Comparative Biochemistry and Physiology Part B 136(2003) 913–920
1096-4959/03/$- see front matter䊚 2003 Elsevier Inc. All rights reserved. doi:10.1016/j.cbpc.2003.09.002
Serum estradiol-17b and testosterone levels during silvering in wild
Japanese eel
Anguilla japonica
Yu-San Han , I-Chiu Liao , Wann-Nian Tzeng , Yung-Sen Huang , John Yuh-Lin Yu *
a b a c d, Institute of Zoology, College of Life Science, National Taiwan University, Taipei, Taiwan, ROCa
Taiwan Fisheries Research Institute, 199 Hou-Ih Road, Keelung, Taiwan, ROC b
National Museum of Marine Biology and Aquarium, 2 Houwan Road, Checheng, Pingtung, Taiwan, ROC c
Endocrinology Laboratory, Institute of Zoology, Academia Sinica, Taipei, Taiwan, ROC d
Received 19 June 2003; received in revised form 5 September 2003; accepted 6 September 2003
Abstract
To understand the changes of serum levels of sex steroids in the wild Japanese eelAnguilla japonica during silvering
process, eels collected from the Kaoping River of Taiwan from August 2000 through June 2001 were examined. The maturational stages of female eels before and during silvering were divided into four stages: juvenile, sub-adult, pre-silver and pre-silver stages based on skin coloration and oocyte diameter. Male eels were investigated only in the pre-silver stage. Radioimmunoassays were employed to measure serum levels of estradiol-17b (E ) and testosterone (T). The2
mean liver mass of the female eels increased significantly during silvering, but the mean hepatosomatic index remained constant. In contrast, mean ovarian mass and gonadosomatic index increased significantly during silvering. Serum concentrations of E in females increased significantly during silvering2 (P-0.05), while E was undetectable in silver2
males. The mean serum T concentrations increased significantly in females (P-0.05) during silvering, with lowest
mean values in the juvenile stage and highest mean value in the silver stage. The mean serum T level in the silver males was significantly lower than in silver females(P-0.05). In conclusion, both serum E and T concentrations increased2
with ovarian development of wild Japanese eels during silvering, while serum E was undetectable in the silver male2
eels. The findings support the idea that androgen, but not estrogen, plays a major role in silvering process of the eels in both sexes.
䊚 2003 Elsevier Inc. All rights reserved.
Keywords: Japanese eel Anguilla japonica; Silvering; Sex steroid; Estradiol-17b; Testosterone; Gonadal development; Liver;
Vitellogenesis
1. Introduction
Catadromous anguillid eels have a complex life cycle. After a long growth period in rivers(4–20 years), eels undergo significant morphological and physiological changes from yellow to silver stages
(process known as ‘silvering’) before the seawater *Correspondence author. Tel.: q886-2-27899509; fax: q 886-2-27858059.
E-mail address: [email protected](J.Y.L. Yu).
spawning migration (Tesch, 1977; Jessop, 1987; Han et al., 2001, 2003b). The morphological mod-ifications include a change in belly color from yellow to silverybronze and in back and pectoral
fins from whiteygray to black (Tesch, 1977; Han
et al., 2003b), and increased eye size (Pankhurst, 1982; Rohr et al., 2001; Han et al., 2003b). The
physiological changes include degeneration of the digestive tract(Pankhurst and Sorensen, 1984; Han
(Pank-hurst, 1982), more developed swim bladder
(Kleckner, 1980; Yamada et al., 2001), higher
density of branchial chloride cells(Fontaine et al.,
1995), higher muscle fat contents (Han et al.,
2001), and more developed gonads (Colombo et al., 1984; Jessop, 1987; Lokman and Young, 1998; Lokman et al., 1998; Han et al., 2003b). These
modifications of silver eels have been proposed to be a pre-adaptation for marine environment of the spawning migration (Pankhurst, 1982; Fontaine and Dufour, 1991; Fontaine et al., 1995; Han et
al., 2003b).
Under conditions of cultivation, both sexes of eels have immature gonads, but can be induced to become fully mature by multiple injections of salmon pituitary homogenates or human chorionic gonadotropin (HCG) (Yamamoto et al., 1974;
Ohta et al., 1996). It has been shown that the skin color of European yellow eel can be altered by testosterone, although the degree of alteration was not always repeatable(Fontaine, 1994). In
Anguil-la australis, injection of 11-ketotestosterone
(11-KT) caused significant gonadal development and silvering-related changes, such as chisel-shaped snouts, black and large pectoral fins and enlarged eyes and livers (Rohr et al., 2001). In European
eel A. anguilla, injection of HCG could
simulta-neously induce the development of gonads and the degeneration of alimentary tract (Pankhurst and
Sorensen, 1984). Meanwhile, changes of skin
col-or, eye size, and digestive tract were found to be synchronous with gonadal development (Han et al., 2003b), and the expression levels of
gonado-tropin mRNAs were significantly increased during silvering of the Japanese eel (Han et al., 2003a).
Accordingly, these findings imply that the hypo-thalamus-pituitary-gonad(HPG) axis may play an important role in the silvering process.
The circulating estradiol-17b(E ) and testoster-2
one (T) levels in the Anguilla dieffenbachii and
A. australis were higher in the silver stage than in
the yellow stage(Lokman and Young, 1998;
Lok-man et al., 1998). In American eel A. rostrata, plasma E2 levels were higher in the migrating silver eels than in resident yellow eels(Cottrill et al., 2001). To our knowledge, no information is
yet available with respect to the corresponding changes in the serum E2 and T levels during silvering of the wild Japanese eels. We, therefore, investigated the changes of serum E and T levels2
for better understanding the sex steroid profiles of
Japanese eels in relation to the HPG axis during eel silvering.
2. Materials and methods
2.1. Eel collection and morphometric measure-ments
Wild Japanese eels were collected by eel traps in the lower reach of Kaoping River of southwest Taiwan (1208509 E and 228409 N) from August 2000 to June 2001 as described previously (Han et al., 2003a). Briefly, the eel pots were set at the bottom of the riverside in the estuary, where they are influenced by tidal cycles. The eel pots were set in the daytime ebb tide and eels were collected in the next ebb tide over approximately 24 h. The eels were trapped in the eel pots when foraging in the nighttime. After capture, eels were stunned with ice and immediately transferred to the labor-atory for processing. The total length (TL, "0.1
cm), body mass (BW, "0.1 g), and horizontal
and vertical diameters("0.01 mm) of the left eye
of the eels were measured before decapitation for blood collection. The ocular index(OI), a measure
of eye area to total length of the eel (Pankhurst, 1982), was calculated as follows: OIsw((AqB)y 4) =2 pyTLx=100%, Where A and B are
horizon-tal and vertical diameters of the left eye, respectively. The gonad mass (GW, "0.01 g), liver mass(LW, "0.01 g) and digestive tract mass
(DW, "0.01 g) were measured; the gonadosomatic
index (GSI), hepatosomatic index (HSI) and
digestosomatic index (DSI) were estimated
according to the formulas GSIs100%=wGW(g)y
BW (g)x, HSIs100%=wLW (g)yBW (g)x, and
DSIs100%=wDW(g)yBW (g)x, respectively. For
examination of the sex and maturity of eels, gonads were fixed in Bouin’s solution, sectioned and stained with hematoxylin and eosin for histology. The mean oocyte diameter (OD, "1 mm) was calculated from randomly selected 20 round oocytes with complete nucleus. Maturation status of oocytes and spermatogonia were determined according to Yamamoto et al. (1974) and Miura et al.(1991), respectively.
In our previous investigation, the maturity of wild female Japanese eels before and during sil-vering was divided into three stages(yellow,
pre-silver and pre-silver) based on skin color and
histological observations of ovarian development
915
Y.-S. Han et al. / Comparative Biochemistry and Physiology Part B 136 (2003) 913–920
yellow eels were further divided into juvenile and sub-adult stages based on oocyte diameter for better comparison(Table 1). Eels with OD below
65 mm were grouped in the juvenile stage, while eels with OD ranging between 66 and 90 mm were grouped in sub-adult stage. The oocytes of the pre-silver eels have OD ranges between 91 and 130
mm, and oocytes of the silver eels have OD above
131 mm. Since the sex ratio of the eels was strongly skewed to the females in the Kaoping River (Tzeng et al., 2002), considerably lower numbers of males were trapped; thus only silver males were investigated in the present study.
2.2. Radioimmunoassays (RIA) of serum levels of
estradiol-17b and testosterone
The RIA protocols of estradiol-17b (E ) and2
testosterone (T) were similar to those described
by Yu et al.(1988, 1991).
2.3. Statistical analysis
Data were analyzed using the software SPSS
(SPSS Inc., USA). Significant differences in mean
values among developmental stages of the female eels were conducted by one-way analysis of vari-ance(ANOVA) followed by Tukey’s HSD multi-ple-comparison test. The significance of differences in mean values between sexes were assessed by Student’st-test. Differences were
con-sidered significant atP-0.05.
3. Results
3.1. Morphometric indices during silvering
The mean TL and BW of female Japanese eels significantly increased during silvering(P-0.05),
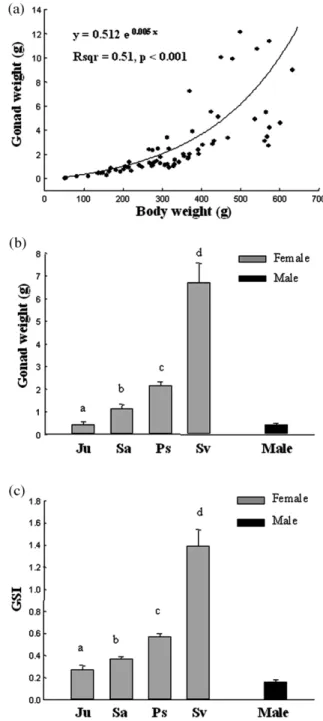
but no significant difference was found between pre-silver and silver stages (P)0.05, Table 1). The mean OI of the females increased, while mean DSI decreased significantly during silvering (P-0.05, Table 1). The mean liver mass (mean"S.E.) of the female eels increased during eel silvering: 1.65"0.28 g in the juvenile stage, 2.66"0.23 g in the sub-adult stage, 3.74"0.36 g in the pre-silver stage and 4.59"0.31 g in the pre-silver stage
(Fig. 1). The mean liver mass of the male eels
was 3.54"0.43 g. However, the mean HSI were not significantly different among the developmen-tal stages in female eels ( juvenile: 1.09"0.07;
sub-adult: 1.05"0.05; pre-silver: 1.03"0.04; and silver: 0.97"0.05) because of the good linear correlation between body mass and liver mass
(Fig. 1). The mean HSI of the silver males was
1.13"0.08 (Fig. 1). The mean gonad mass increased during eel silvering: 0.45"0.11 g in the juvenile stage, 1.16"0.17 g in the sub-adult stage, 2.15"0.18 g in the pre-silver stage, and 6.72"0.84 g in the silver stage(Fig. 2). The mean
gonad mass of the male eels was 0.44"0.06 g. The mean GSI of the female Japanese eels also significantly increased during silvering because of the positively exponential correlation existing between body mass and gonad mass(Fig. 2). The mean GSI of the silver males was 0.16"0.02(Fig. 2). The mean TL, BW, and GSI of the silver males
were significantly lower than those of the silver females(P-0.05), but the OI, DSI and HSI were similar in both sexes of silver stage (P)0.05)
(Table 1, Figs. 1 and 2).
Ovaries of juvenile eels contained oocytes pre-dominantly in chromatin nucleolus stage. Ovaries of sub-adult eels contained also predominantly chromatin nucleolus stage oocytes but with larger OD. In the pre-silver eels, the oocytes grew rapidly and were mainly in peri-nucleolus stage. The first oil drops became apparent at periphery of the oocytes. In the silver eel, the oocytes continued to grow and the oil drops accumulated and filled the whole cytoplasm. They were mainly in oil-drop stage. In silver males, the spermatogonia in the nests, mainly in late type B stage, were actively in mitosis. More advanced spermatids or spermat-ozoa were not found.
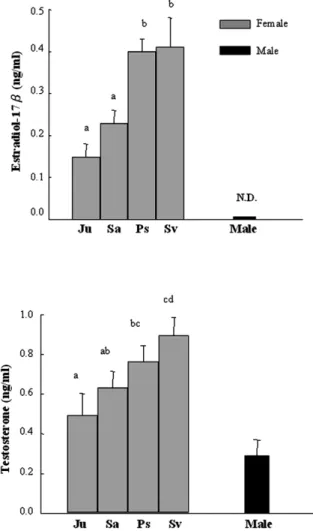
3.2. Serum estradiol-17b levels during silvering
In silver males, the serum E levels were unde-2
tectable (-10 pgyml) (Fig. 3a). In females, on
the contrast, the mean serum E2 levels
(mean"S.E.) increased with stages of ovarian
development during silvering: 0.15"0.03 ngyml
in the juvenile stage, 0.23"0.03 ngyml in the
sub-adult stage, 0.40"0.03 ngyml in the pre-silver
stage, and 0.41"0.07 ngyml in the silver stage.
The mean values were significantly different among stages (Fs9.01)F s2.74,
P-0.05, 69
( )
0.001), and the differences were significant
between juvenileysub-adult and pre-silverysilver
Y.-S. Han et al. / Comparative Biochemistry and Physiology Part B 136 (2003) 913–920 Table 1
The mean("S.E.) morphometric indices of the wild Japanese eel among different developmental stages
Female Tukey’s HSD Male
Juvenile Sub-adult Pre-silver Silver Silver
n 12 20 22 16 6
Collection time August, September, August, September, August, September, September, October, December 2000 October 2000 October 2000 October 2000 December 2000
March, 2001 February, March, May 2001 June 2001 February 2001 February 2001 TL(mm) 475.6"23.2 570.9"8.3 620.8"10.2 657.9"13.5 Ju-Sa-PssSv 603.0"24.2 (367–596) (506–647) (558–744) (570–756) (515–675) BW(g) 150.2"23.3 247.0"14.1 386.0"22.6 474.7"24.1 Ju-Sa-PssSv 293.5"41.3 (52.2–313.7) (137.3–347.8) (216.3–573.6) (289.5–633.4) (182.4–452.1) OI 2.81"0.21 3.36"0.17 3.58"0.16 4.88"0.27 Ju-SasPs-Sv 5.56"0.45 (1.62–3.93) (2.38–5.59) (2.15–4.71) (3.82–6.92) (4.20–6.90) DSI 1.65"0.06 1.64"0.07 1.46"0.07 0.56"0.06 JusSasPs)Sv 0.46"0.09 (1.05–2.63) (1.18–2.31) (0.91–2.30) (0.24–1.03) (0.25–0.85) OD(mm) 57.7"2.1 78.8"1.3 105"2.3 176.3"8.7 Ju-Sa-Ps-Sv — (50–65) (66–90) (91–130) (131–225) GSI 0.27"0.04 0.37"0.02 0.57"0.03 1.39"0.15 Ju-Sa-Ps-Sv 0.16"0.02 (0.04–0.42) (0.27–0.52) (0.35–0.85) (0.55–2.43) (0.10–0.19) HSI 1.09"0.07 1.05"0.05 1.03"0.04 0.97"0.05 JusSasPssSv 1.13"0.08 (0.87–1.64) (0.63–1.44) (0.71–1.41) (0.73–1.62) (1.03–1.43)
TL: total length; BW: body mass; OI: ocular index; DSI: digestosomatic index; OD: oocyte diameter; GSI: gonadosomatic index; HSI: hepatosomatic index; Ju: juvenile; Sa: sub-adult; Ps: pre-silver; Sv: silver.P-0.05 for all significance differences. Numerals in the parentheses indicate range.
917
Y.-S. Han et al. / Comparative Biochemistry and Physiology Part B 136 (2003) 913–920
Fig. 1. Changes in liver mass and its correlation to body mass during silvering of the wild Japanese eels.(a) Regressions of
liver mass on body mass in females.(b) Mean liver masss in
four different stages of females and in silver males.(c) Mean
HSIs in four different stages of females and in silver males. Ju: juvenile; Sa: sub-adult; Ps: pre-silver; Sv: silver. Different letters above the histograms indicate that the differences are statistically significant(P-0.05). The numbers of each stage
are shown in the Table 1.
Fig. 2. Changes in gonad mass and its correlation to body mass during silvering of the wild Japanese eels.(a) Regressions of
gonad mass on body mass in females.(b) Mean gonad masss
in four different stages of females and in silver males. (c)
Mean GSIs in four different stages of females and in silver males. Ju: juvenile; Sa: sub-adult; Ps: pre-silver; Sv: silver. Different letters above the histograms indicate that the differ-ences are statistically significant(P-0.05). The numbers of
each stage are shown in the Table 1.
3.3. Serum testosterone levels during silvering
During silvering, the mean serum T levels in female eels increased gradually with stages of
Fig. 3. Serum E and T levels in different stages of the wild2 Japanese eels. Ju: juvenile; Sa: sub-adult; Ps: pre-silver; Sv: silver. Different letters above the histograms indicate that the differences are statistically significant(P-0.05). The numbers
of each stage are shown in the Table 1.
ovarian development during silvering: 0.49"0.11 ngyml in the juvenile stage, 0.63"0.08 ngyml in
sub-adult stage, 0.76"0.08 ngyml in pre-silver
stage, and 0.89"0.09 ngyml in silver stage. The
mean values were significantly different among stages (Fs2.95)F0.05, 69s2.74, Ps0.04) (Fig.
( )
3b). Mean serum T level in silver males was
0.29"0.08 ngyml, which was significantly lower
than that of silver female eels(P-0.001, Fig. 3b). 4. Discussion
In cultured yellow Japanese eels before matu-ration induction by weekly injection of salmon pituitary homogenate the serum E levels averaged2
approximately 0.27 ngyml, which tended to
increase (up to 2.5 ngyml) after 12 weeks and showed a further increase (5.6–15.8 ngyml) after
completion of vitellogenesis(Ijiri et al., 1995). In
wild silverA. australis and A. dieffenbachii, mean
plasma E2 levels were also significantly higher than yellow eels (Lokman et al., 1998). In
Amer-ican eel, the mean plasma E levels of the silver2
eels were higher than those of yellow eels, and paralleled the development of the ovary (Cottrill
et al., 2001). In the present study, we also found that the mean serum E2 levels in wild female Japanese eels increased significantly during silver-ing (Fig. 3a). The changes in serum E levels2
during silvering were in conformity with the pro-gress of ovarian development (Table 1 and Fig. 3). We have demonstrated that the transcript levels of the pituitary glycoprotein hormone a, gonado-tropin I b and II b mRNAs significantly increased during silvering process of the Japanese eel(Han
et al., 2003a). Accordingly, the increasing levels
of serum E2 in the wild Japanese eels are in conformity with their increasing transcript levels of the gonadotropins, reflecting an enhanced activ-ity of the pituitary-gonad axis during eel silvering. In fish, E is also responsible for the production2
of the hepatic vitellogenin, the yolk precursor molecules (Yu et al., 1981; Wahli, 1988; Teo et al., 1998). In the present study, although the mean liver mass increased during silvering in female eels, the mean HSI did not change significantly
(Fig. 1). No significant difference of HSI was also
observed between yellow stage and silver stage of female American eels(Lewander et al., 1974) and
European eels (Sbaihi et al., 2001). The HSI
remained constant in conger eels and European eels when their GSIs, respectively, increased from 0.1 to 4.8 and from 0.1 to 2.2(Sbaihi et al., 2001).
However, it was shown that a 50–60% increase of HSI was observed between non-migratory and migratory stages in both females of A. australis
and A. dieffenbachii (Lokman et al., 1998). The
GSIs increased from 0.21 to 3.27 with oocytes mainly in the oil-droplets and early vitellogenic stages in A. australis and from 0.08 to 7.22 with
oocytes mainly in the earlyymid vitellogenic stages
in A. dieffenbachii, respectively (Lokman et al.,
1998). These findings suggest that the higher mean
HSI in the migratory silver females may be due to active vitellogenesis. It has been shown that the oocytes of the hormone induced Japanese eels progress through vitellogenesis in approximately
919
Y.-S. Han et al. / Comparative Biochemistry and Physiology Part B 136 (2003) 913–920
200–700 mm diameter(Ijiri et al., 1995). Lokman et al. (1998) showed that the early vitellogenetic oocytes started approximately 220 mm diameter in bothA. australis and A. dieffenbachii. As indicated
in Table 1, the mean OD of the silver females was 176 mm with a range of 131 to 225 mm. Histolog-ical examination revealed that the oocytes of silver females were accumulated with oil drops. Presum-ably they were in the beginning phase of early vitellogenesis and, therefore the HSIs did not vary significantly during silvering.
In the present study, the mean serum T levels of the female eels were significantly increased during silvering(Fig. 3). Previous studies indicat-ed that the high serum T levels in the females could promote oogenesis in the Japanese eel(Lin
et al., 1991) and coho salmon (Oncorhynchus
mykiss) (Fitzpatrick et al., 1994), and the existence
of the androgen receptors in the ovary of the Japanese eel was also proved (Ikeuchi et al.,
1999). Such findings support the idea that
andro-gen is involved in ovarian maturation in eels and salmonids. In addition, androgen may also contrib-ute to the developments of secondary sexual char-acteristics; osmoregulation, skin coloration and thickness and increased red muscle mass and heart size in salmonids(Idler et al., 1961; Schmidt and Idler, 1962; Thorarensen et al., 1996). In the present study, serum T levels were higher in silver females than in silver males (Fig. 3). It was also
demonstrated that the plasma T levels were higher in migratory silver females(Lokman et al., 1998) than in non-migratory yellow males (Lokman et
al., 1998) in A. australis; but plasma T levels were similar between the both sexes ofA. dieffenbachii
(Lokman and Young, 1998; Lokman et al., 1998).
It thus appears that sexual difference of circulating T levels is likely varied with different species of eels. Furthermore, it was also found that plasma 11-KT levels were higher in males than in females and that 11-KT levels were higher than T levels in both sexes of A. australis and A. dieffenbachii
(Lokman and Young, 1998; Lokman et al., 1998).
It is shown that the skin color of European yellow eel can be altered by T(Fontaine, 1994), and that administration of 11-KT induces silvering-related changes in immature femaleA. australis (Rohr et
al., 2001). As demonstrated in the present study,
E was undetectable in serum of the male Japanese2
eels. Our findings, together with the observations by others, thus support the idea that androgen (T
and 11-KT), but not E , plays a major role in the2
silvering process of eels.
In conclusion, the changes in the serum E and2
T levels during silvering of the wild female Japa-nese eels observed in this study are in conformity with their corresponding changes in the expression levels of gonadotropin mRNAs(Han et al., 2003a).
The serum E levels were undetectable in silver2
males. These results together with findings by others thus support that androgen, but not estrogen, plays a major role in silvering process of eels of both sexes.
Acknowledgments
This study was financially supported by the National Science Council (NSC 89-2313-B056-008, NSC 90-2313-B056-005) and Academia Sin-ica, Taiwan, ROC. The authors are grateful to Mr G.H. Cheng and Mr J.T. He for sample collection and gonadal histology.
References
Colombo, G., Grandi, G., Rossi, R., 1984. Gonad differentia-tion and body growth inAnguilla anguilla L. J. Fish Biol.
24, 215–228.
Cottrill, R.A., McKinley, R.S., van der Kraak, G., Dutil, J.D., Reid, K.B., McGrath, K.J., 2001. Plasma non-esterified fatty acid profiles and 17b-oestradiol levels of juvenile immature and maturing adult American eels in the St Lawrence river. J. Fish Biol. 59, 364–379.
Fitzpatrick, M.S., Gale, W.L., Schreck, C.B., 1994. Binding characteristics of an androgen receptor in the ovaries of coho salmon,Oncorhynchus mykiss. Gen. Comp.
Endocri-nol. 95, 399–408.
Fontaine, Y.A., 1994. L’argenture de l’anguille: metamorphose,´ anticipation, adaptation. Bull. Fr. Peche Piscic. 225,ˆ 171–185.
Fontaine, Y.A., Dufour, S., 1991. The eels: from life cycle to reproductive endocrinology. Bull. Inst. Zool. Acad. Sinica Monogr. 16, 237–248.
Fontaine, Y.A., Pisam, M., Moal, C.L., Rambourg, A., 1995. Silvering and gill ‘mitochondria-rich’ cells in the eel,
Anguilla anguilla. Cell Tissue Res. 281, 465–471.
Han, Y.S., Tzeng, W.N., Huang, Y.S., Liao, IC., 2001. Silvering in the eel: changes in morphology, body fat content, and gonadal development. J. Taiwan Fish. Res. 9, 119–127. Han, Y.S., Liao, IC., Huang, Y.S., Tzeng, W.N., Yu, J.Y.L.,
2003a. Profiles of PGH-a, GTH I-b, and GTH II-b mRNA transcript levels at different ovarian stages in the wild female Japanese eelAnguilla japonica. Gen. Comp.
Endo-crinol. 133, 8–16.
Han, Y.S., Liao, IC., Huang, Y.S., He, J.T., Chang, C.W., Tzeng, W.N., 2003b. synchronous changes of morphology and gonadal development of silvering Japanese eelAnguilla japonica. Aquaculture 219, 783–796.
Idler, D.R., Bitners, I.I., Schmidt, P.J., 1961. 11-ketotestoster-one: an androgen for sockeye salmon. Can. J. Biochem. Physiol. 39, 1737–1742.
Ijiri, S., Kazeto, Y., Takeda, N., Chiba, H., Adachi, S., Yamauchi, K., 1995. Changes in serum steroid hormones and steroidogenic ability of ovarian follicles during artificial maturation of cultivated Japanese eel, Anguilla japonica.
Aquaculture 135, 7–16.
Ikeuchi, T., Todo, T., Kobayashi, T., Nagahama, Y., 1999. cDNA cloning of a novel androgen receptor subtype. J. Biol. Chem. 274, 25 205–25 209.
Jessop, B.M., 1987. Migrating American eels in Nova Scotia. Trans. Am. Fish. Soc. 116, 161–170.
Kleckner, W.B., 1980. Swim bladder volume maintenance related to initial oceanic migratory depth in silver-phase
Anguilla rostrata. Science 208, 1481–1482.
Lewander, K., David, G., Johansson, M.L., Larsson, A., Lid-˚ man, U., 1974. Metabolic and hematological studies on the yellow and silver phases of the European eel, Anguilla anguilla L.–I. Carbohydrate, lipid, protein and inorganic ion
metabolism. Comp. Biochem. Physiol. B 47, 571–581. Lin, H.R., Zhang, M.L., Zhang, S.M., Kraak, G.V.D., Peter,
R.E., 1991. Stimulation of pituitary gonadotropin and ovar-ian development by chronic administration of testosterone in female Japanese silver eel,Anguilla japonica.
Aquacul-ture 96, 87–95.
Lokman, P.M., Young, G., 1998. Gonad histology and plasma steroid profiles in wild New Zealand freshwater eel (Anguil-la dieffenbachia and A. australis) before and at the onset
of the natural spawning migration. II. males. Fish Physiol. Biochem. 19, 339–347.
Lokman, P.M., Vermeulen, G.D., Lambert, J.G.D, Young, G., 1998. Gonad histology and plasma steroid profiles in wild New Zealand freshwater eel(Anguilla dieffenbachia and A. australis) before and at the onset of the natural spawning
migration. I. females. Fish Physiol. Biochem. 19, 325–338. Miura, T., Yamauchi, K., Nagahama, Y., Takahashi, H., 1991. Induction of spermatogenesis in male Japanese eel,Anguilla japonica, by a single injection of human chorionic
gonad-otropin. Zool. Sci. 8, 63–73.
Ohta, H., Kagawa, H., Tanaka, H., Okuzawa, K., Hirose, K., 1996. Milt production in the Japanese eelAnguilla japonica
induced by repeated injections of human chorionic gonado-tropin. Fish. Sci. 62, 44–49.
Pankhurst, N.W., 1982. Relation of visual changes to the onset of sexual maturation in the European eelAnguilla anguilla (L.). J. Fish Biol. 21, 127–140.
Pankhurst, N.W., Sorensen, P.W., 1984. Degeneration of the alimentary tract in sexually maturing European Anguilla
anguilla (L.) and American eels Anguilla rostrata (Le
Sueur). Can. J. Zool. 62, 1143–1149.
Rohr, D.H., Lokman, P.M., Davie, P.S., Young, G., 2001. 11-Ketotestosterone induces silvering-related changes in imma-ture female short-finned eels, Anguilla australis. Comp.
Biochem. Physiol. A 130, 701–714.
Sbaihi, M., Fouchereau-Peron, M., Meunier, F., Elie, P., Mayer, I., Burzawa-Gerard, E., et al., 2001. Reproductive biology of the conger eel from the south coast of Brittany, France and comparison with the European eel. J. Fish Biol. 59, 302–318.
Schmidt, P.J., Idler, D.R., 1962. Steroid hormones in the plasma of salmon at various stages of maturation. Gen. Comp. Endocrinol. 2, 204–241.
Teo, B.Y., Tan, N.S., Lim, E.H., Lam, T.J., Ding, J.L., 1998. A novel piscine vitellogenin gene: Structural and functional analyses of estrogen-inducible promoter. Mol. Cell. Endo-crinol. 146, 103–120.
Tesch, F.W., 1977. The Eel. Biology and Management of Anguillid Eels. Chapman and Hall, London.
Thorarensen, H., Young, G., Davie, P.S., 1996. 11-Ketotestos-terone stimulates growth of heart and red muscle in rainbow trout. Can. J. Zool. 74, 912–917.
Tzeng, W.N., Han, Y.S., He, J.T., 2002. The sex ratios and growth strategies of wild and captive Japanese eelsAnguilla japonica. In: Small, B., MacKinlay, D.(eds.), Developments
in Understanding Fish Growth. International Congress on the Biology of Fish. University of British Columbia, Van-couver, Canada, pp. 25–42.
Wahli, W., 1988. Evolution and expression of vitellogenin genes. Trends Genet. 4, 227–232.
Yamada, Y., Zhang, H., Okamura, A., Tanaka, S., Horie, N., Nikawa, N., et al., 2001. Morphological and histological changes in the swim bladder during maturation of the Japanese eel. J. Fish Biol. 58, 804–814.
Yamamoto, K., Morioka, T., Hiroi, O., Omori, M., 1974. Artificial maturation of female Japanese eels by the injection of salmonids pituitary. Bull. Jpn. Soc. Sci. Fish. 40, 1–7. Yu, J.Y.L., Dickhoff, W.W., Swanson, P., Gorbman, A., 1981.
Vitellogenesis and its hormonal regulation in the Pacific hagfish, Eptatretus stouti L. Gen. Comp. Endocrinol. 43,
492–502.
Yu, J.Y.L., Liaw, J.J., Chang, C.L., Lee, S.N., Chen, M.C., 1988. Seasonal changes in circulating levels of gonadal steroids during an estrous cycle of Holstein-friesian cows. Bull. Inst. Zool. Acad. Sinica(ROC) 27, 133–143.
Yu, J.Y.L., Shen, S.T., Wu, Y.C., 1991. Gonadotropin specific-ity and species diversspecific-ity of gonadal steroid hormone for-mation in fish. Bull. Inst. Zool. Acad. Sinica (ROC)