Sputtered Cr and Reactively Sputtered CrN Serving as
Barrier Layers Against Copper Diffusion
Jui-Chang Chuang,* Shuo-Lun Tu, and Mao-Chieh Chen**
Department of Electronics Engineering and the Institute of Electronics, National Chiao-Tung University, Hsinchu 300, Taiwan
ABSTRACT
The barrier capabilitr of sputter deposited Cr and reactively sputter deposited CrN films against Cu diffusion in a structure of Cu/barrier/p n junction diodes was investigated by means of t1ermal annealing at elevated temperatures in conjunction with electrical measurements and material analysis. For a 500 A thick barrier layer, the barrier capability of a pure Cr layer was limited to temperatures up to 500°C, while CrN films sputter deposited in a gas mixture of Ar and N2 showed improved barrier capabilities. With Ar/N2 flow rates of 24/6 to 24/12 standard cubic centimeters er minute, the deposited CrN films possessed a much improved barrier capability. In particular, the Cu/CrNr (24/9)/p n junction diodes were capable of sustaining 30 mm of thermal anneal at temperatures up to 700°C without degradation of the diodes' electrical characteristics. The failure of Cu/Cr/p*n and Cu/CrN/pn junction diodes under extreme thermal treat-ment was presumed to arise from two mechanisms: grain boundary diffusion for lightly nitrogen doped CrNr and pure Cr barriers, and localized defect (microcrack) diffusion for excessively nitrogen doped CrN, barriers.
lnfroduction
Withthe continuing progress of integrated circuit tech-nology, the requirement to increase device density necessi-tates the improvement of multilevel metallizations."2 As
the linewidth of aluminum (Al) interconnects is scaled
down to deep submicrometer dimensions, the electrical re-sistivities of Al and its alloys are no longer low enough to reduce the interconnect propagation delay.3 The Al inter-connection line also suffers from earlier electromigration
failure under high current density.4-6 Furthermore, hillocks form on Al films at processing temperatures as low as
300°C, and cause interlevel (as well as intralevel) shorting by penetrating through the dielectric layers that separate neighboring interconnect lines.7
Copper (Cu) has been extensively studied as a potential substitute for Al and Al alloys in multilevel metallization
of semiconductor devices and integrated circuits.1'2 Copper
metallization for multilevel interconnects has many advan-tages, such as high melting point,8'9 high electromigration
resistance,'° low electrical resistivity,1'8 and thus low
inter-connect RC time delay, low reaction tendency with com-monly used diffusion barrier materials,"8'° and, probably, overall back-end process simplicity. However, there are a number of difficulties associated with the application of Cu in silicon-based integrated circuits, for example, poor
ad-hesion to dielectric layers,1 difficulty in dry etching,3'1' easy
diffusion in silicon and Si03, deep level trap in silicon, and formation of Cu silicides at low temperatures. All of these drawbacks have detrimental effects on the device reliabili-ty'2-'7 Therefore, the use of barrier layers between Cu and Si as well as Cu and Si02 becomes essential to the success-ful use of Cu in integrated circuit applications.
A large number of barrier layers have been studied.'7 Among them, refractory metals (Cr, Nb, Ti, Mo, W, and Ta)
and their nitrides have been investigated as diffusion
bar-riers because of their high melting points, high thermal
stability, good adhesion to dielectrics, and good electrical
conductivity.'72' Chromium (Cr) and its nitride (CrN)
films are widely used as scratch and corrosion passivation
layers in field application.22'23 Nonetheless, little work has
been reported regarding the properties of Cr-related thin films that serve as diffusion barriers in Cu metallization
systems. In this work, Cu/Cu/n-Si and Cu/CrNjn-Si
structures were investigated with respect to the barrier capability of the sputtered Cr and reactively sputtered
CrN films against Cu diffusion.
*ElectrochemicalSociety Student Member, ** ElectrochemicalSociety Active Member,
Experimental
For this study, Cu/Cr/pn and Cu/CrNr/pn junction
diodes were fabricated from n-type, (100) oriented, 4 in.
diam Si wafers with a nominal resistivity of 1-7 (1 cm.
After a standard RCA cleaning,24 5000 A Si02 was ther-mally grown in a pyrogenic steam atmosphere at 1050°C. Square active regions with areas ranging from 100 x100
to 1000 X 1000 p.m were defined by the conventional pho-tolithographic method. Then a 250 A screen oxide was
thermally grown in dry oxygen atmosphere at 900°C. The
pn junctions were formed by BF2 implantation at an energy of 40 keV to a dose of 3 X i0' cm2 followed by thermal annealing in a N2 atmosphere at 900°C for 30 mm. The screen oxide was then removed using a dilute HF
solu-tion. The junction made by this process was estimated to be 0.25-0.30 p.m. The wafers were loaded into a sputtering
system immediately following the screening oxide
re-moval. A 500 A, Cr or CrN barrier layer was sputter
de-posited, followed by another deposition of 2000 A Cu over-layer without breaking the vacuum. The Cr film was
sputter deposited using a pure Cr target (99.9999% purity) in Ar atmosphere, while the CrNr layers were deposited by reactive sputtering using the same Cr target in a gas mix-ture of Ar and N3 with various flow rates. The base pres-sure of the sputtering chamber was 2 X iO Ton, and films were sputtered at a pressure of 7.6 mTorr. For the purpose of easy identification, the CrN films deposited with vari-ous flow rates of Ar and N3 are designated as CrN (a/b) hereafter, where a and b stand for Ar and N2 flow rates in standard cubic centimeters per minute (sccm), respective-ly, in the sputtering gas mixture. After the deposition of the Cu overlayer, the Cu/Cr as well as the Cu/CrN dual layers were patterned using the lift-off method.
The Cu/barrier/pn samples were thermally annealed in a furnace for 30 mm at temperatures ranging from 400 to 800°C in a N3 atmosphere. At the end of the thermal treat-ment, samples were retained in the furnace with N2 flush-ing until the temperature was below 200°C; then samples were removed from the furnace. Reverse bias leakage cur-rents were measured at a reverse bias of 5 V for the sam-ples with various junction areas. Typically, for each case, eighteen randomly chosen diodes were measured using an HP4145B semiconductor parameter analyzer.
Unpatterned samples of Cr/Si, CrNJSi, Cu/Cr/Si, and Cu/CrN/Si structures processed in the same process run were also prepared for material analysis. Sheet resistance was measured using a four-point probe on the unpatterned samples. The phase composition was identified by X-ray diffraction (XRD) analysis. Scanning electron microscopy 4290 J. Electrochem. Soc., Vol. 145, No. 12, December 1998 The Electrochemical Society, Inc.
(SEM) was used to investigate the surface morphology and
the cross section of the samples. The nitrogen content
(atomic concentration, designated as N atom %) in the as-deposited CrN film was obtained by Auger electron spec-troscopy (AES) analysis.
Resultsand Discussion
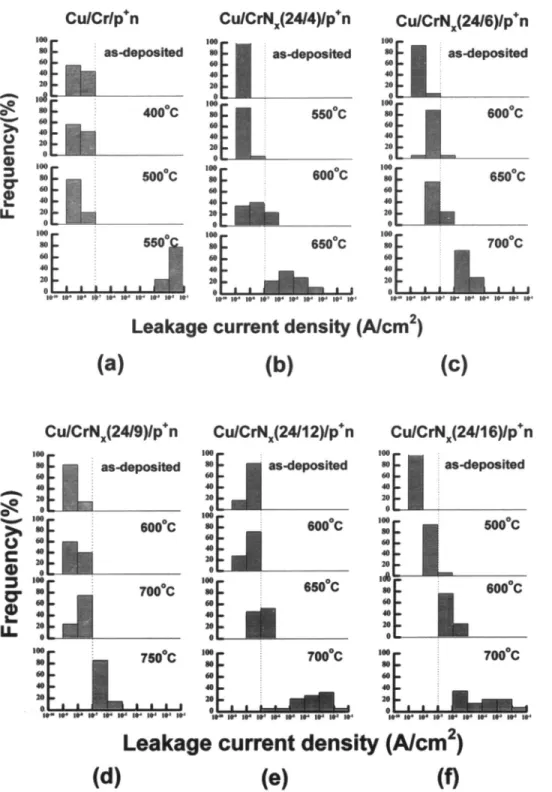
Thermal stability of Cu/barrier/pn junction diodes.— Figure 1 shows the statistical distributions of reverse bias (5 V) leakage current densities for the Cu/Cr (500 A)/pn junction diodes as well as for the Cu/CrNr(500 A)/pn junc-tion diodes annealed at various temperatures. In this study,
we defined a leakage current density larger than 1 X i0
A/cm2 as a failure. The Cu/Cr/pin junction diodes (Fig. la)
were able to sustain a 30 mm thermal treatment up to
500°C without causing degradation; however, annealing at
550°C resulted in severe degradation. For the
Cu/CrN(24/4)/pn junction diodes (Fig. ib), diode degra-dation after annealing at temperatures above 600°C was
observed, showing an at least 50°C improvement of the
thermal stability over the Cu/Cr/pn diodes. Further im-provement was observed for the Cu/CrN,/24/6)/pn and
Cu/CrNr(24/12)/pn junction diodes (Fig. ic and e), which
were able to sustain the thermal treatment up to 600°C. The
best resu1t was observed for the Cu/CrN(24/9)/p*n junc-tion diodes (Fig. id), which remained stable at
tempera-tures up to 700°C. This is an improvement of 200°C as com-pared to the Cu/Cr/pn junction diodes. However, the thermal stability of Cu/CrN(24/16)/pn diodes (Fig. if) showed only slight improvement over that of Cu/Cr/pn
diodes.
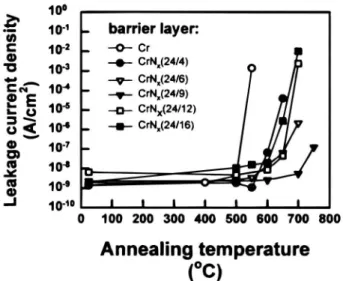
Average values of leakage current density vs. annealing
temperature for all the Cu/CrN/pn junction diodes are
summarized in Fig. 2. The results indicate that the thermal stability of Cu/Cr/pn junction diodes can be substantial-ly improved by using reactivesubstantial-ly sputtered CrN films with appropriate nitrogen contents in a sputtering gas mixture of Ar and N2 instead of a pure Cr barrier layer.
Leakage current density (AIcm2)
(e)
CuICr!pn
Cu!CrN(24I4)!pn
CuICrN(24I6ypn
1I0 ILL
iL LL
600°C':
650°C650°C
:
700°C—
. i
r_L_L._L_IlII__L._L_J
I.- I.' ii' is' is' is' is. is. I,- 10. is is. II- is• I. is is'
I
I
(a)
(b)
(c)
Leakage current density (A/cm2)
Cu!CrN(24I9)Ipn
Cu!CrN(24I1 2)/pn
CuICrN(24I1 6pn
-I.
as-depositedj
asdeposited as-depositedFig. 1. Statistical distributions
of reverse bias (5 V) leakage
current density for the
Cu/barn-er/pn 'unction diodes having
500 A thick barrier layers
annealed at various tempera-hires for 30 mm: (a) Cu/Cr/pn,
(b) Cu/CrN,(24/4)/pn, (c) Cu / Cr N ,( 24 / 6)/ p (d) Cu/CrN.(24/9)/pn, (e) Cu/CrN.(24/12)/pn, and (f) Cu/CrN(24/ 1 6)/pn. IN N N N 20 IN N SI a 20
i.
N N*
20 C ISO N N N 20 ISO 00 60 40 20 IN N N 40 20 Kc
600°C 700°CLL2
':
I
600°C 650°C 700°C (i
I.LL11
LL
IN 700°C NII- iI is. is' is' is• is' is' is' is'
(d)
is- is. is. is. is' is. is. is. is. is.
C
0
-C0
0
-JPhysicalproperties of Cr and CrN films deposited on an Si substrate—Figure 3 shows the nitrogen content (N atom
%) of various as-deposited CrN films. It reveals the
creasing trend of N atom % in the CrNr films with the in-crease of nitrogen volume fractions (N2/Ar + N2) in the
sputtering gas mixture. Nonetheless, N atom % eventually
reached a saturated value of about 40 atorn %.Figure 4 shows the surface morphology of the 2000 A thick as-de-posited Cr and various CrNr films on Si substrates. The Cr
film (Fig. 4a) revealed a slightly granular surface, while the
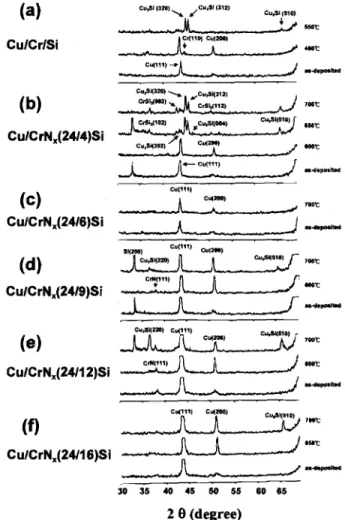
CrN films revealed granular surfaces with various grain sizes and roughness (Fig. 4b-d). Also, microcracks were clearly observable on the surface of the CrN(24/l6) film (Fig. 4d), which was sputtered in a gas mixture with a high content of nitrogen (flow rate 16 sccm). Figure 5 shows XRD spectra for the as-deposited and thermally annealed Cr and various CrN films deposited on Si substrates. For the as-deposited samples (Fig. 5a), the Cr/Si sample
exhib-ited a strong Cr(ll0) preferred orientation as well as a
weak diffraction peak corresponding to the Cr oxide, pre-sumably due to air exposure of the samples before taking
the XRD analysis, because Cr is inherently an oxygen
absorber.8'9 For the CrNJSi samples, the Cr nitride (CrN) phase appeared, and the intensity of the diffraction peak became stronger with increasing nitrogen content in the sputtering gas mixture. The CrNr(24/4)/Si (not shown) and CrN(24/6)/Si samples revealed similar XRD spectra, i.e., neither Cr nor CrN diffraction peaks were observed. The
CrN(24/9)/Si sample revealed a polycrystalline CrN phase ________
Fig. 3. N atom % of as-deposited CrN, films vs. N2 volume frac-tion in the sputtering gas mixture as determined by AES.
barrier layer: —0—Cr —•— CrN,(24/4) —ç-- CrN(24/6) —-- CrN(24/9) —.0.- CrN(24I12) -— CrN,(24/16) 100 10.1 102 iO-10-s
10
10-' 10-i 10-' 10-' 10.10 0 100 200 300 400 500 600 700 800Annealing temperature
(°C)
Fig.2. Average reverse bias leakage current density vs.
anneal-ing temperature for various Cu/barrier/pn junction diodes.
of (111) and (200) orientations. The CrN(24/l2) and
CrNr(24/16) (not shown) samples showed similar spectra, in which only CrN(1 11) X-ray reflections were found. For the thermally annealed samples (Fig. 5b), the XRD spectrashowed basically the same nitride phase detected in the
corresponding as-deposited samples. However, a silicide (CrSi2) phase appeared on the Cr/Si sample annealed at
(a)
(b)
(c)
(d)
z
60 50 40 30 20 10 0 0 10 20 2500A 30 40 50N2 volume fraction (%)
Fig. 4. SEM micrographs showing the surface morphology of
as-deposited Cr and CrN, films: (a) Cr, (b) CrN(24/6), (c) CrN(24/9), and (d) CrN(24/ 16).
J. Electrochem. Soc., Vol. 145, No. 12, December 1998 The Electrochemical Society, Inc.
(a)
0.8 JC,NA2A45IJcflJlllO)
30 35 40 45 50 55 60 65
29 (degree) CiSI4)10) CS,)112) IOOt,c,)sl30 35 40 45 50 55 60 65
20 (degree)Fig. 5. XRD spectra for (a) as-deposited and (b) thermally
annealed Cr/Si and various CrN/Si samples.
temperaturesof 600°C and above. Figure 6 shows the resis-tivity of CrN films vs. nitrogen volume fraction in the sputtering gas mixture, showing an N-shaped variation.
Figure 7showsthe deposition rate of CrN films for vari-ous sputtering powers vs. nitrogen volume fraction in the sputtering gas mixture. As was expected, higher sputtering powers resulted in higher deposition rates. Furthermore, higher volume fractions of nitrogen in the sputtering gas mixture resulted in lower deposition rates.
Thermal annealing of Cu/Cr/Si and Cu/CrNJSi
struc-tures.—XRD spectra of as-deposited and thermally
annealed Cu/Cr/Si and Cu/CrNJSi samples having 500 Athick barrier films are illustrated in Fig. 8. For the
as-deposited samples, the phase composition of the Cr and CrN films remained unchanged as compared with the cor-responding samples without a Cu overlayer. In addition, there were no obvious differences between the spectra of as-deposited and the corresponding low temperature
nealed samples. Figure 9 shows sheet resistance vs. an-nealing temperature for various Cu/CrN/Si samples.
After annealing at a temperature of 400°C, all samples re-vealed a slight decrease of sheet resistance, presumably due to the healing of sputter induced defects and the crys-tallization of the Cr and CrN, films.1"125 For the Cu/Cr/Si sample, sheet resistance revealed a drastic increase after
0 10 20 30 40 50
N2 Volume fraction (%)
Fig. 6. Resistivity of as-deposited CrN0 films vs. N2 volume
frac-tion in the sputtering gas mixture. Criftill) Cttl.(24112))5I
.2
co
0'—'
a
0
a
(b)
0.7 0.6 0.5 0.4 0.3 0.2 0.1 o 260w o 200w A 160w V 100w450w
C.tl(111) 5M20) 4 Citl(200) 0,01)220)bit.
Cfl442V1))S1 -- bOOt. C01424(ipS( 0 10 20 30 40 0.N2
Volume fraction (%)
Fig. 7. Deposition rate of CrN,, films vs. N2 volume fraction in the
sputtering gas mixture for various sputtering powers.
annealing at 500°C, and the XRD analysis showed the
appearance of Cu3Si phase after annealing at tempera-tures as low as 550°C, but showed no appearance of Cr sili-cide phase (Fig. 8a). For the Cu/CrN(24/4)/Si sample,
Cu3Si and CrSi2 were observed after annealing at 650°C
(Fig. 8b) and the sheet resistance also showed a drastic increase (Fig. 9a). For the samples with Cr or CrNr(24/4) as
a barrier layer, the results of the electrical measurements (Fig. 1 and 9) and the XRD analysis(Fig. 8) consistently
show that the formation of high resistive Cu silicide2°
stand for the failure of the diffusion harriet For the
sam-(a)
Co,SI (320) ._, Cu.5) (312)00.51(110) j..., soot Cu/CriSi ..-____::L::/L__._....__0..___..___....1 a
(b)
Co.3l(320) _.Co,50312) TOOt3I2)jflt
Cu/CrN5(2414)Si(c)
Cu/CrN0(24!6)Si(d)
CuICrN5(24!9)Si(e)
C*)11l) -region 3 - region 2I
9
C
S
>0 'is to to a, 8x103 7x103 6x103 SxlO' 4x103 3x10 Cr 24/6 I I I Co)lll) 0.12W) lootflj
________—
C..5I)22O) 00)111)Cu/CrN5(24/12)Si
_nA_J____.) .oOt
AJ _i —
00)111) 0i4000)
o'UJlO)
CuICrN5(24/16)Si _.__0____a ___k_________J
—
30 35 40 45 50 55 60 65
20 (degree)
Fig.8.XRDspectra for the as-deposited as well as the
therm-ally annealed Cu/barrier/Si samples: (a) Cu/Cr/Si, (b) Cu/ CrN,j24/4)/Si, (c) Cu/CrN5(24/6)/Si, (d) Cu/CrNJ24/9)/Si, (e)
I 0
w
C10
E Io
—0
a) .C 1OFig. 9. Sheet resistance vs. annealing temperature for various Cu/barrier/Si samples.
pieswith a barrier layer of CrN films deposited with a
higher nitrogen content in the sputtering gas mixture
(Ar/N2 flow ratio less than 24/6), Cu suicide was also de-tected by XRD analysis at and above the temperatures of the observation of electrical failure (Fig. 1, 8, and 9d-f); however, no Cr silicide phase was observed. The samples with CrN(24/9), CrN(24/12), and CrN(24/16) barrier
lay-ers showed only a moderate increase of the sheet resist-ance after annealing at temperatures above 600°C; this in-dicated that the interaction between Cu and Si substrates was not sensitively detectable by sheet resistance
meas-urement and XRD analysis, but was serious enough to
detrimentally increase the junction leakage current.
Fig-ure 10 shows SEM micrographs for the Cu/Cr/Si and Cu/CrNJSi samples annealed at elevated temperatures;
formation of Cu silicide can be clearly observed. After an-nealing at high temperatures, tetragonal92628 Cu3Si was observed. Deformed and ruptured films due to the stress induced by the volume change of suicide formation were easily observed for the Cu/Cr/Si sample annealed at 600°C
(Fig. lOa and b) and the Cu/CrN(2 4/4)/Si sample annealed
at 650°C (Fig. bc); they were also observed for the
Cu/CrN(24/6)/Si sample annealed at 800°C (Fig. lOd) and the Cu/CrN(24/9)/Si sample annealed at 750°C (Fig. be),though they were found in fewer numbers. For the
Cu/CrN(24//16)/Si sample that revealed microcracks in
the as-deposited CrN film (Fig. 4d), thermal annealing
resulted in porosity of the Cu overlayer (Fig. lOf),
presum-ably resulting from the fast inward diffusion of Cu
through the microcracks.2°Discussion.—In Fig. 5, it was observed that the sputtered
Cr film revealed a Cr(110) preferred orientation, which
remained stable after annealing at temperatures up to
500°C. For the reactively sputtered CrNr films, diffraction peaks of CrN phase appeared and the intensity of the peaks increased with the nitrogen content in the sputtering gas
Fig. 10. SEM micrographs
showing the surface morpholo-gy and cross-sectional view for the thermally annealed
Cu/bar-rier/Si samples: (a) and (b) Cu/Cr/Si at 600°C, (c) Cu/CrN(24/4)/Si at 650°C, (d) Cu/CrN(24/6)/Si at 800°C, (e) Cu/CrN(24/9)/Si at 750°C, and (0 Cu/CrN(24/16)/Si at 650°C. 0 100 200 300 400 500 600 700 800
Annealing temperature (°C)
mixture. In addition, the CrN phase with the (111)
pre-ferred orientation was detected for the samples of
CrN(24/12) and CrN(24/l6). Because all the CrN1 films were reactively sputtered using the same conditions except the Ar/N2 flow rates, we assume that the nitrogen content in the sputtering gas mixture should be responsible for the orientation preference.
For the CrN films deposited with low nitrogen flow
rates, the incorporated nitrogen did not react with the sput-tered Cr; instead, the nitrogen tended to collect in the grain boundaries of the deposited Cr films. As a consequence of lattice deformation, the resistivity of the Cr films increased with the increase of nitrogen content. This is designated as
region 1 for the N-shaped curve shown in Fig. 6. Eventually,
the crystalline structure of the Cr film was destroyed and
the XRDdatashowed a nearly amorphous spectrum, which
revealed no distinguishable diffraction peak after annealing at temperatures up to 600°C. As the nitrogen content in the
sputtering gas mixture was increased, the chemical reaction
of Cr and nitrogen increased, resulting in the formation of
Cr nitride, and thus decreasing resistivity (region 2 of
Fig. 6). The XRD data were similar to that shown in Fig. 5 for the sample of CrN(24/6)/Si, which revealed very weak
but still distinguishable diffraction peaks of CrN after
annealing at 600°C. As the nitrogen content in the
sputter-ing gas mixture was further increased, a portion of the
incorporated nitrogen turned to decorate grain boundaries of the polycrystalline Cr nitride in addition to increase the
N atom % in the deposited films; thus, resistivity of the film
increased again. However, we assume that there is a com-peting sputter deposition/etching process in the sputtering chamber. The nitrogen in the reactive sputtering process served as a reactant for the formation of Cr nitride and also
supported the N2 plasma, which would sputter etch the deposited Cr nitride film. When the nitrogen flow rate in the
sputtering gas mixture was increased to a notable amount, the rates of sputter etching and sputter deposition became
comparable.25'3° Thus, we observed the ceasing of the increasing trend of N atom % (Fig. 3), the decrease of depo-sition rates irrespective of the depodepo-sition power (Fig. 7), and
the increase of surface damages of the films. Eventually, as
the nitrogen flow rate was increased to an excessive amount (e.g., an N2 flow rate of 16 sccm), microcracks (Fig. 4d) were
created in the films that resulted in a drastic increase of
re-sistivity, as shown in region 3 of Fig. 6.
Thermodynamic data31 suggest that Cr nitrides are more stable than Cr silicides at room temperature but silicides
are more stable at elevated temperatures., The results
shown in Fig. Sb indicate that interaction between Cr and
the nitrogen in N2 atmosphere as well as between Cr and the
silicon in Si substrate occurred at 600°C of the Cr/Si sam-ples, while there was no interaction between CrN and the
Si substrate at temperatures up to 800°C for the CrN/Si
samples. For the reactively sputter deposited CrN films on Si substrate, Cr nitride was formed during the sputter
depo-sition process, and the formation of Cr silicide was deterred
by the presence of Cr nitride.
There are two possible types of Cu diffusion paths in the
Cr-based barrier layers. First, for the Cr and CrN films
deposited with low nitrogen content in the sputtering gas
mixture (N2 flow rates below 6 sccm), Cu diffuses to the Si
substrate through the barrier layers along the grain
bound-aries which are deficient in nitrogen decoration. Second, for
the CrN films deposited with high nitrogen content in the
sputtering gas mixture (N2 flow rates above 12 sccm),
local-ized defects, such as microcracks, provide fast diffusion paths for Cu diffusion. Cu can diffuse to the Si substrate by way of these diffusion paths and deteriorate the electrical
characteristics of the pn junction diodes,1247 and form localized Cu3Si grains as shown in Fig. 10. As the grains of
Cu3Si grew, the volume difference between Cu and its
sili-cide resulted in the deformation of the Cu overlayer and the
protrusion of Cu2Si through the Cu overlayer as well as the intrusion of Cu3Si grains into the underlying Si substrate.
Presumably, the roles of the nitrogen are to collect at the grain boundaries of the as-deposited barrier layers and to
form Cr nitrides. For the CrN,, films deposited with low nitrogen content in the sputtering gas mixture, there was not enough nitrogen incorporation and a Cr nitride phase was not detected. For the CrN films deposited with high nitrogen content in the sputtering gas mixture, the nitrogen
content in the CrN films was increased and CrN was
detected. The abundant supply of nitrogen not oniy formed Cr nitride but also blocked the grain boundaries of the Cr nitride; thus, it should possess a superior barrier capability.
However, CrN films deposited with very high nitrogen con-tent in the sputtering gas mixture (e.g., N2 flow rate of
16 sccm) possessed sputter-induced defects such as
miro-cracks. Therefore, the defect-related failure took dominance
and the electrical characteristics of the pn junction diodes tended to deteriorate with increasing annealing tempera-ture. For the CrN films deposited with medium nitrogen content in the sputtering gas mixture (N2 flow rates from 6 to 12 sccm), failure would occur due to both mechanisms.
Moreover, the barrier capability of CrN films deposited
with nitrogen flow rate of 9 sccm (i.e., the CrN(24/9) film) was the best among all the Cr nitride films investigated.
Conclusion
Thiswork investigated the barrier capability of sputter deposited Cr and reactively sputter deposited Cr14,, films against Cu diffusion in a structure of Cu/barrier/pn junc-tion diodes. The CrN,, films deposited in a sputtering gas mixture of Ar and N2 with Ar/N2 flow ratios from 24/6 to
24/12 were found to possess efficient barrier capability. For
a 500 A thick layei we found that the Cu/CrN,,(24/9)/pn junction diodes were capable of sustaining 30 mm of ther-mal annealing at temperatures up to 700°C without degra-dation to the diodes' electrical characteristics. The dorre-sponding thermal stability of junction diodes using a pure
Cr barrier layer was found to be 500°C. The failure of
Cu/CrN,,/pn junction diodes under extreme thermal treat-ment was presumed to be arising from two mechanisms:
grain boundary diffusion for the lightly nitrogen doped CrN,, including the Cr barriers, and localized defect
(micro-crack) diffusion for the excessively nitrogen doped CrN,, barriers.
Acknowledgment
The authors wish to thank the Semiconductor Research Center of National Chiao-Thng University for providing excellent processing environment. This work was support-ed by the National Science Council, ROC, under contract
no. NSC-86-2215-E-009-040.
Manuscript submitted March 10, 1998; revised manu-script received August 10, 1998.
National Chiao-Tung University assisted in meeting the publication costs of this article.
REFERENCES
1. See, for example, MRSBull.,18 (1993); 19 (1994);
de-voted to the topic of copper metallization for
micro-electronics.
2. K. P Rodell, E. G. Colgan, and C. K. Hu, in Mater Res.
Soc. Symp. Proc., 337, P Murarka, A. Katz, K. N. Th,
and K. Maex, Editors, p. 59, MRS, Pittsburgh, PA
(1994).
3. C. K. Hu, B. Luther, F B. Kaufman, J. Hummel, C.
Uzoh, and D. J. Pearson, ThinSolidFilms, 262, 84
(1995).
4. K. N. Th, J.Vac. Sci. Technol., A,2, 216 (1984).
S. J. K. Howard, J. F White, and P 5. Ho, J. Appl. Phys.,
49, 4083 (1978).
6. C. K. Hu, Thin Solid Films, 260, 124 (1995).
7. S. Wolf and R. N. Tauber, Silicon Processing for the VLSIEra,Vol. 1, Editor; p. 269, Lattice Press, Sunset
Beach, CA (1986).
8. CRC Handbook of Chemistry and Physics, 73rd ed.,
D. R. Lide, Editor, Section 12, CRC Press, Inc., Boca Raton, FL (1992).
9. Binary Alloy PhaseDiagrams,2nd ed., P B. Massalski,
Editor; pp. 1266, 1293, 1304, 1333, 1446, ASM
Inter-national, Materials Park, OH (1990).
10. 5. Shingubara, K. Fujiki, A. Sano, H. Sakaue, and Y.
J. C. Coburn, J. E. Sanchez, Jr., K. P. Rodbell, and
W. F Filter, Editors, p. 441, MRS, Pittsburgh, PA (1994).
11. Y. Igarashi, T. Yamanobe, and T. Ito, Thin Solid Films, 262, 124 (1995).
12. J. D. McBrayer, B. M. Swanson, and T. W Sigmon, J. Electrochem. Soc., 133,1242(1986).
13. Y. Shacham-Diamand, A. Dedhia, D. Hoffstettei and
W. G. Oldham, J. Elect rochem. Soc., 140, 2427 (1993).
14. D. Gupta in Mater Res. Soc. Symp. Proc., S. P. Murar-ka, A. Katz, K. N. Tu, and K. Maex, Editors, p. 209,
MRS, Pittsburgh, PA (1994).
15. G. Raghavan, C. Chiang, P. B. Anders, S. M. Tzeng, R. Villasol, G. Bai, M. Bohr, and D. B. Fraser, Thin Solid Films, 262, 168 (1995).
16. VLSI Technology, 2nd ed., S. M. Sze, Editor, p. 309,
McGraw-Hill, Singapore (1988).
17. 5. Q. Wang, S. Suther, C. Hoeflich, and B. J. Burrow, J. Appi. Phys., 73, 2301 (1993).
18. C. A. Chang, J. Appl. Phys., 67, 6184 (1990).
19. D. Y. Shih, C. A. Chang, J. Paraszczak, S. Nunes, and J. Cataldo, J. Appl. Phys., 70, 3052 (1991).
20. H. Ono, T. Nakano, and T. Ohta, Appl. Phys. Lett., 64,
1511 (1994).
21. L. C. Lane, T. C. Nason, G. R. Yang, T. M. Lu, and H.
Bakhru, J. Appl. Phys., 69, 6719 (1991).
22. F. Cosset, G. Contoux, A. Celerier, and J. Machet, Surf. Coat. Technol., 79, 25 (1996).
23. C. Friedrich, G. Berg, E. Broszeit, and K. H. Kloos,
Surf. Coat. Technol., 74-75, 279 (1995).
24. W. Kern and D. A. Puotinen, RCA Rev., 31,187(1970).
25. J. C. Chuang and M. C. Chen, Thin Solid Films, 322,
213 (1998).
26. C. S. Liu and L. J. Chen, J. Appl. Phys., 74, 3611 (1993).
27. C. S. Liu and L. J. Chen, Thin Solid Films, 262, 187
(1995).
28. C. S. Liu and L. J. Chen, J. Appl. Phys., 75, 2370 (1994).
29. B. E. Reed-Hill, Physical Metallurgy Principles, 2nd
ed., p. 386, Van Nostrand, New York (1972).
30. J. C. Chuang and M. C. Chen, J. Electrochem. Soc., 145, 3170(1998).
31. E. A. Brandes, in Smithells Metals Reference Book, 6th
ed., p. 8, Robert Hartnoll Ltd., Bodmin, Cornwall,
England (1983).
Application of a Semi-insulating Amorphous Hydrogenated
Silicon Nifride Film as a Resistive Field Shield and Its
Reliability
K. Matsuzaki,*,0 1. Horasawa,b G. Tada,c and M. Saga"
°Fuji Electric Corporate Research and Development, Limited, Advanced Device Technology Laboratory, Matsumoto City, Japan 390
bFujj Electric Company, Limited, Matsumoto Factory, IC Fabrication Department, Matsumoto City, Japan 390 °Fuji Electric Company, Limited, Matsumoto Factory, Semiconductor Device Research and Development Center,
Matsumoto City, Japan 390
dFuji Electric Corporate Research and Development, Limited, Yokosuka City, Japan 240
ABSTRACT
In order to develop reliable high voltage integrated circuits (HVICs), the characteristics of semi-insulating plasma-deposited amorphous silicon nitride (a-SiN:H) films as resistive field shields, are examined, the reliability of their appli-cation to HVICs are studied. The surfaces of these semi-insulating films were unstable, and it was concluded that these films must be covered with a final passivation film such as an insulating plasma-deposited a-SiN:H film. The transverse electrical conduction mechanism of these films is briefly discussed.
Infroduction
Ingeneral, high voltage integrated circuits (HVICs) using planar technology employ shallow junctions and alternative means to control the electric field near the surface of a
semiconductor. The depletion layer can be controlled and the breakdown voltage can be improved by using a field plate, a resistive field shield, an equipotential ring, etc."2 The use of semi-insulating polycrystalline silicon (SIPOS) as a resistive field shield has been developed,3" but its use has been limited.6-8 Because a resistive field shield was needed and the problems encountered with SIPOS must be
avoided, Osenbach et al. developed Si-rich
plasma-deposit-ed amorphous silicon nitride hydrogen alloys (a-SiN:H) as
resistive field shields.7 In this paper, we study the reliability of HVICs using such semi-insulating a-SiN:H films.
Experimental
Chemical vapor deposition of a-SiN:H films—A
VDS-5000 plasma chemical vapor deposition (CVD) system
(JPEL Co.) was used for the deposition of a-SiN:H films. This system was operated at 50 kHz, 1 kW radio frequen-cy (rf) power, and 320°C. The pressure of deposition and the flow of Ar carrier gas were kept at 53 Pa and 1.5
stan-dard liters per minute (slm), respectively. The gas flow
*ElectrochemicalSociety Active Member,
ratio R = SiH4/(SiH4+ NH3)was variable. Insulating and
semi-insulating a-SiN:H films were deposited at R =0.15
and R = 0.70—0.77,respectively. The substrates were 4 in.
diam, (100)-polished silicon wafers.
HVIC device structure using semi-insulating a-SiN:H films.—The schematic structure of the device is shown in
Fig. 1. The substrates were 4 in. diam, (100)-polished, p-type (p =100—150tI cm) silicon wafers. N-well (p '—10
l
cm, X '—6p.m), P-base (p "-0.511 cm, X —2p.m), P-offset(p —10l cm, X -'-1.5p.m), gate oxide (t0 =25nm),
poly-Si gate electrodes, and source-drain regions were
succes-sively formed in the substrates. The field region lying
between source and drain region was covered with
local-ized oxidation of silicon (LOCOS) (t0,, =0.7p.m),
phospho-silicate glass (PSG) SiO, (tpsG =1p.m), and finally a semi-insulating a-SiN:H film (ts,N =1p.m, R =0.76typically) as
a resistive field shield. The length of the field region was
"70 p.m. The breakdown voltage of this HVIC was
designed to be over 700 V.
Measurements.—The thickness of the film was deter-mined by a laser-based ellipsometer. The density of the film was calculated using the mass determined by measur-ing the sample weight before and after deposition, and the volume of the film. The transmission infrared spectrum of the film was taken with Fourier transform infrared (FTIR)