Editorial Manager(tm) for Rheumatology International (Clinical and Experimental Investigations)
Manuscript Draft
Manuscript Number:
Title: Association of the C-285T and A5954G Polymorphisms in the DNA repair gene OGG1 with the Susceptibility of Rheumatoid Arthritis.
Article Type: Original Article
Keywords: Rheumatoid Arthritis (RA) ‧ 8-oxoguanine glycosylase 1 (OGG1) ‧ Single nucleotide polymorphisms (SNPs) ‧ Haplotypes.
Corresponding Author: Dr. Shih-Yin Chen,
Corresponding Author's Institution: Graduate Institute of Chinese Medical Science, First Author: Shih-Yin Chen
Order of Authors: Shih-Yin Chen; Lei Wan; Chung-Ming Huang; Yu-Chuen Huang; Jim Jinn-Chyuan Sheu; Ying-Ju Lin; Shih-Ping Liu; Yu-Ching Lan; Chih-Ho Lai; Cheng-Wen Lin; Chang-Hai Tsai; Fuu-Jen Tsai Abstract: Rheumatoid arthritis (RA) is a chronic autoimmune disease and can lead to deformities and severe disabilities, due to irreversible damage of tendons, joints, and bones. Previous study indicated that DNA repair system was involved in the pathology of RA. In this study, we investigated the
association of two 8-oxoguanine glycosylase 1 (OGG1) gene polymorphisms (rs159153 and rs3219008) with the susceptibility to RA in 384 Taiwanese individuals (192 RA patients and 192 controls). Our data showed that statistically significant difference in genotype frequency distributions was found at rs3219008 SNP between RA patients and control groups (p = 5.6E-0.5). Our data also indicated that individuals with the AG genotype at rs3219008 SNP may has a higher risk of developing RA. We did not observe any statistically significant association of OGG1 haplotype frequencies (rs159153 and
rs3219008) with RA progression. The study suggested that OGG1 polymorphisms (rs159153 and rs3219008) are associated with RA progression and that these may be used as molecular markers of RA.
Association of the C-285T and A5954G Polymorphisms in
the DNA repair gene OGG1 with the Susceptibility of
Rheumatoid Arthritis.
Shih-Yin Chen ‧ Lei Wan ‧ Chung-Ming Huang ‧ Yu-Chuen Huang ‧ Jim
Jinn-Chyuan Sheu ‧ Ying-Ju Lin ‧ Shih-Ping Liu ‧ Yu-Ching Lan ‧ Chih-Ho
Lai ‧ Cheng-Wen Lin ‧ Chang-Hai Tsai ‧ Fuu-Jen Tsai
F. J. Tsai () ‧ S. Y. Chen ‧ L. Wan ‧ Y. C. Huang ‧ J. J. C. Sheu ‧ Y. J. Lin ‧
C. H. Tsai
Genetic Center, Department of Medical Research, China Medical University Hospital,
Graduate Institute of Chinese Medical Science, College of Chinese Medicine, China
Medical University, Taichung, Taiwan.
e-mail: [email protected]
C. M. Huang
Division of Immunology and Rheumatology, China Medical University Hospital,
Taichung, Taiwan.
S. P. Liu
Manuscript
Center for Neuropsychiatry, China Medical University and Hospital, Taichung,
Taiwan.
Y. C. Lan
Department of Health Risk Management, China Medical University, Taichung,
Taiwan.
Chih-Ho Lai
Department of Microbiology, School of Medicine, China Medical University,
Taichung, Taiwan.
Cheng-Wen Lin
Department of Medical Laboratory Science and Biotechnology, China Medical
University, Taichung, Taiwan.
S. Y. Chen ‧ L. Wan ‧ C. M. Huang, are joint first authors and equal contribution in
ABSTRACT
Rheumatoid arthritis (RA) is a chronic autoimmune disease and can lead to
deformities and severe disabilities, due to irreversible damage of tendons, joints, and
bones. Previous study indicated that DNA repair system was involved in the
pathology of RA. In this study, we investigated the association of two 8-oxoguanine
glycosylase 1 (OGG1) gene polymorphisms (rs159153 and rs3219008) with the
susceptibility to RA in 384 Taiwanese individuals (192 RA patients and 192 controls).
Our data showed that statistically significant difference in genotype frequency
distributions was found at rs3219008 SNP between RA patients and control groups (p
= 5.6E-0.5). Our data also indicated that individuals with the AG genotype at
rs3219008 SNP may has a higher risk of developing RA. We did not observe any
statistically significant association of OGG1 haplotype frequencies (rs159153 and
rs3219008) with RA progression. The study suggested that OGG1 polymorphisms
(rs159153 and rs3219008) are associated with RA progression and that these may be
used as molecular markers of RA.
KEY WORDS:
Rheumatoid Arthritis (RA) ‧ 8-oxoguanine glycosylase 1 (OGG1) ‧ Single
Introduction
Rheumatoid arthritis (RA) is a joint inflammation disease combining several
peripheral inflammatory conditions [1]. It could involve chronic synovitis, inducing
the demolition of joint tissue, especially containing bone and cartilage, and therefore,
joint function is seriously impaired. The prevalence is about 1% with some variation
among ethnic groups [1-3]. Its genetic contribution was well documented by multiple
family studies, and multiple whole-genome sib-pair linkage studies have been
reported with limited consistency among them [5, 6]. The previous studies on
RA-susceptible genes were published from a group based on a high-throughput single
nucleotide polymorphism (SNP) genotyping facility that adopts case-control linkage
disequilibrium (LD) mapping on a large scale as an initial survey method without
using subjects that were used for preceding linkage studies [7-9]. One of them
identified functionally relevant polymorphisms of peptidylarginine deiminase 4, an
enzyme that catalyzes the post-translational citrullination of proteins, as a RA gene
[10, 11]. Besides, the major histocompatibility (MHC) class II region is an important
susceptibility factor, and the human leukocyte antigen (HLA)–DR4 has been
DNA repair system was involved in the pathology of RA [14]. It had been implicated
that T cells of RA patients failed to producesufficient transcripts and protein of the
DNA repair kinaseataxia telangiectasia (AT) mutated (ATM). Therefore, we decided
to study the effect of genetic polymorphism of DNA repair genes as modifiers of the
risk for RA as well in this study.
Moreover, DNA repair enzymes modulate free-radical production after DNA
damage. Among such enzymes, 8-oxoguanine glycosylase 1 (OGG1) seems to be
most important since OGG1 is primarily responsible for removing 8-oxoguanine in
DNA, which is a major product of DNA damage formed by free radicals, and can
mispair with adenine residues instead of the usual cytosine residues, leading to an
increased frequency of G:C to T:A transversion mutations [15, 16].
In the present study, we aimed to identify genetic polymorphisms in potential
candidate genes for RA, and we therefore investigated the association of OGG1 gene
polymorphisms with RA in a Taiwanese population. Our findings are expected to help
us understand the role of OGG1 gene polymorphisms in RA disease and its
progression; this knowledge can point us toward possible management strategies for
this common nephropathy.
Study Population
The study subjects including a total of 192 patients with RA and 192 healthy subjects
were recruited from China Medical University Hospital in Taiwan. Patients with RA
according to the revised America College of Rheumatology criteria[17] were enrolled.
Nephelometry was used to detect rheumatoid factor (RF). Values ≧ 30 IU/ml were
defined as positive. The presence or history of extra-articular manifestations in
patients with RA was recorded [18]. The healthy control from the general population
was selected from health examination. All individuals’ samples were collected by
venipuncture for genomic DNA isolation. Informed consent was from all participants
and was approved by the local Ethics Committee.
Genomic DNA Extraction and Genotyping
Genomic DNA was prepared from peripheral blood according to standard protocols of
the DNA extraction kit (Qiagen, Valencia, CA, USA). The two OGG1 polymorphisms
(rs159153 and rs3219008) were detected by restriction enzyme (RE) digestion. PCRs
for OGG1 gene polymorphisms were carried out in a 50-μL reaction mixture
containing 50 ng of genomic DNA, 2 to 6 pmole of each primers, 1× Taq polymerase
buffer (1.5 mM MgCl2), and 0.25 U of AmpliTaq DNA polymerase (Applied
Biosystems). The primers, PCR conditions, and RE cutting sites used to determine
Statistical Analysis
Chi-squaretest or Fisher’s exact tests will be used to determine statistically significant
differences in allele/genotype frequencies between case and control groups. Allelic
frequencies will be expressed as percentage of the total number of alleles. The
Hardy–Weinberg equilibrium will be tested for each marker using χ2-test. The
haplotype combination at rs159153 and rs3219008 in OGG1 gene was estimated
using Haploview version4.1 [19]. The differences in the distribution ofthe haplotype
frequencies between the two groupswere assessed with a χ2-test. Odds ratios [ORs]
and 95% confidence intervals (95% CIs) were obtained using logistic regressions to
determine associations between OGG1 alleles/genotypes/haplotypes and RA
susceptibility. The Kaplan-Meier method was used to estimate cumulative survival.
Differences in survival were analyzed with the log-rank test. All data were analyzed
with SPSS Version 15.0 software (SPSS Inc., Chicago, IL, USA). A p value < 0.05
was considered statistically significant.
Results
The genotypic and allelic frequencies of rs159153 and rs3219008 are shown in Table
allele to be the major one at the rs159153 polymorphism both in RA patients (90.6%;
348/384) and controls (89.6%; 344/384). The A allele was the major one at the
rs3219008 polymorphism in RA patients (55.7%; 214/384) and controls (51.8%;
199/384). When we compared the genotype distribution between RA patients and
control groups, statistically significant differences in genotype frequency distributions
were noted for the rs3219008 SNP in RA patients and controls (p = 5.6E-05). Our
data indicated that individuals with the AG genotype at rs3219008 SNP may have a
higher risk of developing RA (Table 2).
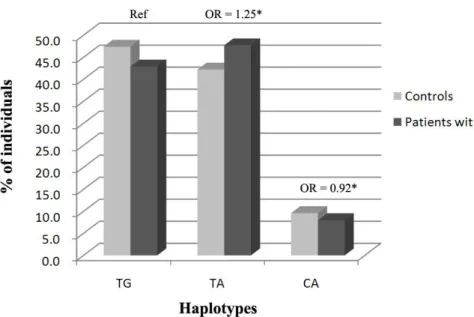
Haplotype frequencies were estimated using the rs159153 and rs3219008 SNPs.
Three haplotypes of the OGG1 were present in the study population. The TG and TA
were the common haplotypes both in RA patients (42.9% and 47.7%, respectively)
and health control (47.4% and 42.2%, respectively) groups. Subsequently, we
constructed haplotypes and considered haplotype with wild-type alleles of the two
polymorphisms as reference (TG) and analyzed the risk of RA in individuals with
other haplotypes. The trend of frequency distribution of three haplotypes (TG, TA and
CA) obtained in control individuals. Regression analysis revealed that there was no
association with combined effect of these two polymorphisms with RA risk (Fig. 1).
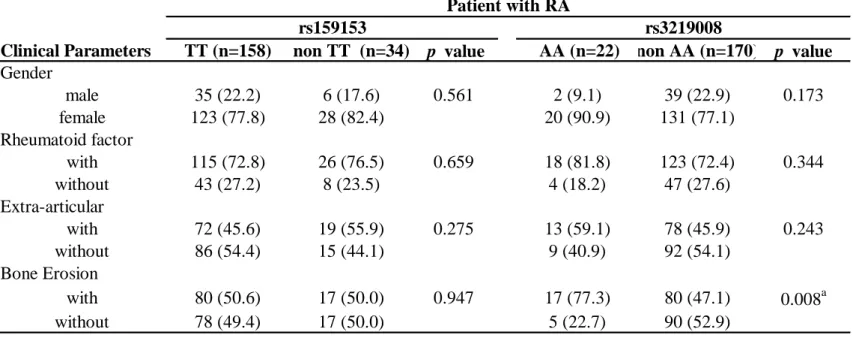
Comparisons of the clinical features of RA patients with the different
rheumatoid factor accompaniment, and incidence of extra-articular. We observed the
percentage of bone erosion occurrence in RA patients with the AA genotype at
rs3219008 SNP was much higher than RA patients with non-AA genotype (p = 0.008).
Briefly, our data indicated that individuals with the AA genotype at rs3219008 SNP
may have a higher risk of developing RA (Table 3).
Discussion
Currently, RA is considered to be a chronic inflammatory disease with
tissue-destructive potential that occurs in genetically susceptible individuals.
Polymorphic gene sequences of cytokines known to be involved in the pathogenesis
of RA are potential markers of disease susceptibility. Previous studies have examined
the relationship between cytokine gene polymorphisms and the incidence of RA,
including urokinase, IL-6, IL-8, CD4 cells [20-22]. Moreover, One effect involves a “snow-balling” mechanism of increased levels of cellular damage and death leading
to more inflammation, which in turn produces more ROS [23]. The question remains
whether the defect in DNA damage repairfunctions to render individuals susceptible
to RA or is a consequenceof disease. Accumulation of DNA damage may also have
In this study, we focused on the variants of the OGG1 gene that had previously
been investigated for breast cancer, bladder cancer, and Alzheimer's disease [24-26].
We found a statistically significant association between RA and the rs3219008
polymorphism. The AG genotype frequency at rs3219008 was significantly higher in
RA than in the control participants (Table 1). Our results also indicated that the T-A
haplotype of the OGG1 gene was estimated to be present in approximately 47.7% of
RA patients. We observed that the T-A haplotype seems appeared to be an “at-risk”
haplotype for RA progression, although the difference was not statistically significant
(Fig. 1).
The treatment strategies for RA patients are also an important issue. We
observed the percentage of bone erosion occurrence in RA patients with the AA
genotype at rs3219008 SNP was much higher than RA patients with non-AA genotype
(p = 0.008). Despite the similar mode of treatment given to our patients, greater
rheumatoid factor accompaniment and incidence of extra-articular were observed in
the AA subgroup than in the subgroups with non-AA genotype at rs3219008, although
the difference was not statistically significant (Table 3). These data suggest that a
dose readjustment in the drugs given may be required according to the different
genotype. In addition, more specific drugs that interact with OGG1 could be given in
at rs3219008.
In conclusion, our findings strongly suggest an association between OGG1
(rs3219008) genetic variants and RA disease susceptibility; further, we indicated that
these polymorphisms contribute to the genetic background of RA pathogenesis.
Moreover, the absence of the AA genotype at rs3219008 is associated with the bone
erosion occurrence in RA patients. The findings should prompt specific considerations
for the treatment of RA patients with the AA genotype at rs3219008.
Acknowledgements
We thank all colleagues at the Genetic Center, Department of Medical Research,
China Medical University Hospital for their feedback and technical support. This
work was supported by China Medical University (CMU98-N1-18), China Medical
University Hospital (DMR-98-042 and DMR-98-144) and National Science Council
in Taiwan (NSC99-2314-B-039-005-MY3).
References
therapy. N Engl J Med 322:1277–1289
2. Ziff M (1990) Rheumatoid arthritis--its present and future. J Rheumatol 17:
127-133
3. Feldmann M, Brennan FM, Maini RN (1996) Rheumatoid arthritis. Cell 85:
307-310
4. Yamada R, Ymamoto K (2005) Recent findings on genes associated with
inflammatory disease. Mutat Res 573:136–151
5. Weyand CM, Klimiuk PA, Goronzy JJ (1998) Heterogeneity of rheumatoid
arthritis: from phenotypes to genotypes. Springer Semin Immunopathol 20: 5-22
6. Lynn AH, Kwoh CK, Venglish CM, Aston CE, Chakravarti A (1995) Genetic
epidemiology of rheumatoid arthritis. Am J Hum Genet 57: 150-159
7. Ohnishi Y, Tanaka T, Ozaki K, Yamada R, Suzuki H, Nakamura Y (2001) A
high-throughput SNP typing system for genome-wide association studies. J Hum
Genet 46:471–477
8. Ozaki K, Ohnishi Y, Iida A, Sekine A, Yamada R, Tsunoda T, et al (2002)
Functional SNPs in the lymphotoxin-alpha gene that are associated with
susceptibility to myocardial infarction. Nat Genet 32: 650–654
9. Suzuki A, Yamada R, Chang X, Tokuhiro S, Sawada T, Suzuki M, et al (2003)
deiminase 4, are associated with rheumatoid arthritis. Nat Genet 34: 395–402
10. Speckman RA, Wright Daw JA, Helms C, Duan S, Cao L, Taillon-Miller P, et al
(2003) Novel immunoglobulin superfamily gene cluster, mapping to a region of
human chromosome 17q25, linked to psoriasis susceptibility. Hum Genet 112:
34–41
11. Worthington J, John S (2003) Association of PADI4 and rheumatoid arthritis: a
successful multidisciplinary approach. Trends Mol Med 9: 405–407
12. van Zeben D, Hazes JM, Zwinderman AH, Cats A, Schreuder GM, D'Amaro J,
Breedveld FC (1991) Association of HLA-DR4 with a more progressive disease
course in patients with rheumatoid arthritis. Results of a follow up study. Arthritis
Rheum 34: 822–830
13. Wagner U, Kaltenhauser S, Sauer H, Arnold S, Seidel W, Häntzschel H, et al
(1997) HLA markers and prediction of clinical course and outcome in rheumatoid
arthritis. Arthritis Rheum 40: 341–351
14. Shao L, Fujii H, Colmegna I, Oishi H, Goronzy JJ, Weyand CM (2009)
Deficiency of the DNA repair enzyme ATM in rheumatoid arthritis. J Exp Med
206: 1435-1449
15. Mabley JG, Pacher P, Deb A, Wallace R, Elder RH, Szabo C (2005) Potential role
290–292
16. Cooke MS, Evans MD, Dizdaroglu M, Lunec J (2003) Oxidative DNA damage:
mechanisms, mutation, and disease. FASEB J 17: 1195–1214
17. Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS, et al
(1988) The American Rheumatism Association 1987 revised criteria for the
classification of rheumatoid arthritis. Arthritis Rheum 31: 315-324
18. Yen JH, Chen JR, Tsai WJ, Tsai JJ, Liu HW (1995) HLA-DRB1 genotyping in
patients with rheumatoid arthritis in Taiwan. J Rheumatol 22: 1450-1454
19. Barrett JC, Fry B, Maller J, Daly MJ (2005) Haploview: analysis and visualization
of LD and haplotype maps. Bioinformatics 21: 263–265
20. Huang CM, Chen CL, Tsai JJ, Tsai CH, Tsai FJ (2004) Association between
urokinase gene 3'-UTR T/C polymorphism and Chinese patients with rheumatoid
arthritis in Taiwan. Clin Exp Rheumatol 22: 219-222
21. Lo SF, Huang CM, Lin HC, Chen WC, Tsai CH, Tsai FJ (2008) Cytokine (IL-6)
and chemokine (IL-8) gene polymorphisms among rheumatoid arthritis patients in
Taiwan. Clin Exp Rheumatol 26: 632-637
22. Lo SF, Wan L, Lin HC, Huang CM, Tsai FJ (2008) Association of CD4 enhancer
gene polymorphisms with rheumatoid arthritis and systemic lupus erythematosus
23. Meira LB, Bugni JM, Green SL, Lee CW, Pang B, Borenshtein D, et al (2008)
DNA damage induced by chronic inflammation contributes to colon
carcinogenesis in mice. J Clin Invest 118: 2516-2525
24. Rossner P Jr, Terry MB, Gammon MD, Zhang FF, Teitelbaum SL, Eng SM, et al
(2006) OGG1 polymorphisms and breast cancer risk. Cancer Epidemiol
Biomarkers Prev 15: 811-815
25. Figueroa JD, Malats N, Real FX, Silverman D, Kogevinas M, Chanock S, et al
(2007) Genetic variation in the base excision repair pathway and bladder cancer
risk. Hum Genet 121: 233-242
26. Dorszewska J, Kempisty B, Jaroszewska-Kolecka J, Rózycka A, Florczak J,
Lianeri M, et al (2009) Expression and polymorphisms of gene 8-oxoguanine
glycosylase 1 and the level of oxidative DNA damage in peripheral blood
Table 1 Characteristics of the OGG1 genetic polymorphisms and PCR condition for genotyping analysis.
Gene SNPs rs number Position Alleles Primers PCR product size PCR conditions Restriction enzyme DNA fragment size (bp) (annealing temperature)
OGG1 C-285T rs159153 3:9729875 C/T Forward:5'-AGGGCAAAGGGGATACAAAG-3' 347 57℃ DdeI C:154+113+80
OGG1 Reverse:5'-CTGGTTGAAGAGCCAGGTTT-3' T:267+80
OGG1 A5954Grs3219008 3:9735543 A/G Forward:5'-ATTCACCTCTTCCGGCTTCT-3' 320 60℃ MspI A: 12+308
Table 2 Genotypic and allelic frequencies of OGG1 genetic polymorphisms in the patients with RA and controls.
dbSNP ID Patients with RA Controls p value
(N=192) (N=192) rs159153 Genotype TT 158 (82.3) 153 (79.7) 0.629a TC 32 (16.7) 38 (19.8) CC 2 (1.0) 1 (0.5) CC + TC 34 (17.7) 39 (20.3) Allele frequency T 348 (90.6) 344 (89.6) 0.717 C 36 (9.4) 40 (10.4) rs3219008 Genotype AA 22 (11.5) 25 (13.0) 5.6E-05a AG 170 (88.5) 149 (77.6) GG 0 (0) 18 (9.4) GG + AG 170 (88.5) 167 (87.0) Allele frequency A 214 (55.7) 199 (51.8) 0.311 G 170 (44.3) 185 (48.2) a
Genotype distribution between patients and control were calculated by 2 x 3 chi-square test
Fig. 1 Association of OGG1 gene haplotypes with rheumatoid arthritis
Table 3 Association between clinical feature and genotype distribution of OGG-1 polymorphisms in the RA patients.
Clinical Parameters TT (n=158) non TT (n=34) p value AA (n=22) non AA (n=170) p value
Gender male 35 (22.2) 6 (17.6) 0.561 2 (9.1) 39 (22.9) 0.173 female 123 (77.8) 28 (82.4) 20 (90.9) 131 (77.1) Rheumatoid factor with 115 (72.8) 26 (76.5) 0.659 18 (81.8) 123 (72.4) 0.344 without 43 (27.2) 8 (23.5) 4 (18.2) 47 (27.6) Extra-articular with 72 (45.6) 19 (55.9) 0.275 13 (59.1) 78 (45.9) 0.243 without 86 (54.4) 15 (44.1) 9 (40.9) 92 (54.1) Bone Erosion with 80 (50.6) 17 (50.0) 0.947 17 (77.3) 80 (47.1) 0.008a without 78 (49.4) 17 (50.0) 5 (22.7) 90 (52.9) rs3219008 Patient with RA rs159153 a
Dear Editor,
Attacheded please find a copy of our manuscript entitled, “Association of the C-285T and A5954G Polymorphisms in the DNA repair gene OGG1 with the Susceptibility of Rheumatoid Arthritis.” to be considered for publication in Rheumatology International.
This manuscript comprised a study of the human Rheumatoid Arthritis (RA). We tested a hypothesis that the OGG1 genetic polymorphism confers RA susceptibility. Study participants were Taiwanese RA patients and a healthy control group. Our data indicated that OGG1 genetic polymorphism (rs3219008) contributes to the susceptibility of RA.
"None of the authors has any potential financial conflict of interest related to this manuscript."
Thank you for your time and consideration. We look forward to hearing from you in due time. Should you have any questions, please feel free to contact me.
Very truly yours,
Shih-Yin Chen, Ph.D. Assistant Professor,
Graduate Institute of Chinese Medical Science, China Medical University,
91 Hsueh-Shih Road,Taichung, Taiwan 40402, R.O.C Tel:+886- 4-22052121 ext 2033
Fax:+886- 4-22033295
e-mail: [email protected]