non-cultured arthropodsINTRODUCTION
White spot syndrome baculovirus (WSBV) (Lightner
1996) has been found across different shrimp species and
in different Asian countries. The principal clinical sign of
the disease associated with this virus is the presence of
white spots in the exoskeleton and epidermis of the dis-
eased shrimp. The causative virus itself consists of an
enveloped, rod-shaped nucleocapsid. It is extremely vir-
ulent, has a wide host range and targets various tissues
'Addressee for correspondence. E-mail: ghkouOccms.ntu.edu.tw
(Chou et al. 1995, Wang et al. 1995, Chang et al. 1996,
Lo
et al. 1996). The rapid onset and lethality of this disease
are remarkable. Based on the histopathology of the
affected animals and the characteristics of the virus,
WSBV is either identical or closely related to Thailand's
systemic ectodermal and mesodermal baculovirus
(SEMBV) (Wang et al. 1995, Wongteerasupaya et al.
1995,
C.Wongteerasupaya pers. comm.). WSBV infec-
tion and its associated mortality is emerging as one of the
most significant problems for the global shrimp industry.
The amplification of selected
DNA sequences
bypolymerase chain reaction
(PCR) is fulfilling its promise
as a powerful diagnostic tool for the identification of
0 Inter-Research 1996
Dis Aquat Org 27: 215-225, 1996
pathogens (Erlich et al. 1988, Oste 1988). In recent
years, a number of researchers have drawn attention to
the need for more sensitive diagnostic techniques using
'high-tech' approaches for crustacean viruses (Brock
1992, Lightner et al. 1992, Bruce et al. 1993, 1994,
Chang et al. 1993, Man et al. 1993, 1995, Poulos et al.
1994. Wang e t al. 1995). In the case of WSBV, we have
already developed PCR primers a n d nucleic acid
probes for diagnosis (Lo et al. 1996). The specificity of
these WSBV PCR primers has already been established
(Lo e t al. 1996), and in the present paper w e use the
more sensitive nested PCR to detect the latent or pre-
patent carrier state of WSBV infection in shrimp and
other arthropod populations collected from shrimp
farms or from the natural environment.
MATERIALS AND METHODS
Oligonucleotide primers for WSBV diagnostic PCR.
A nested primer set derived from the sequence of a
cloned WSBV
Sal11461 bp DNA fragment (Lo et al.
1996) including 146F1, 5'-ACT ACT AAC TTC AGC
CTA TCT AG-3'; 146R1,5'-TAA TGC GGG TGT AAT
GTT CTT ACG A-3'; 146F2, 5'-GTA ACT GCC CCT
TCC ATC TCC A-3'; 146R2, 5'-TAC GGC AGC TGC
TGC ACC TTG T-3'; 146F4, 5'-AGA AGG TTA GCG
AAT GGA CTG-3'; and 146R3, 5'-CGA AGA GTA
GTG TTA GAA GAG GA-3' was utilized for l-step or
2-step WSBV diagnostic PCR. Four of these were used
in our previous study (Lo e t al. 1996).
As described previously (Lo et al. 1996), the quality
of the DNA extracted from tested organisms was
checked with a primer set amplifying a decapod gene
before the application of WSBV diagnostic PCR. For
this purpose, 2 primers, 143F (5'-TGC CTT ATC AGC
TNT CGA TTG TAG-3', where N represents G, A, T o r
C) a n d 145R (5'-TTC AGN TTT GCA ACC ATA CTT
CCC-3'), derived from a highly conserved region of the
1 8 s rRNA sequence of decapods (Kim
&Abele 1990,
Lo et al. 1996) were used.
One-step WSBV diagnostic PCR. Three primer pairs,
146F1/146R1, 146F2/146R2 and 146F4/146R3, were
used for l-step WSBV diagnostic PCR. The DNA
sample extracted from the muscle of 1 diseased black
tiger shrimp naturally infected with WSBV was used as
template DNA for demonstrating the specificity a n d
sensitivity of the 3 primer pairs in l-step WSBV diag-
nostic PCR. The DNA samples used for amplification
totaled 0.1 pg in a 100 p1 reaction mixture containing
10 m M Tris-HC1, pH 8.8 a t 25"C, 50 mM KC1, 1.5 mM
MgCl,, 0.1
%Triton X-100, 200 pM of each dNTP,
100 pm01 of each primer,
2units of DynaZymeTMII
DNA Polymerase (Finnzymes Oy, Riihitontuntie 14 B,
FIN-02200 Espoo, Finland). The amplification was per-
formed in a AG-9600 Thermal Station (Biotronics Corp.
Lowell, MA, USA) for 1 cycle of 94°C for
4min, 55°C
for 1 min. 72°C for 2 min; 39 cycles of 94°C for
1 min,55°C for
1 min, 72°C for 2 min, plus a final 5 min ex-tension at 72OC after 40 cycles. Control reactions con-
taining no template DNA were run for all PCR reac-
tions. A portion (10 pl) from each of the completed PCR
reactions was mixed with 1 p1 loading buffer and sub-
jected to electrophoresis on
1 %agarose gels contain-
ing ethidium bromide at a concentration of 0.5 pg ml-',
and visualized by ultraviolet transillurnination.
Two-step WSBV diagnostic PCR. The first PCR step
was performed as described above with the outer
primer pair 146F1 a n d 146R1. After completion of the
first step, 10 p1 of the reaction mixture was added to
90 p1 of PCR cocktail containing the inner primer pair,
146F2 and 146R2, and this was then subjected to a
second step of amplification over the same 40 cycles.
For the ana!ysis of PCR products, 10 p1 of the final reac-
tion mixture was used.
Evaluation of the effect of 2-step
PCR with nested
primers o n the sensitivity of WSBV diagnostic PCR.
The relative sensitivity of l-step PCR, 2-step PCR, 1-
step PCR with Southern hybridization and l-step PCR
with dot hybridization was investigated as follows:
First step o f amplification:
Serially 10-fold diluted
(10-' to IO-') DNA samples of deproteinized DNA ex-
tracted from the muscle of diseased black tiger shrimp
naturally infected with WSBV served as template DNA
samples in the first step of amplification with the
146F1/146R1 primer set.
Second step o f amplification:
An aliquot (10 p1) of
each PCR product yielded in the first step of amplifi-
cation served a s template DNA in the second step of
amplification.
One-step PCR with Southern hybridization:
Southern
hybridization was performed with the gel used to ana-
lyze the products yielded in the first step of amplification.
After application of alkali (1.5 M NaCl and 0.5 N NaOH)
to denature the DNA, the gel was neutralized with 1 M
Tris (pH 7.4) and 1.5 M NaCl and subsequently trans-
ferred to a Hybond-N nylon membrane (Amersham,
Little Chalfont, Buckinghamshire, UK) using a Hoefer
TE 80 vacuum transfer unit (Hoefer Scientific Instru-
ments, San Francisco, CA, USA) for 60 min. A solution
of 20x SSC ( l x SSC = 150 mM NaC1, 15 mM sodium
citrate, pH 7.0) was used as transfer buffer. After cross-
linking the DNA with the membrane by UV light, the
blot was used for hybridnation with a probe derived
from the 941 bp PCR product of a reaction mixture with
the 146F2 and 146R2 primers. This probe was non-
radioactively 1abel.ed with digoxigenin (DIG)-dUTP
(Boehringer Mannheirn, Germany) by a random priming
method. The blot was hybridized at 37°C for 16 h with
the DIG-labeled probe, after prehybridization at 37°C for
Lo et a1 : WSBV d e t e c t ~ o n In arthropods
12 h in 50 % formaniide, 5 x SSC, 1 mM EDTA, 50 mM Tris (pH 8) a n d 5 x Denhardt's reagent [0.1% Ficoll-400, 0.1 % polyvinyl pyrrolidone, 0.1 % bovine serum alburmn (BSA)]. After hybridization, detection of the DIG-labeled nucleotides in blots was accomplished using a DIG Luminescent Detection f i t (Boehnnger Mannheim). The blot was exposed to Kodak XAR-5 film at 37OC for 15 to 30 min to record the chemiluminescent signal.
One-step PCR
with dot hybridization:
An aliquot (10 p1) of each of the completed PCR reactions in the first step amplification was incubated with 16 p1 of a n alkali solution (1.5 M NaCl, 0.5 N NaOH) and 24 p1 of H 2 0 a t room temperature for 5 to 7 min. After being neutralized with 4 p1 of neutralization solution (1.5 M NaCl, 1 M Tris, pH 7.4) for 10 min, the reaction niix- tures were spotted onto Hybond-N nylon paper using a 96-well dot-blot vacuum filtration manifold appara- tus (Schleicher a n d Schuell, Inc. Keene, NH, USA). The blot was rinsed with 2 x SSC. After cross-linking the DNA with the membrane by UV light, the blot was used for hybridization with a DIG-labeled probe derived from the 941 b p PCR product. The procedure for the hybridization a n d signal detection was the same a s that for Southern hybridization.Detection of WSBV in cultured decapods. Cultured Penaeus monodon (black tiger shrimp), l? japonicus (kuruma shrimp), l? penjcillatus (red tail shrimp), Meta- p e n a e u s ensis (sand shrimp), Macrobrachlum rosen- bergii (the giant freshwater prawn) and Scylla sen-ata (mud crab) that displayed white spot syndrome (WSS) were collected at various growth stages, from various geographic regions of Taiwan, and in different seasons. These shrimp specimens were subjected to WSBV diagnostic PCR. From each specimen, 1 pereio- pod was excised a n d placed in a small vial, rapidly frozen in liquid nitrogen a n d then stored a t -70°C until use for PCR template DNA preparation. DNA was iso- lated following the method described by Lo et al. (1996) with some modification. Briefly, a piece of pereiopod (approx. 100 mg) was placed directly in a microfuge tube containing 1.2 m1 digestion buffer (100 mM NaC1, 10 mM Tris-HC1, pH 8, 25 mM EDTA, pH 8 , 0 . 5 % N- lauryl sarcosine, 0.5 mg ml-' proteinase K) a n d crushed with a disposable stick. After incubation at 65'C for 1 h , 5 M NaCl was added to adjust the NaCl concen- tration of the DNA solution to 0.7 M. The solution was then treated with 1 % N-cetyl N,N,N-trimethylammo- nium bromide (CTAB) for 10 nlin at 65°C followed by successive extractions with 1 X volume of chloroform/ isoamyl alcohol once, 1 X volume of phenol 2 to 3 times, a n d 2 X volume of chloroform/isoamyl alcohol once.
The DNA was recovered by ethanol precipitation, dried a n d resuspended in 0.1 X Tris-ethylenediamine- tetraacetic acid (1 X TE = 10 mM Tris-base, 1 mM EDTA.2Na, pH 7.6) buffer at 65OC for 30 min, before
being stored at 4°C until use for WSBV diagnostic PCR. The PCR products recovered by ethanol precipitation were further analyzed with HaeIII, HpaII, RsaI a n d Sa3A I restriction endonucleases.
Detection of WSBV in wild-captured decapods. From July through December 1995, 66 Penaeus monodon (body weight. 59 to 128 g ) , 23 l? japonicus (35 to 100 g ) , 32 l? semisulcatus (80 to 127 g ) , a n d 27 l? penicillatus (18 to 39 g ) in the adult stages and of wild origin were captured from their natural environment in the coastal waters around southern Taiwan. On their arrival at Tung Kang Marine Laboratory, specimens were ex- amined visually and found to b e in good health with no evidence of WSS. In order to minimize the effect of stressful conditions which can trigger increased viral replication in the shrimp, excision of 1 pereiopod from each of the specimens was performed immediately, a n d the excised pereipod was then subjected to 2-step WSBV diagnostic PCR. When the results of the first step of amplification became available 4 h later, one of the specimens whose pereiopod was positive for WSBV by l - s t e p PCR was killed a n d its tissues were prepared for histological observation using light iiiicroscopy ds described previously (Chou et al. 1995). The remaining shrimp specimens w e r e maintained in 500 1 tanks for a period of further observation.
Detection of WSBV in non-cultured arthropods from shrimp farms. Specimens of copepods, pest crab Hehce tridens, small pest Palaemonidae prawn a n d pupae of a n Ephydridae insect were collected from shrimp farms in epizootic areas a n d subjected to 2-step WSBV diagnostic PCR in order to ascertain whether WSBV was present in these non-cultured arthropods.
RESULTS
Oligonucleotide primers and amplicons of WSBV diagnostic PCR
The relative positions of oligonucleotide primers cor- responding to the sequences of the WSBV Sal1 1461 b p DNA fragment for WSBV diagnostic PCR a n d their aniplicons a r e indicated in Fig. 1A. Fig. 1B shows the 1- step PCR product primed by 146F1/146R1 (lane l ) ,
146F2/146R2 (lane 2) a n d 146F4/146R3 (lane 3) with each lane using the same amount of template DNA iso- lated from 1 WSBV-infected specimen of black tiger shrimp. The primer pairs achieved higher sensitivity when they yielded shorter amplicons. In the nested protocol for 2-step PCR in this study, 146F1/146R1 w a s used for outer PCR while 146F2/146R2 was used for inner PCR. However, several other combinations of primer pairs for the nested protocol were tested a n d also yielded good results (data not shown).
Dis
Aquat Org 27: 215-225, 1996WSBV Sail l461 bp DNA fragment
Fig. 1. Pnmers and amplicons of WSBV diagnostic PCR. (A) Location and sequence of primers for WSBV diagnostic PCR; (B) amplicons primed by 146F1/146Rl (lane l ) , 146F2/146R2 (lane 2) and 146F4/146R3 (lane 3). M: pGEM DNA size marker
Evaluation of the effect of 2-step PCR with nested
primers on the sensitivity of WSBV diagnostic PCR
The sensitivity of the 2-step PCR was 103 to 104 times
higher than that of the l-step PCR (Fig.
2). The 10 1.11of completed PCR reaction mixture of the first step of
amplification, which served as the DNA source for the
second step of amplification, contained some 1447 bp
PCR products as well as some excess from the
146F1/146R1 primer set. Because of this, the DNA samples
that yielded a visible band of 1447 bp fragment in the
first step of amplification usually yielded 2 visible
bands (1447 and 941 bp) in the second step of amplifi-
cation (Fig. 2B). The sensitivity of 2-step PCR was also
higher than that of l-step PCR with Southern or dot
hybridization, by a factor of about 102
to 103
(Fig. 2B, C,
D).
One-step PCR
A b p l 2 3 4 5 6 7 8 9
Two-step PCR
B b p 1 2 3 4 5 6 7 8 9
One-step PCR+Southern hybridization
C b p 1 2 3 4 5 6 7 8 9
Detection of
WSBV incultured decapods
WSS was found in 4 major cultured species of marine
shrimp, i.e. Penaeus monodon, P japonicus,
l? penicil-latus and Metapenaeus ensis (Fig. 3 ) . Except for
I!
penicillatus, WSS was readily observed in diseased
cultured shrimp, with the white spots being quite
marked (Fig. 3A,
B, D ) . In diseased P penicillatus,
however, the white spots were not easily seen (Fig.
3 C )until after the carapace had been removed. One-step
amplification of WSBV DNA from the DNA samples of
shrimp that displayed WSS consistently yielded a
1447 bp PCR product as shown in the left panel of
Fig. 4. The results confirmed the presence of a rela-
tively large amount of
WSBV in the tested specimens.
Interestingly, we found that the amount of the WSBV
in shrimp from the same pond collected at the same
One-step PCR+dot hybridization 1 2 3 4 5 6 7 8 9
E::
Fig. 2. Detection of WSBV DNA in serially 10-fold diluted DNA extracted from naturally infected black tiger shrimp by (A) l-step PCR; (B) 2-step PCR; (C) 1-step PCR with Southern hybridization; and (D) l-step PCR with dot hybridization. (A) l-step PCR using 146F1 and 146R1 primer pair, (B) 2-step (nested) PCR using 146F1 and 146R1. as a n outer primer pair, and 146F2 and 146R2 a s a n inner primer pair; (C) Southern hybridization after l-step PCR using a DIG-labeled 146F2- 146R2 probe; ( D ) dot hybridization after l-step PCR using a DIG-labeled 146F2-146R2 probe. Lanes 1 to 9 correspond to 10-' to 10-Q dilution of infected shnmp DNA. PCR products
Lo et al.. WSBV detection in arthropods
Fig. 3. Cultured marine shrimp with white spot syndrome. ( A ) Penaeus monodon, ( B ) P japonicus, (C) P penicillatus, ( D ) Meta-
penaeus ensis. Scale bars = 1 cm
time was similar; this is evidenced by
the similar intensity of their PCR prod-
uct bands as shown in the left panel of
Fig.
4,in which 2 specimens of each
shrimp species collected from the same
pond at the same time are shown. No
1
differences were observed in the rest-
riction profiles of PCR products yielded
by l? monodon, l? japonicus,
l?penicil-
latus,
and Metapenaeus ensis,
analyzed
with
HaeIII, HpaII, RsaI
and
Sau3AI
restriction endonucleases (Fig.
4,right
panel), which indicates the close relat-
edness of WSBV found in the different
species of shrimp collected from various
geographic regions of Taiwan and in
different seasons.
The WSS displayed by the cultured
marine crab
Scylla serrata
was readily
seen in pereiopods rather than in the
carapace. When these crabs were sub-
jected to WSBV diagnostic PCR, some
tested specimens gave a positive result
in the first step of amplification, while
others gave a positive result only after
reamplification. Unexpectedly, all tested
specimens of the cultured freshwater
prawn Macrobrachium rosenbergii with
WSS gave WSBV-positive results only
One-step WSBV DNA PCR M Pm Pi Po Me N
Haelll Hpall Rsa l Sau3Al
M Pm
pr
Pp Me Pm Pj Pp - Me E m3
Pp
- - - - Me Pm P) Pp _M+Fig. 4. WSBV-specific DNA fragments amplified frolll L c l l l p l O L c ylYm 3 C L I I I p I c J p l c p a l c U I I V I ~ cultured marine shrimp Penaeus monodon (Pm), P: japonicus (Pj),
P
pen~cillatus (Pp), and Metapenaeus ensis (Me) that displayed marked white spot syndrome. Two specimens for each species ofP
monodon, P: japonicus, and l? penicillatus were collected from the same pond at the same time. Left panel: l-step WSBV PCR products. Right panel: result of the PCR products analyzed with HaeIII, HpaII, RsaI and Sau3AI220 Dis Aquat Org 27: 215-225. 1996
A
1st Step PCRB
C
2nd Steo PCR Hoa II Rsa l Fig. 5. WSBV-spec~f~c DNA fragments amplified from template DNA samples prepared from cultured mud crab Scylla serrata and cultured freshwater shrimp Macrobrachium rosenbergii
that displayed white spot syndrome. (A) l-step WSBV PCR
products. (B) 2-step WSBV PCR products. (C) 2-step PCR prod-
ucts amplified from Penaeus monodon (Pm), S. serrata (Ss) and
M. rosenbergii (Mr) cleaved with HpalI and RsaI restriction
endonucleases. M: pGEM DNA size marker
after the second step of amplification, in
which they yielded a
94 1bp PCR fragment.
However, analysis of this PCR product with
Hpa I1 and Rsa
I restriction endonucleases
showed similar restriction profiles to those
of the 941 bp fragments yielded by
P mon-
odon and the mud crab S. serra1.a (Fig.
5 ) .Detection of WSBV in wild-captured decapods
Captured
specimens
of
apparently
healthy, wdd Penaeus monodon,
P japo-
nicus,
P semisulcatus and P penicillatus
were immediately tested by PCR analysls
upon their arnval at Tung Kang Manne
Laboratory. The signs of WSS, even when
present, were not readily seen in these
examined specimens (Fig 6).
The results of PCR analysis (Table
1)indicated the presence of WSBV in all
4of
the species investigated. Of the 66 Penaeus
4
monodon collected
50were positive by
'2.- 4step WSBV diagnostic PCR and 22 of these
were positive in the first step of amplifica-
tion. The l-step WSBV PCR-positive indi-
viduals usually died within 1 to
3 d after
arrival at Tung Kang Manne Laboratory.
Small white spots were observed on cara-
I
ited the typical histopathological changes
caused by WSBV infection in tissues of the
gill, the stomach, the pleopods and the
lyrnphoid organ. Cells with characteristic
hypertrophied nuclei were readily seen in
these tissues (Fig.
7 ) .Occasionally, crabs were found amongst
Fig. 6. (A) Penaeus monodon,
(B)
P japonicus, (C) P semlsulcatus, and (D)the captured shrimp, specifically Charyb-
P penicillatus captured from coastal waters near southern Taiwan. Scale
dis feriatus
( lPo*unus
pelagi-
Lo et al.: WSBV detection in arthropods 221
Table 1. Results of 2-step WSBV diagnostic PCR with adult, captured Penaeus monodon, P japonicus, P semisulcatus and
P
penicillatus. Values represent no. of shrimp positive in the first and second step PCR per no. of shrimp examined WSBV diagnostic PCR F! monodon P japonicus F! seniisulcatus P penicillatusl -step PCR 22/66 5/23 0/32 0/27
2-step PCR 50/66 14/23 2/32 3/27
(1 specimen). All of these crabs were positive In the
second step of 2-step WSBV diagnostic PCR.
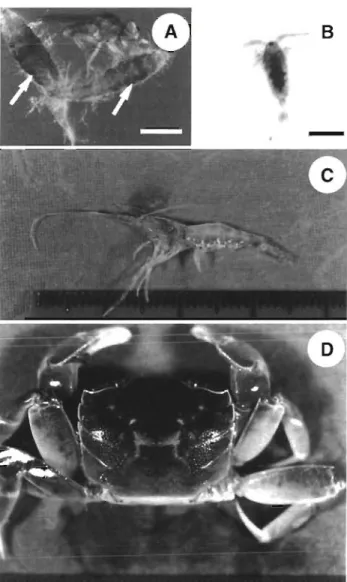
Detection of WSBV in non-cultured arthropods from
shrimp farms
Copepods, pest crab Helice tndens, small pest Palae-
monidae prawn, and pupae of an Ephydridae insect
were collected from shrimp farms in epizootic areas of
WSBV infection (Fig. 8).
Template DNA samples were
prepared from the pereiopods of
15specimens of small
Palaemonidae pest prawns and 9 specimens of H.
tri-
d e n s pest crab. They were also prepared from 10
coarctate larvae removed from the protective case of
Ephydridae pupae and 6 pooled, pelleted samples of
copepods. These samples were subjected to 2-step
WSBV diagnostic PCR. As shown in Table 2, WSBV-
positive cases were found in copepods, pest crab, pest
prawn and insects which had been recently (within
half a day) collected from shrimp farms. Four out of
10Ephydridae pupae, 2 out of the 6 pooled samples of
copepods,
10out of 15 pest prawn specimens and
4out
of11
pest crab specimens were positive by l-step
WSBV diagnostic PCR. Thus copepods, pest crab H.
tridens
and small pest Palaemonidae prawn a s well
as an Ephydridae insect are evidently reservoir hosts
for WSBV in shrimp culture facilities.
Fiq. 7. Penaeus monodon. (A]
selected l-siep
WSBV
PCR-I
positive
P:
rno-nodon exhibit- ing the typical histopatho- logical changes caused byWSBV
attacks. Degenerated cells characterized by hyper- trophied nuclei (arrows) are readily seen in these tissues.222 DIS Aquat Org 27. 215-225, 1996
DISCUSSION
The specificity of the primers used here as well as
the standardization of preparation procedures for
template
DNAa n d its quality control have been d e -
scribed previously (Lo et al. 1996). Furthermore, in
pilot studies of WSBV tissue tropism in various deca-
pods using histopathological examination,
in situ
hybridization assay a n d transmission electron micro-
scopic observation, the preliminary data show that in
most cases, a positive PCR result coincides with WSBV
histopathology a n d the presence of WSBV positive
cells as revealed by
i n situ hybridization (data not
included in t h s manuscript). Nonetheless, although
2-step PCR is very sensitive a n d reliable, there is a
need for stringent controls to eliminate the increased
chance of false positives. Therefore, in addition to a
template-free reagent control reaction, a PCR reaction
with a template is run as a supplemeniary negative
control for all PCR reactions (Lo et al. 1996). In the pre-
sent study, a sample from a previously prepared batch
of WSBV-free shrimp abdominal muscles (100 mg in
each vial; stored at -70°C) was always included in our
DNA
extraction samples (data not shown). This pro-
vided some reassurance that positive results did not
arise from contamination during processing, but false
positives a n d physical environmental contamination
a r e complex issues that will b e d s c u s s e d more fully in
papers currently in preparation.
WSBV was previously classified by Wang et al.
(1995) as the genus
Non-Occluded [sic] Baculovirus
( N O B ) of the subfamily Nudibaculovirinae of the
family
Baculoviridae in accordance with the fifth
report of the International Committee on the Taxon-
Fig. 8. (A) Insect (family: Ephydridae), (B) copepod (subclass: Copepoda, Schmackeria dubia), (C) pest prawn (farmly: Palaemonidae, Exopalaemon orientis.), and ( D ) pest crab
(Hehce tridens) collected from shrimp farms in epizoot~c ar- eas of WSBV lnfectlon. White scale bar In (A) = 3 mm; black scale bar in (B) = 0 5 mm. Arrows indicate the pupae of an
Ephydridae insect
omy of Viruses (ICTV) (Francki et al. 1991). In the sixth
report of the ICTV, however, the subfamily
Nudi-
baculovirinae a n d the genus NOB a r e not listed (Mur-
phy et al. 1995). The 2 included genera of the family
Baculoviridae a r e Nucleopolyhedro~irus
a n d
Granulo-
virus, a n d WSBV is unlikely to belong to either. The
insect viruses Oryctes rkinoceros virus (OrV) and
Heliothis zea virus 1 (HzV-l), which were previously
classified a s nonoccluded members of the family
Baculoviridae, have now been placed
in the list of unassigned invertebrate
Table 2. Results of 2-step WSBV d~agnostlc PCR in copepods (subclass: Cope- viruses,
than
a
poda), pest crab Hellce tndens, small pest prawn (family: Palaemonidae) a n d a nnonoccluded baculoviruses have been
insect (famlly: Ephydridae) collected from s h n m p farms In epizootic areas of
found associated with a number of
WSBV infect~on. Values represent no. of specimens positwe ln the f ~ r s t and
arthropod hosts, including lnsecta,
second step PCR per no. of specimens examined
Arachnida a n d Crustacea, very Little is
WSBV diagnostic PCR Insect Copepod Pest prawn Pest crab
-
known about their molecular biology
(Sano et al. 1981, Lester e t al. 1987,
l -step PCR 4/10 2/6 10/15 4/11 2-step PCR 8/10 4/6 12/15 5/11
Johnson 1988, Johnson
&Lightner
1988, Burand 1991, Crawford 1994,
Wang e t al. 1995, Wongteerasupaya
Lo et al.. WSBV detection ~n arthropods 223
et al. 1995). To date, it is not clear whether WSBV and
other nonoccluded baculoviruses should in fact be
included in the Baculoviridae or not. Genome sequenc-
ing, genome organization, gene order, strategy of
replication and other genetic considerations of insect
and crustacean nonoccluded baculoviruses, which are
becoming increasingly important as viral taxonomic
criteria (Murphy et al. 1995), are in urgent need of
further study before the taxonomic status of these
viruses can be decided. Notwithstanding the uncer-
tainty in the taxonomic position of nonoccluded bac-
uloviruses, in a recently published handbook of patho-
logy and diagnostic procedures for diseases of penaeid
shrimp (Lightner 1996), the present virus is called
white spot syndrome baculovirus (WSBV). Conse-
quently, in the interests of nomenclaturial consistency,
this is the name that we have used here.
Wang e t al. (1995) demonstrated that negatively
stained preparations of WSBV virions frequently had
tail-like projections extending from one end, that the
nucleocapsid components form parallel cross-striations
which are perpendicular to the longitudinal axis of the
nucleocapsid, and that the full length of WSBV
genonlic
DNAwas longer than 150 kbp. WSBV is very
close to SEMBV from Thailand
in
terms of viral mor-
phology (size, bacilliform with a tail, banding of nuc-
leocapsid) and type and size of genome. WSBV EcoR I
and BamH I restriction profiles (authors' unpubl. data)
were also found to be similar to those published for
Thailand's SEMBV (Wongteerasupaya et al. 1995). [In
this regard, please note that Thailand's SEMBV group
has informed us that the names of the restriction
enzymes were inadvertently transposed in the caption
to Fig.
4of their article in Diseases of Aquatic Organ-
isms (Vol. 21, p. 75)
(C.Wongteerasupaya pers.
cornm.).] Thus, SEMBV and WSBV are at least very
similar if not identical.
In several respects including viral morphology and
genome size, WSBV is distinct from the Hz-l virus but
shares some characteristics with the Oryctes rhinoc-
eros virus, a pathogenic virus of invertebrates, which
infects coleopteran insects in the family Scarabaeidae
(Burand 1991, Crawford 1994, Murphy et al. 1995,
Wang et al. 1995, Wongteerasupaya et al. 1995). Never-
theless, WSBV a n d OrV are alike only to a certain
extent, and their very similarity raises questions. What,
if any, is the relationship between insect viruses and
crustacean viruses? Are these viral agents present in
insects and crustaceans in the natural environment?
Could interactions between insects and cultured crus-
taceans contribute to the increased number of out-
breaks of shrimp viral disease? Most immediately, is it
possible that insects or other non-cultured animals act
a s reservoir hosts of crustacean viruses or vice versa?
In the present study, we have shown PCR evidence of
WSBV in the pupae of a n Ephydridae insect. It was
also detected in copepods, another non-decapod. So
far, it is not clear whether WSBV replicates in either
insects or copepods, or whether it can cause disease in
them. The interaction between cultured shrimp and
insects or other reservoir hosts that inhabit farms, estu-
aries and coastal waters are of great interest. The
dynamics involved may be very complex but under-
standing them should help prevent viral diseases.
We found that about 40% of the pest crab Helice
tri-
dens and Palaemonidae prawn collected from epi-
zootic areas of WSBV infection were positive for WSBV
by l-step diagnostic PCR. These animals were ob-
tained from farms where mass mortality of cultured
shrimp had occurred and only very few cultured
shrimp still survived. We carefully examined a large
number of carapaces removed from Palaemonidae
specimens immediately after being collected from
these farms and all of them were exceptionally clear.
Thus, these pest species are very likely asymptomatic
carriers of WSBV in farms. However, when transferred
to the laboratory, the number of the specimens positive
by l-step diagnostic
PCR increased over time (data
not shown) and all died within a few days. White spot
syndrome was readily observed in these moribund
specimens. Thus, under stressful conditions, WSBV
was able to cause disease in these pest prawns.
In Taiwan, there are
3different sources of salt water
for shrimp farms: oceanic surface water, beach sand-
filtered sea water and water from saline wells (Chen
1990). Underground sea water (i.e. that from saline
wells) is the most popular water source for small
shrimp culture ponds in Taiwan while large earthen
ponds tend to use oceanic surface water which is
exchanged through canals by gravity flow during high
and low tides. Beach sand-filtered sea water and
underground water systems are normally pest-free,
and the threat of pathogens is much reduced. By con-
trast, there is a high risk that tidal input can introduce
pathogens, microscopic eggs and larvae of such pests
a s Palaemonidae prawn and pest crabs. We have
shown here that these pests may be major reservoir
hosts of WSBV and it is now important to continue to
examine these species in greater detail (histology,
in situ hybridization, bioassays, etc.) to determine to
what extent they might represent a real threat to the
shrimp aquaculture industry. Furthermore, the direct
discharge of water from shrimp culture systems into
drainage canals can release WSBV carriers and WSBV
itself into coastal waters. This could have a severe
negative impact on natural decapod populations and
other susceptible animals. A number of wild shrimp
populations now also carry infectious hypodermal a n d
hematopoietic necrosis virus (IHHNV) (Lotz 1992),
possibly as
a
consequence of nearby shrimp culture
224 Dis Aquat Org 27: 215-225, 1996
activities (Fulks & M a i n 1992). It is clear t h a t w a t e r s u p p l y s y s t e m s a n d poor m a n a g e m e n t of t h e culture a n d w a s t e w a t e r c a n s p e e d t h e s p r e a d of t h e virus a n d h a v e a s e v e r e n e g a t i v e i m p a c t o n t h e n a t u r a l envi- r o n m e n t .
It w a s disappointing to l e a r n t h a t WSBV w a s p r e s e n t
i n wild populations of
Penaeus monodon,
F!japonicus,
F!
semisulcatus,
and
F!penicillatus.
S e v e r a l possible factors m i g h t e x p l a i n t h e h i g h p r e v a l e n c e ofWSBV
infection i n F!monodon
w h e n c o m p a r e d to t h e o t h e r s p e c i e s s t u d i e d . First, t h e black tiger s h r i m p f r e q u e n t s w a t e r s of less t h a n 30 m i n d e p t h . Unlike m a n y of t h e o t h e r p e n a e i d s h r i m p , it d o e s n o t b u r y itself i n sand b u t s t a y s o n t h e s u r f a c e of t h e s u b s t r a t e ( C h e n 1990). C o n - s e q u e n t l y , it h a s m o r e c h a n c e of b e i n g e x p o s e d t o WSBV i n coastal w a t e r s polluted b y t h e d i s c h a r g e from affected p o n d s . Secondly, m a n y p a r t s of Asia, includ-ing T a i w a n , h a v e b e e n conducting p r o g r a m s of releas-
ing h a t c h e r y - r e a r e d postlarvae (Fulks
& M a i n 1992) o r30 g f e m a l e s h r i m p ( C h e n 1990) into coastal w a t e r s i n
o r d e r t o r e p l e n i s h n a t u r a l s h r i m p stocks. Before w e h a v e effective diagnostic tools for all t h e k n o w n s h r i m p viruses a n d c a n certify s h r i m p stocks, t h e practice of r e l e a s i n g f a r m e d s h r i m p into t h e n a t u r a l e n v i r o n m e n t s h o u l d b e a v o i d e d b e c a u s e it m a y h a v e a n e g a t i v e i m p a c t o n n a t u r a l stocks. Conversely, t h e w i d e s p r e a d u s e of c a p t u r e d broodstock of t h e black t i g e r s h r i m p o r k u r u m a s h r i m p to p r o d u c e fry for stocking farms m a y c o n t r i b u t e to t h e viral d i s e a s e p r o b l e m s f a c e d by t h e s h r i m p c u l t u r e industry i n T a i w a n a n d m a n y o t h e r a r e a s .Acknowledgements. This work was supported by the Council of Agriculture under grant no. 85-AST-1.1-FAD-49(27)A, 85-AST-1.1-FAD-49(27)B and the National Science Council under grant no. NSC85-2311-B002-012-B04. We are indebted to Dr Jung-Yaw Lin. Institute of Biochemistry. College of Medicine, National Taiwan University for his constructive suggestions, and for access to data bases. We thank Dr I-Chiu Liao, Director General, Taiwan Fisheries Research Institute
(TFRI), for his constructive suggestions. We thank Dr Timothy
William Flegel of Mahidol University for numerous dscus- sions on Thailand's SEMBV. We are indebted to Mr Paul
Barlow for his helpful criticism of the manuscript.
LITERATURE CITED
Brock JA (1992) Current diagnostic methods for agents and diseases of farmed marine shrimp. In: Fulks W, Main KL (eds) Diseases of cultured penaeid shrimp in Asia and the United States. The Oceanic Institute, Honolulu, HI, p 209-232
Bruce LD, Lightner DV, Redman RM (1994) Comparison of traditional and molecular detection methods for baculo- virus penaeid infections in larval Penaeus vannamei J Aquat Anirn Health 6:355-359
Bruce LD, Redman RM, Lightner DV, Bonaml JR (1993) Appli- cation of gene probes to detect a penaeid shrimp baculo-
virus in fixed tissue using in situ hybridization. Dis Aquat Org 17:215-221
Burand J (1991) Molecular biology of the HzV-I and Oryctes
nonoccluded baculoviruses. In: Kurstak E (ed) Viruses of
Invertebrates Marcel Dekker, Inc., New York, p 11 1-126 Chang PS, Lo CF, Kou GH, Lu CC, Chen SN (1993) Punflca-
tion and ampllflcation of DNA from Penaeus monodon-
type baculovirus (MBV). J Invertebr Path01 62:116-120
Chang PS, Lo CF, Wang YC, Kou GH (1996) Idenhfication of white spot syndrome associated baculovirus (WSBV) target organs in the shrimp Penaeus monodon by in situ hybridization. Dis Aquat Org 27:131-139
Chen LC (1990) Aquaculture in Taiwan. The Alden Press, Oxford
Chou HY, Huang CY, Wang CH, Chiang HC, Lo CF (1995)
Pathogenicity of a baculovirus infection causing white spot syndrome in cultured penaeid shrimp in Taiwan. Dis Aquat Org 23:165-173
Crawford A (1994) Nonoccluded baculoviruses. In: Webster RG, Granoff A (eds) Encyclopedia of virology. Academic Press, New York, p 133-139
Erlich HH, Gelfand DH, Saiki RK (1988) Specific DNA ampli-
fication. Nature 331:461-462
Francki RIB, Fauquet CM, Knudson DL, Brown F (1991) Classification and nomenclature of viruses. Arch Virol (Suppl) 2:1-450
Fulks W, Maln KL (1992) Diseases of cultured penaeld shrimp in Asia and the United States. The Oceanic Institute, Honolulu, H1
Johnson PT (1988) Rod-shaped nuclear viruses of crustaceans:
hemocyte-infecting species. Dis Aquat Org 5:111-122
Johnson PT, Lightner DV (1988) Rod-shaped nuclear viruses
of crustaceans: gut-infecting species. Dis Aquat Org 5: 123-141
IOm W, Abele LG (1990) Molecular phylogeny of selected decapod crustaceans based on 18s rRNA nucleotide sequences. J Crust Biol 10:l-13
Lester RJG, Doubrovsky A, Paynter JL, Sambhi SK, Atherton
JG (1987) Light and electron microscope evidence of
baculovirus infection in the prawn Penaeus plebejus. Dis
Aquat Org 3:217-219
Lightner DV (ed) (1996) A handbook of pathology and diag- nostic procedures for diseases of penaeid shrimp. World Aquaculture Soc., Baton Rouge
Lightner DV, Poulos BT, Bruce L. Redrnan RM, Mari J , Bonami JR (1992) New developments in penaeid virology: application of biotechnology in research and disease diag- nosis for shrimp viruses of concern in the Americas. In:
Fulks W, Main KL (eds) Diseases of cultured penaeid
shrimp in Asia and the United States. The Oceanic Insti- tute, Honolulu, HI, p 233-256
Lo CF, Leu JH. Ho CH, Chen CH. Peng SE, Chen YT, Chou CM, Yeh PY. Huang CJ, Chou HY, Wang CH. Kou GH (1996) Detection of baculovirus associated with white spot syndrome (WSBV) in penaeid shrimps using polymerase chain reaction. Dis Aquat Org 25:133-141
Lotz JM (1992) Developing speclflc pathogen-free (SPF) ani- mal populations for aquaculture: a case study for IHHN virus of penaeld shrimp. In: Fulks W, Main KL (eds) Dis- eases of cultured penaeid shrimp in Asia and the United States. The Oceanic Institute, Honolulu, HI, p 258-269 Man J , Bonami JR, Lightner DV (1993) Partial cloning of the
genome of infectious hypodermal and hematopoietic necrosis virus, an unusual parvovirus pathogenic for penaeid shrimps; diagnosis of the disease using a specific probe. J Gen Virol 74:2637-2643
Lo et al.. WSBV detection in arthropods
cloning of the genome of a n unusual shrimp parvovirus
(HPV): use of gene probes in disease diagnosis.
Dis
Aquat Org 22:129-134~Murphy FA. Fauquet CM, Bishop DHL, Ghabrial SA. Jarvis
AW. Martelli GP, Summers MD (1995) Classification and
nomenclature of viruses. Arch Virol (Suppl) 10:l-586 Oste C (1988) Polymerase chain reaction. Biotechniques 6:
162-167
Poulos BT, Mari J , Bonami J R , Redman R. Lightner DV (1994) Use of non-radioactively labeled DNA probes for the detection of a baculovirus from Penaeus monodon (PmSNPV=MBV) by in situ hybridization on fixed tissue. J Virol Methods 49:187-194
Sano T, Nishimura T, Oguma K, Momoyama K, Takeno N Responsible Subject Editor: J. E. Stewart, Dartmouth. Nova Scotia, Canada
(1981) Baculovirus infection of cultured kuruma shrimp, Penaeus japonicus, in Japan. Fish Path01 15:185-191 Wang CH, Lo CF. Leu JH, Chou CM, Yeh PY. Chou HY, Tung
MC, Chang CF, Su MS, Kou GH (1995) Purification and genomic analysis of baculovirus associated with white spot syndrome (WSBV) of Penaeus monodon. Dis Aquat Org 23:239-242
Wongteerasupaya C, Vickers JE, Sriurairatana S, Nash GL, Akarajamorn A, Boonsaeng V, Panyim S, Tassanaka- jon A, Withyachumnarnkul B, Flegel T W (1995) A non- occluded, systemic baculovirus that occurs In cells of ecto- dermal and mesodermal origin and causes high mortality in the black tiger prawn Penaeus monodon. Dis Aquat Org 21:69-77
Manuscript first received: May 8, 1996 Revised version accepted: August 28, 1996