Synthesis and characterization of novel nanocomposite membrane of
sodium titanate/Nafion
®
Yongsheng Wei
a, Liangbo Shen
b, Fanghui Wang
c, Wein-Duo Yang
d, Hong Zhu
c,⁎
,
Zhongming Wang
c, Kefei Han
ca
School of Science, Beijing Jiaotong University, Beijing 100044, PR China
b
Beijing No. 4 High School, Beijing 100034, PR China
c
Institute of Modern Catalysis, Department of Organic Chemistry, State Key Laboratory of Chemical Resource Engineering, Beijing University of Chemical Technology, Beijing 100029, PR China
d
Department of Chemical and Materials Engineering, National Kaohsiung University of Applied Sciences, Kaohsiung 807, Taiwan, ROC
a b s t r a c t
a r t i c l e i n f o
Article history:
Received 8 November 2010 Accepted 25 February 2011 Available online 3 March 2011 Keywords:
Nanocomposite membrane Sodium titanate nanotubes FTIR
Nanocomposites Fuel cell
Novel nanocomposite membrane of sodium titanate/Nafion based on sodium titanate nanotubes with Nafion®
were prepared by solvent casting techniques. Nanotubes of sodium titanate were synthesized by hydrothermal method. TEM, XRD, and FTIR were employed to characterize the crystal phase, microstructure, and other physicochemical properties of the membrane and the nanotube samples. FTIR results showed us that the nanotube material of Na2Ti3O7has existed in the nanocomposite membrane of Na2Ti3O7/Nafion. The
existence of sodium titanate nanotubes in Nafion®improves the methanol crossover and makes promising
practical value of blocking methanol in direct methanol fuel cells.
© 2011 Elsevier B.V. All rights reserved.
1. Introduction
Fuel cells have recently attracted considerable attention as an alternative energy-conversion technology that meets the demand of
sustainable development [1,2]. Direct methanol fuel cell (DMFC)
using an aqueous solution of methanol as fuel is one kind of PEMFC and is thought to be the most promising power generating system for
applications in portable electronic devices[3–5]. Proton exchange
membrane is one of key materials whether in DMFC. Presently,
commercially available perfluorinated membranes including Nafion®
have the problem of methanol crossover and reduce the performance of DMFC, which slowed the commercial process of direct methanol
fuel cell[6,7].
After literatures study, we found that modification of the
perfluorinated sulfonic membrane is a doable method. It is a new
direction to prepare organic/inorganic composite membrane this way. Nanotubes of sodium titanate showed the special performance of conductivity and the water holding, which meets the requirements of
proton exchange membrane for better performance[8,9].
In this study, the sodium titanate nanotubes of micro-diameter
were synthesizedfirst, and then they were casted with Nafion®to
change the structure of the novel nanocomposite membrane.
2. Experimental 2.1. Materials
Nafion®solution (5 wt.%) was purchased from DuPont, USA. N,
N-dimethylformamide (DMF) was purchased from Beijing Yili Fine Chemicals Ltd., China. Methanol and pure sulfuric acid were purchased from Beijing Chemicals Ltd., China. Deionized water was used throughout the study.
2.2. Synthesis of sodium titanate nanotubes (Na2Ti3O7)
2 kg of anatase TiO2 (80–200 nm) is weighed into a flask, and
15 ml of 10 M NaOH solution is added and then stirred with ultrasound for 2 h. The mixture is transferred into a high-pressure
reaction kettle lined with Teflon on the inner wall and sealed. The
water-heated mixture is allowed to react at 130 °C for 3 days. The reaction kettle was then cooled under natural condition to room
temperature and taken out, and the contentfiltered. A white solid was
obtained, which was then washed with deionized water until the pH was 7 and dried to yield sodium titanate nanotubes.
2.3. Preparation of sodium titanate/Nafion®nanocomposite membranes
Sodium titanate/Nafion® nanotube composite membrane was
prepared. The base membranes were prepared by a solvent casting
technique in which the low-boiling-point alcohols in Nafion®
solution Materials Letters 65 (2011) 1684–1687
⁎ Corresponding author. Tel.: +86 10 82161887. E-mail address:[email protected](H. Zhu).
0167-577X/$– see front matter © 2011 Elsevier B.V. All rights reserved. doi:10.1016/j.matlet.2011.02.081
Contents lists available atScienceDirect
Materials Letters
plays important role of net, which can prevent the methanol go
through the Nafion membrane and reduce the methanol crossover.
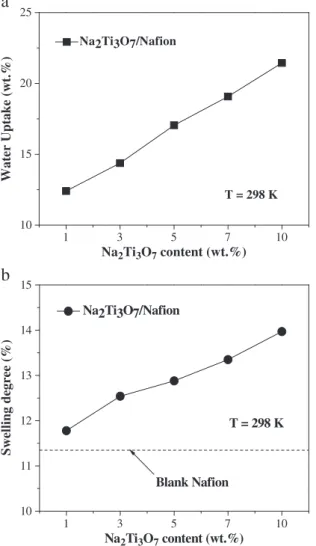
3.6. Water uptake
For most proton conductive polymers, water acts as the carrier for proton transportation through the membrane. Thus, adequate hydration of electrolyte membranes is crucial for high proton
conductivity. The results of water uptake for the Na2Ti3O7/Nafion®
composite membrane with different quality of nanotubes at room
temperature at different temperatures are shown inFig. 5-a. Water
uptake increases with the quality of the nanotubes. The water uptake
of the Na2Ti3O7/Nafion®composite membrane is higher than that of
Nafion®112 which is 10.4 wt.% at room temperature. It is believed that
Ti–OH and H2O can form hydrogen bonds, which increases the
absorption of water. Besides, the nanotubes have a very high specific
surface area and may also lead to higher water adsorption. Overall, these results show that the composite membrane has higher water uptake and may lead to better performance in a fuel cell.
3.7. Swelling degree
The swelling degree of composite membrane affects the mem-brane performance. Too large a swelling degree will adversely affect the mechanical properties of the membrane and may crack the fuel cell and shorten its life.
The water swelling results of composite membranes of different
nano-material contents are shown inFig. 5-b. The swelling degree
increases with the nano-material content. Generally speaking, the change of nano-material content has little effect on the dimensions of the membrane. This indicates that composite membranes have good resistance to swelling. The reason is that there are abundant hydroxyl groups on the surface of nanotubes and these hydroxyl groups are highly hydrophilic. After the sodium titanate nanotubes have been
added to Nafion®and acidified, the sodium ions are replaced by the
smaller hydrogen ions. The collapse of the original tubular structure leaves more space to accept water molecules without affecting the membrane volume. The introduction of water molecules provides the physical foundation for proton conductivity in the membrane.
4. Conclusions
Novel nanocomposite membranes of Na2Ti3O7/Nafion®for direct
methanol fuel cell are prepared by solvent casting techniques. The base material of sodium titanate nanotubes was synthesized by hydrothermal method. TEM and SEM test results show us that the diameter of nanotubes is in nanometer orders of magnitude and lies
the same direction as the nanocomposite membrane of Na2Ti3O7/
Nafion®. Introduction of nanotubes of Na
2Ti3O7 affect the proton
conductivity of the nanocomposite membrane of Na2Ti3O7/Nafion®.
More meaning is that the methanol crossover can be improved. The nanotubes have dual role of blocking methanol crossover and better
ability in water holding. Na2Ti3O7/Nafion®composite membrane is
expected to have better performance and further promising in direct methanol fuel cell applications.
Acknowledgements
The authors gratefully acknowledge the financial support from
International S&T Cooperation Program of China (No. 2006DFA61240 and No. 2009DFA63120), the National Science Foundation of China (No. 50674006, No. 20876013, and Key Program No. 20636060).
References
[1] Yi Baolian, Chemical industrial publishing, Beijing, 2003:2–10 2003.
[2] O'Hayre Ryan, Cha Suk-Won, Colella Whitney, Prinz Fritz B. New York: Wiley; 2005.
[3] Aricò AS, Srinivasan S, Antonucci V. Fuel Cells 2001;1:133.
[4] Antonucci PL, Aric AS, Cret P, Ramunni E. Solid State Ionics 1999;125:431–7. [5] Neburchilov V, Martin J, Wang HJ, Zhang JJ. J Power Sources 2007;169:221–38. [6] Seo Sang Hern, Lee Chang Sik. Applied Energy 2010;87:2597–604.
[7] Valdez TI, Narayanan SR. Proton Exchange Membrane Symposium Boston, MA, USA; 1998.
[8] Baliteau S, Sauvet A-L, Lopez C, Fabry P. Solid State Ionics 2007;178:1517–22. [9] Zhang Yanyan, Fu Wuyou, Yang Haibin, Li Minghui, Li Yixing, Zhao Wenyan, et al.
Sensors Actuators B 2008;135:317–21.
[10] Shao ZG, Joghee P, Hsing IM. Membr Sci 2004;229:43–51.
[11] Armstrong AR, Armstrong G, Canales J, Bruce PG. Angew Chem Int Ed 2004;43: 2286–8.
[12] Falk M. Can J Chem 1980;58:1495–501.
1 3 5 7 10 10 15 20 25 T = 298 K Na2Ti3O7/Nafion
Water Uptake (w
t.%)
Na
2Ti
3O
7content (wt.%)
1 3 5 7 10 10 11 12 13 14 15T = 298 K
Na2Ti3O7/Nafion
Swelling degree (%)
Na
2Ti
3O
7content (wt.%)
Blank Nafion
b
a
Fig. 5. (5-a) Water uptake of composite membranes with for different nanotubes contents at room temperature. (5-b) Swelling degree of composite membranes at room temperature.
1687 Y. Wei et al. / Materials Letters 65 (2011) 1684–1687