Adjunctive Traditional Chinese Medicine Therapy Improves Survival of Liver
Cancer Patients
Yueh-Hsiang Liao1,2, Cheng-Chieh Lin3,4,5, Hsueh-Chou Lai4,6, Jen-Huai Chiang7,8,9,
Jaung-Geng Lin1*, Tsai-Chung Li8,9,10*
1. School of Chinese Medicine, China Medical University, Taichung, Taiwan 2. School of Chinese Medicine for Post-Baccalaureate, I-Shou University,
Kaohsiung, Taiwan
3. Department of Family Medicine, China Medical University Hospital, Taichung, Taiwan 4. School of Medicine, College of Medicine, China Medical University, Taichung, Taiwan 5. Department of Medical Research, China Medical University Hospital, Taichung, Taiwan 6. Division of Hepatogastroenterology, Department of Internal Medicine, China
Medical University Hospital, Taichung, Taiwan
7. Management Office for Health Data, China Medical University Hospital, Taichung, Taiwan
8. Research Center for Chinese Medicine & Acupuncture, China Medical University, Taichung, Taiwan
9. Graduate Institute of Biostatistics, China Medical University, Taichung, Taiwan 10. Department of Healthcare Administration, College of Medical and Health
Science, Asia University, Taichung, Taiwan
* Correspondence to: Tsai-Chung Li and Jaung-Geng Lin
China Medical University, 91 Hsueh-Shih Road, Taichung, 40402, Taiwan, Tel: 886-4-2205-3366 ext. 6605, Fax: 886-4-2207-8539, e-mail: [email protected]
Electronic word count for main body of manuscript: 2500 words. Number of figures and tables: 4 tables and 1 figure.
Abbreviations: HBV, hepatitis B virus; HCV, hepatitis C virus; TACE, transcatheter arterial chemoembolization; TCM, traditional Chinese medicine; NHI, National Health Insurance; NHIRD, National Health Insurance Research Database; ICD-9-CM, International Classification of Diseases, Ninth Revision, Clinical Modification; CI, confidence interval; HR, hazard ratio.
Disclosure: The authors have declared no conflicts of interest.
Funding: This study is supported in part by Taiwan Ministry of Health and Welfare Clinical Trial and Research Center of Excellence (MOHW 104-TDU-B-212-113002), Health and welfare surcharge of tobacco products, China Medical University Hospital Cancer Research Center of Excellence (MOHW 104-TD-B-111-03, Taiwan) and China Medical University under the Aim for Top University Plan of the Ministry of Education, Taiwan.
Key points box:
1. The adjunctive therapy with TCM may improve the overall survival in liver cancer patients.
2. A significant protective effect of TCM use with a 35% reduction on overall survival was observed.
3. Similar significant protective effects of TCM use were found across various subgroups of chronic liver diseases, indicating consistency in the current findings. 4. Jia Wei Xiao Yao San and Chai Hu Shu Gan Tang were the most effective TCM
Abstract
Background: Traditional Chinese medicine (TCM) is an alternative treatment for
cancer with its effect by stimulating host immune response for cytotoxic activity
against liver cancer. No studies evaluated TCM treatment on survival of liver cancer
patients.
Patients and methods: This study determined whether the combination of TCM and
conventional cancer treatment affects the survival of liver cancer patients. A
retrospective cohort study was conducted in 127,237 newly diagnosed liver cancer
patients from 2000 to 2009 in the National Health Insurance Program database. Results: Among these patients, 30,992 (24.36%) used TCM for liver cancer care. All
patients were followed up until 2011. The mean follow-up was 5.67 years (SD 1.47)
for TCM users and 5.49 years (SD 3.64) for non-TCM users. Compared with patients
without TCM use, patients with TCM use were significantly associated with a
decreased risk of death [hazard ratio (HR) = 0.65, 95% confidence interval (CI) =
0.64 to 0.66] with multivariate adjustment. A similar significant protective effect of
TCM use across various subgroups of chronic liver diseases was also observed. Jia
Wei Xiao Yao San (HR = 0.89, 95% CI = 0.81 to 0.96) and Chai Hu Shu Gan Tang
improved survival.
Conclusions: This cohort study provided information that adjunctive therapy with
TCM may improve the survival in liver cancer patients. Further studies are needed to
confirm the potential role of TCM in HCC.
Introduction
Liver cancer is the second leading cause of cancer death in men and the sixth in
women worldwide [1]. Hepatitis B virus (HBV) and hepatitis C virus (HCV)
infections have high prevalence rates. Taiwan and China have the third and fourth
highest incidence rates of liver cancer worldwide, and their age-standardized
incidence rates are 48.62 [2] and 29.0 [1] per 100,000 persons, respectively [1, 2]. In
Taiwan, liver cancer accounts for more than 34.9% of total deaths [2] and ranks as the
second most common cancer in both men and women, accounting for approximately
20% of all cancer mortalities. The annual standardized mortality incidence increased
from 22.7 per 100,000 in 1991 to 24.7 per 100,000 in 2012 [2]. In Hong Kong, liver
cancer accounts for more than 10% of total deaths and ranks as the fourth most
common cancer in 2010 [3].
Liver cancer has poor prognosis with a five-year survival rate of 12% [4].
Prompt diagnosis of early-stage liver cancer is hindered by the lack of a sensitive
screening for this disease. More than 60% of newly diagnosed liver cancer cases are
at an advanced stage [5], with therapeutic options limited to palliative approaches
using transcatheter arterial chemoembolization (TACE) or chemotherapeutic agents
[4]. However, many patients poorly respond to TACE or suffer from poor outcomes
Complementary and alternative medicine for liver cancer care has gained
worldwide popularity. Traditional Chinese medicine (TCM) is popular in Chinese and
East Asian societies and plays an active role in modern health care system covered by
the National Health Insurance (NHI) program in Taiwan. The effect of TCM has
targeted the stimulation of the host immune response for cytotoxic activity against
liver cancer by inhibiting proliferation and promoting apoptosis of tumor cells [2, 7, 8,
9], thereby alleviating chemoradiotherapy-related or gene-therapy-related side effects
[10, 11]. However, these studies on TCM treatment for liver cancer have been
conducted in laboratories. Only one multi-center randomized control trial has
evaluated the effects of TCM use in preventing recurrence after resection of small
hepatocellular carcinoma [12]. To date, no study has evaluated the effects of TCM
treatment on survival for liver cancer. Thus, the current study was designed to
determine whether the combination of TCM and conventional cancer treatment
affected the survival of liver cancer patients. Methods
Research Database
The NHI program in Taiwan is a compulsory and single-payer program that was
The national government-run Bureau of National Health Insurance conducts an expert
review of random samples from every 50 to 100 ambulatory and inpatient claims in
each hospital and clinic quarterly, and false reports of diagnoses are severely
penalized [13]. The datasets of the study consisted of registry for beneficiaries,
ambulatory and inpatient care claims, and registry for catastrophic illness from 1999
to 2009 from the National Health Insurance Research Database (NHIRD), which was
released by the Taiwan National Health Research Institute. The registry for
catastrophic illness database contains data from insurers who suffer from major
diseases and are granted exemption from co-payment. All cancer cases registered in
the catastrophic illness database should be confirmed by pathological reports. For a
patient that meets the criteria of the catastrophic illness database, the attending
physicians and medical institution should provide assistance for the patient’s
application for the catastrophic illness certificate. The NHIRD database contains the
following information: demographic data; dates of clinical visits; diagnostic codes by
the International Classification of Diseases, Ninth Revision, Clinical Modification
(ICD-9-CM); details of prescriptions; and expenditure amounts of beneficiaries. Data
for detailed diagnoses and treatments provided by Chinese medicine physicians are
Study subjects
A retrospective study was conducted using the registration and claim datasets of
the catastrophic illness database for 1999 to 2009. Liver cancer patients (aged ≥30
years) who were diagnosed with ICD-9-CM code 155 were identified from the
catastrophic illness database covering 2000 to 2009 and were followed up until
December 31, 2011. However, some patients withdrew from the NHI program, or
some had died. To protect the patients’ privacy, data regarding their identities and
institutions were cryptographically scrambled in the NHIRD. The current study was
exempted from obtaining the institutional review board approval of the Public Health,
Social and Behavioral Science Committee Research Ethics Committee, China
Medical University and Hospital.
Variables for coexisting diseases and treatment of liver cancer
In this study, coexisting diseases were identified from the disease records in the
ICD-9-CM for outpatient, inpatient, and catastrophic illness registry files. Data for
these coexisting conditions were collected for a 48-month period preceding cohort
entry. Histories of diabetes mellitus (ICD-9-CM code 250), cerebral vascular disease
(ICD-9-CM codes 430–438), renal failure (ICD-9-CM codes 584, 585, and 586),
nonalcoholic fatty liver disease (ICD-9-CM code 571.8), HBV (ICD-9-CM codes
070.2, 070.3, and V02.61), and HCV (ICD-9-CM codes 070.41, 070.44, 070.51,
070.54, and V02.62) were identified as comorbidities before the index date.
Treatment type was the treatment that a patient with liver cancer received after the
index date but two years prior to the end of the follow-up period. The treatment types
included hepatectomy, partial hepatectomy, hepatic lobectomy, total hepatectomy,
liver transplantation, radiofrequency ablation, TACE, systemic chemotherapy, and
radiation therapy. Primary outcome
The primary outcome was all-cause mortality during the 11-year follow-up.
Patients’ date of death was determined from the registry for catastrophic illness of the
NHIRD. All eligible patients were followed up from the index date to the death of the
patient, withdrawal from NHI, or the end of 2011, whichever occurred first. Statistical analysis
Continuous variables were reported as mean and 95% confidence interval (CI),
whereas categorical variables were reported as number, percentage, and 95% CI.
Differences in proportions and means were assessed using χ2- or t-test. Survival
analysis was performed using Kaplan–Meier method, and significant difference was
were used to examine the effect of TCM use on survival, which was determined by
adjusted hazard ratio (HR) with 95% CI. All reported p values were obtained from
two-sided tests. Statistical significance was set to p < 0.05. All analyses were
performed using SAS version 9.3 (SAS Institute Inc., Cary, NC, USA). Results
Baseline sociodemographic factors, comorbidities, and type of treatment
between TCM users and non-users among liver cancer patients are shown in Table 1.
TCM users were linked with the following factors: higher proportions of age groups
<50, 50s, and 60s; female gender; with insured amount of 20,000 to 39,999, 40,000 to
59,999, and ≥60,000 NT$/month; residence in an area with urban levels 1 and 2 or at
central area; insured units by government/school employees; private enterprise
employees and members of occupational sector; with administered partial
hepatectomy, lobectomy of liver, liver transplantation, radiofrequency ablation,
TACE, systemic chemotherapy, and radiation therapy; lower prevalence of TCM use
one year prior to liver cancer diagnosis; and with diabetes, cerebral vascular disease,
renal failure, and cirrhosis.
A total of 97,748 deaths were observed during the follow-up period with crude
without and with TCM use, respectively. Kaplan–Meier analysis demonstrated that
the survival curves were significantly lower in TCM users than in non-TCM users
(log-rank test, p < 0.001). The mean follow-up period was 5.67 years (SD 1.47) and
5.49 years (SD 3.64) for TCM and non-TCM users, respectively. Table 2 shows the
crude and adjusted HRs of survival for TCM use status along with sociodemographic
factors, comorbidity, and treatment types in liver cancer patients. Compared with
non-TCM users, non-TCM users were significantly associated with a decreased risk of death
(HR = 0.64, 95% CI = 0.63 to 0.64) without adjustment. The HR remained the same
after adjusting for sociodemographic factors, comorbidity, and treatment types (HR =
0.65, 95% CI = 0.64 to 0.66). The other risk factors that significant increased risk of
death were as follows: old age; male gender; higher levels of insured amount
(≥20,000 NT$/month); urban levels of 2 and 3; insured units of private enterprise
employees; members of occupational sector; low-income household/veterans;
comorbidity of diabetes, cerebral vascular disease, renal failure, and cirrhosis; and
administration or performance of systemic chemotherapy and radiation therapy. By
contrast, the risk factors that significantly reduced risk of death were as follows:
nonalcoholic fatty liver disease; and administration or performance of hepatectomy,
radiofrequency ablation, and TACE.
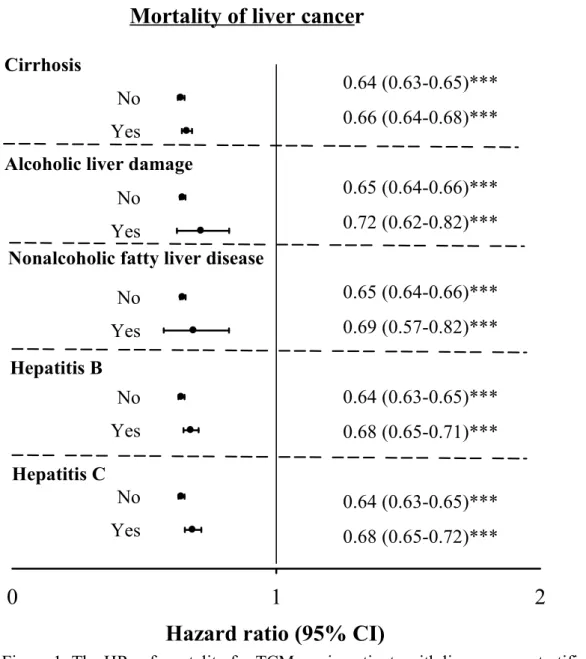
The HRs of mortality for TCM use in liver cancer patients stratified by
cirrhosis, alcoholic liver damage, nonalcoholic fatty liver disease, hepatitis B, and
hepatitis C are shown in Figure 1. Similar significant protective effects of TCM use
were found across various subgroups of chronic liver diseases, indicating consistency
in the current findings.
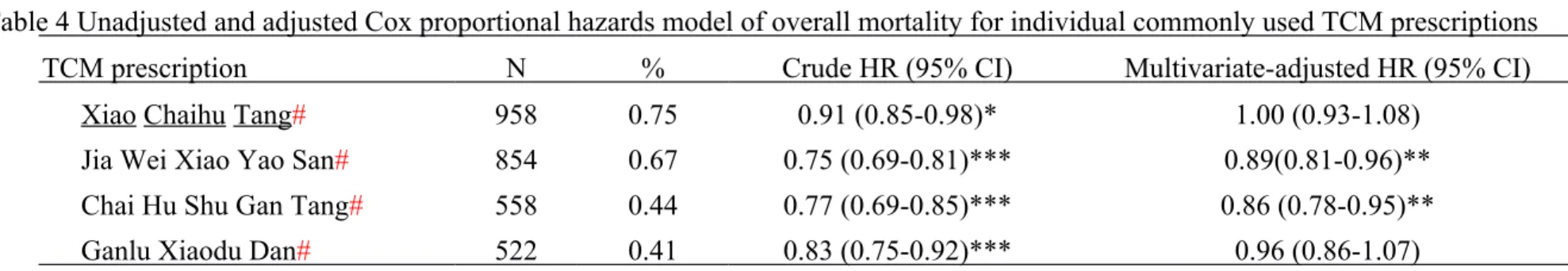
The six most common prescriptions were Xiao Chaihu Tang (n = 958, 0.75%),
Jia Wei Xiao Yao San (n = 854, 0.67%), Yinchen Wuling San (n = 715, 0.56%),
Xiang Sha Liu Jun Zi Tang (n = 632, 0.5%), Chai Hu Shu Gan Tang (n = 558,
0.44%), and Ganlu Xiaodu Dan (n=522, 0.41%). Among these prescriptions, Yinchen
Wuling San and Xiang Sha Liu Jun Zi Tang were not used to treat liver cancer but
hyperlipoproteinemia [14], chronic gastritis, and appetite loss [15]. Thus, these
prescriptions were not considered in the further analysis. Table 3 shows the TCM
name, ingredient or generic name, and functional classification of the commonly used
TCM prescriptions in liver cancer patients. Cox proportional hazard regression
analysis demonstrated that the two most effective TCM agents in increasing survival
were Jia Wei Xiao Yao San (HR = 0.89, 95% CI = 0.81 to 0.96), and Chai Hu Shu
Discussion
This study is the first large-scale cohort that focused on the effect of TCM use
on the survival among liver cancer patients. The association between TCM use and
survival in a large cohort of liver cancer patients in Taiwan was evaluated. This
retrospective cohort study included 127,237 liver cancer patients, and a significant
protective effect of TCM use with a 35% reduction on survival was observed. The most common TCM prescriptions used by liver cancer patients in the
present study were similar to those reported by Chen et al. [16]. Among these
prescriptions, Jia Wei Xiao Yao San and Chai Hu Shu Gan Tang exhibited significant
protective effect on survival. Jia Wei Xiao Yao San was also the most common
prescription used in treating patients with colon cancer or breast cancer [17, 18]. This study is the first large-scale cohort of liver cancer patients for TCM use in
Taiwanese society, and this study includes all diagnosed liver cancer cases confirmed
by pathological reports. The NHI databases are representative of the general
population because the NHI covered more than 99% of Taiwan’s 23 million
population and 93% of the medical institutes. Moreover, given that the NHI covers
TCM health care, the TCM prescription data obtained are completely recorded, which
records includes drug names, dose, and days of administration. These drugs were
prescribed by board-certified Chinese medicine physicians. The complete list of TCM
prescription facilitates the evaluation of the effects of each type of prescription on the
survival of liver cancer patients, which is more comprehensive than merely evaluating
a particular type of TCM prescription.
Randomized controlled trials for TCM plus TACE to treat patients with
unresectable HCC have demonstrated the effect of TCM on the stimulation of host
immune response that has cytotoxic activity against HCC [19-21]. The findings of
these randomized controlled trials revealed that TCM plus TACE improved patient
survival, quality of life, alleviation of symptoms, and tumor response [19]. TCM also
significantly increases the survival, complete or partial response, non-deterioration
performance, T cells, natural killer cells, and white blood cell count as well as
decreased the level of blood alpha-fetoprotein concentration and significantly lowered
the incidence of nausea and vomiting [20]. This type of treatment also increased the
proportions of cluster differentiation CD3(+) T cells, CD4(+) T cells, and natural
killer cells as well as the ratio of CD4(+)/CD8(+) before and after treatment [21]. Jia
Wei Xiao Yao San displayed hepatoprotective effect on dimethylnitrosamine-induced
of Chai Hu Shu Gan Tang that has significant potential in combination with adjuvant
remedy with tumor necrosis factor-alpha for cancer patients by inhibiting the
activated T lymphocytes via suppression of NF-k B, NF-AT, and AP-1 signaling [23]. This study has several limitations. First, the findings were limited by potential
residual and unrecognized confounding variables because of the observational study
design. Second, measurement errors could be attributed to the administrative claim
data gathered from clinical practice. For instance, the clinical cases with diseases or
disorders such as nonalcoholic fatty liver disease, hepatitis B, hepatitis C,
hypercholesterolemia, and hypertension were those who seek health care services,
accounting for a small proportion of the entire patients with these diseases/disorders.
Thus, the prevalence of these diseases or disorders was underestimated and was
associated with health care utilization when case ascertainment was based on ICD-9
codes. Thus, these diseases or disorders were not considered in the present study.
Although this study did not consider these diseases or disorders, the possibility of
confounding effects arising from these diseases or disorders were small because there
was no evidence that these diseases or disorders were associated with higher
probability of TCM use in patients with HCC because HCC has been linked with
TCM, were not covered by the NHI program, and visits at clinics not contracted with
the NHI were not considered in the current study. Less than 10% of TCM clinics are
not contracted with NHI. Cancer patients also need rare and expensive Chinese herbal
medicines, which were not covered by the NHI program. Thus, the use of TCM could
be underestimated. Fourth, clinicopathology such as cancer staging, which was
associated with prognosis, was not available in the study. In our study, we only had
information about distant metastases and treatment type, which had been adjusted for
their confounding effects. The proportions of distant metastases, systemic
chemotherapy, and radiation therapy were slightly higher in TCM use than in
non-TCM use, indicating that the severity of cancer may be higher in non-TCM use. Thus, the
effect of TCM use on increased survival may be underestimated, which is a lesser
threat to the validity of our study finding. Lastly, only the date of death was recorded
in the registry for catastrophic illness in the NHIRD. Therefore, the effect of TCM use
on liver cancer-specific mortality cannot be completely analyzed. Conclusion
This cohort study showed that adjunctive therapy with TCM may improve the
survival of liver cancer patients. TCM may be used as an integral element of effective
Reference
1. Worldwide Health Office. Cancer Incidence, Mortality and Prevalence Worldwide in 2012. http://globocan.iarc.fr (July 2014, date last accessed). 2. Ministry of Health and Welfare R..O.C.
http://www.mohw.gov.tw/cht/DOS/Statistic.aspx?
f_list_no=312&fod_list_no=2747 (July 2014, date last accessed). 3. Hospital Authority Hong Kong Cancer Registry.
www3.ha.org.hk/cancereg/c_stat.asp (July 2014, date last accessed).
4. El-Serag HB. Hepatocellular carcinoma. N Engl J Med. 2011; 365(12): 1118-1127.
5. Llovet JM, Di Bisceglie AM, Bruix J, et al. Design and endpoints of clinical trials in hepatocellular carcinoma. J Natl Cancer Inst. 2008; 100(10): 698-711. 6. Andreas Pircher, Michael Medinger, Joachim Drevs. Liver cancer: targetedfuture
options. World J Hepatol. 2011; 3(2): 38-44.
7. Cao Z, Lin W, Huang Z, Chen X, et al. Ethyl acetate extraction from a Chinese herbal formula, Jiedu Xiaozheng Yin, inhibits the proliferation of hepatocellular carcinoma cells via induction of G0/G1 phase arrest in vivo and in vitro. Int J Oncol. 2013; 42(1): 202-210.
8. Wu GG, Li WH, He WG, et al. Mir-184 post-transcriptionally regulates SOX7 expression and promotes cell proliferation in human hepatocellular carcinoma. PLoS One. 2014; 9(2): e88796.
9. Ling CQ, Yue XQ, Ling C. Three advantages of using traditional Chinese medicine to prevent and treat tumor. J Integr Med. 2014; 12(4): 331-335. 10. Konkimalla VB and Efferth T. Evidence-based Chinese medicine for cancer
therapy. J Ethnopharmacol 2008; 116: 207-210.
11. Ling CQ, Wang LN, Wang Y, Zhang YH, Yin ZF, Wang M, Ling C. The roles of traditional Chinese medicine in gene therapy. J Integr Med. 2014; 12(2): 67-75.
12. Zhai XF, Chen Z, Li B, Shen F, Fan J, Zhou WP, Yang YK, Xu J, Qin X, Li LQ, Ling CQ. Traditional herbal medicine in preventing recurrence after resection of small hepatocellular carcinoma: a multicenter randomized controlled trial. J Integr Med. 2013; 11(2): 90-100.
13. Liao YH, Lin CC, Li TC, et al. Utilization pattern of traditional Chinese medicine for liver cancer patients in Taiwan BMC Complementary and Alternative Medicine 2012; 12: 146.
14. Yu R, Wang DS, Zhou H. Clinical and experimental study on effects of yinchen wuling powder in preventing and treating hyperlipoproteinemia. Zhongguo Zhong Xi Yi Jie He Za Zhi. 1996; 16(8): 470-473.
15. Complementary and Alternative Healing University.
http://alternativehealing.org/xiang_sha_liu_jun_zi_tang.htm (July 2014, date last accessed).
16. Chen FP, Kung YY, Chen YC, et al. Frequency and pattern of Chinese herbal medicine prescriptions for chronic hepatitis in Taiwan. J Ethnopharmacol. 2008; 117(1): 84-91.
17. Wang BR, Chang YL, Chen TJ, et al. Coprescription of Chinese herbal medicine and Western medication among female patients with breast cancer in Taiwan: analysis of national insurance claims. Patient Prefer Adherence. 2014; 8: 671-682.
products for post-surgery colon cancer patients in Taiwan. J Ethnopharmacol. 2014; 155(1): 702-708.
19. Meng MB, Cui YL, Guan YS, et al. Traditional Chinese medicine plus transcatheter arterial chemoembolization for unresectable hepatocellular carcinoma. J Altern Complement Med 2008; 14(8): 1027–1042.
20. Cho WC, Chen HY. Transcatheter arterial chemoembolization combined with or without Chinese herbal therapy for hepatocellular carcinoma: meta-analysis. Expert Opin Investig Drugs 2009; 18(5): 617–635.
21. Meng MB, Wen QL, Cui YL, et al. Meta-analysis: traditional Chinese medicine for improving immune response in patients with unresectable hepatocellular carcinoma after transcatheter arterial chemoembolization. Explore (NY) 2011; 7(1): 37–43.
22. Chien SC, Chang WC, Lin PH, et al. A Chinese Herbal Medicine, Jia-Wei-Xiao-Yao-San, Prevents Dimethylnitrosamine-Induced Hepatic Fibrosis in Rats. ScientificWorldJournal. 2014; 2014: 217525.
23. Wong VK, Zhang MM, Zhou H, et al. Saikosaponin-d Enhances the Anticancer Potency of Tα via Overcoming Its Undesirable Response of Activating NF-Kappa B Signalling in Cancer Cells. Evid Based Complement Alternat Med. 2013; 2013: 745295.
Table1. Liver cancer patient characteristics during the period 2000-2009
Characteristic
TCM Nonusers TCM Users P value
Total % Total % Patient no. 96245 75.64 30992 24.36 Age <0.001 <50 18239 18.95 6988 22.55 50s 20499 21.3 7801 25.17 60s 25076 26.05 8853 28.57 70s 32431 33.7 7350 23.72 Gender <0.001 Female 29326 30.47 10925 35.25 Male 66919 69.53 20067 64.75
Visit one year ago <0.001
TCM Nonusers 72746 75.58 12807 41.32
TCM Users 23499 24.42 18185 58.68
Insured amount (NT$/month) <0.001
<20,000 45493 47.27 12789 41.27 20,000-39,999 35874 37.27 12494 40.31 40,000-59,999 9132 9.49 3543 11.43 >=60,000 5746 5.97 2166 6.99 Urban Level <0.001 1 23883 24.81 7993 25.79 2 26380 27.41 9153 29.53 3 14740 15.32 4847 15.64 4 16718 17.37 5183 16.72 5 14524 15.09 3816 12.31 Residential Area <0.001 Northern 11319 11.76 3337 10.77 Taipei 28739 29.86 8277 26.71 Central 15404 16.00 7440 24.01 Southern 20745 21.55 6282 20.27 Eastern 2288 2.38 540 1.74 Kao-Ping 17750 18.44 5116 16.51 Insured unit <0.001 Government, school employees 6604 6.86 2478 8.00
Private enterprise employees 22991 23.89 8377 27.03 Member of occupational 16195 16.83 5811 18.75
Farmers, fishermen 28051 29.15 7548 24.35 Low-income households
Veterans, other regional 22404 23.28 6778 21.87 Comorbidities
Diabetes 22157 23.02 6348 20.48 <0.001
Cerebral vascular disease 7042 7.32 1493 4.82 <0.001
Renal failure 5476 5.69 1090 3.52 <0.001 Cirrhosis 31785 33.03 9226 29.77 <0.001 Type of treatment Hepatectomy 241 0.25 83 0.27 0.64 Partial hepatectomy 10454 10.86 4634 14.95 <0.001 Lobectomy of liver 3316 3.45 1408 4.54 <0.001 Total hepatectomy 95 0.10 39 0.13 0.24 Liver transplantation 713 0.74 324 1.05 <0.001 Radiofrequency ablation 215 0.22 126 0.41 <0.001 TACE 6609 6.87 2700 8.71 <0.001 Systemic chemotherapy 30123 31.30 12278 39.62 <0.001 Radiation therapy 7549 7.84 3003 9.69 <0.001 Distant metastases 0.02 No 81497 84.68 26069 84.12 Yes 14748 15.32 4923 15.88
Table 2 The hazard ratios (HRs) of mortality of liver cancer for sex, age, time, insurance premium, urbanization level, comorbidities and type of treatment
Variable Crude HR (95% CI) Multivariate-adjusted HR (95% CI) Visit one year
TCM Nonusers 1.00 1.00 TCM Users 0.64 (0.63-0.64)*** 0.64 (0.63-0.66)*** Age <50 1.00 1.00 50s 1.11 (1.09-1.13)*** 1.14 (1.12-1.17)*** 60s 1.23 (1.20-1.25)*** 1.25 (1.23-1.28)*** 70s 1.67 (1.64-1.70)*** 1.58 (1.55-1.62)*** Gender Female 1.00 1.00 Male 1.20 (1.18-1.22)*** 1.27 (1.25-1.29)***
Visit one year ago
TCM Nonusers 1.00 1.00
TCM Users 0.92 (0.91-0.93)*** 1.09 (1.08-1.11)***
Insured amount (NT$/month)
<20,000 1.00 1.00 20,000-39,999 0.60 (0.59-0.61)*** 0.50 (0.49-0.50)*** 40,000-59,999 0.60 (0.59-0.62)*** 0.56 (0.55-0.57)*** >=60,000 0.57 (0.55-0.59)*** 0.53 (0.51-0.55)*** Urban Level 1 1.00 1.00 2 1.02 (1.00-1.04)* 0.96 (0.95-0.98)*** 3 1.13 (1.11-1.15)*** 1.04 (1.02-1.06)*** 4 1.14 (1.12-1.16)*** 1.00 (0.98-1.02) 5 1.18 (1.15-1.20)*** 0.99 (0.96-1.01) Residential Area Northern 1.00 1.00 Taipei 0.94 (0.92-0.96)*** 0.97 (0.95-0.99)* Central 1.07 (1.05-1.10)*** 1.10 (1.08-1.13)*** Southern 1.07 (1.04-1.09)*** 1.04 (1.02-1.06)*** Eastern 1.43 (1.37-1.50)*** 1.38 (1.32-1.44)*** Kao-Ping 1.09 (1.07-1.12)*** 1.05 (1.02-1.07)*** Insured unit Government, school employees 1.00 1.00
TCM: traditional Chinese Medicine; TACE: transarterial chemoembolization. *:P<0.05; **:p<0.01; ***: p<0.001.
Table 3. The TCM name, ingredients or generic name, and functional classification of the commonly used TCM prescriptions in patients with liver cancer
TCM Name Ingredients or Generic Name Functional Classification No. of
Users
%
Xiao Chaihu Tang Bupleurum chinense, Scutellariae Radix, Pinellia ternate, Codonopsis pilosula, Radix Glycyrrhiza, Zingiber officinale, Zizyphus Jujuba
Regulating exterior and interior qi activity by bearing and reducing smooth blood flow and balancing between Yin and Yang.
958 0.75
Jia Wei Xiao Yao San Angelica sinensis, Atractylodes macrocephala, Bupleurum chinensis, Gardenia jasminoides, Glycyrrhiz auralensis, Mentha haplocalyx, Paeonia lactiflora, Paeonia suffruticosa, Poria cocos, Zingiber officinale
Harmonizing and releasing formulae 854 0.67
Yinchen Wuling San Artemisia capillaris, Polyporus umbellatus Fr., Poria cocos, Alisma canaliculatum, Atractylodis Rhizoma, Cinnamomi Ramulus
Heat-clearing and dehumidification 715 0.56
Xiang Sha Liu Jun Zi Tang
Amomum villosum, Atractylodes macrocephala, Aucklandia lappa, Citrus reticulata, Radix Glycyrrhizae, Panax ginseng, Pinellia ternata, Poria cocos, Zingiber officinale
Tonifying and replenishing formulae 632 0.50
Fructus Citri Aurantii,Cyperus
rotundus,Ligusticum chuanxiong,Paeonia lactiflora Pall.,Radix Glycyrrhizae,
follow and relieving chest pain
Ganlu Xiaodu Dan Pulvis talci, Scutellaria baicalensis Georgi, Artemisia capillaries, Grassleaf Sweelflag Rhizome, Bulbus fritillariae cirrhosae, Akebia longeracemosa Matsum, Agastache rugosa, Forsythia suspense, Alpinia tonkinensis Gagnep, Mentha haplocalyx, Belamcanda chinensis
Table 4 Unadjusted and adjusted Cox proportional hazards model of overall mortality for individual commonly used TCM prescriptions
TCM prescription N % Crude HR (95% CI) Multivariate-adjusted HR (95% CI)
Xiao Chaihu Tang# 958 0.75 0.91 (0.85-0.98)* 1.00 (0.93-1.08)
Jia Wei Xiao Yao San# 854 0.67 0.75 (0.69-0.81)*** 0.89(0.81-0.96)**
Chai Hu Shu Gan Tang# 558 0.44 0.77 (0.69-0.85)*** 0.86 (0.78-0.95)**
Ganlu Xiaodu Dan# 522 0.41 0.83 (0.75-0.92)*** 0.96 (0.86-1.07)
Abbreviation: 95% CI, 95% confidence interval; HR, hazards ratio; TCM, traditional Chinese medicine. #: the reference group included patients without TCM use and patients with TCM use but without the specific prescription.
Multivariate-adjusted for age, gender, insured amount, urban level, residential area, insured unit, and type of treatment *:P<0.05; **:p<0.01; ***: p<0.001.
Figure Legends:
Figure 1: The HRs of mortality for TCM use in patients with liver cancer stratified by status of cirrhosis, alcoholic liver damage, nonalcoholic fatty liver disease, hepatitis B and hepatitis C.
Hazard ratio (95% CI)
0
1
2
No Yes No Yes No Yes No Yes No YesMortality of liver cancer
0.64 (0.63-0.65)*** 0.66 (0.64-0.68)*** 0.65 (0.64-0.66)*** 0.72 (0.62-0.82)*** 0.65 (0.64-0.66)*** 0.69 (0.57-0.82)*** 0.64 (0.63-0.65)*** 0.68 (0.65-0.71)*** 0.64 (0.63-0.65)*** 0.68 (0.65-0.72)***
Alcoholic liver damage Cirrhosis
Nonalcoholic fatty liver disease
Hepatitis B
Hepatitis C
Figure 1: The HRs of mortality for TCM use in patients with liver cancer stratified by status of cirrhosis, alcoholic liver damage, nonalcoholic fatty liver disease, hepatitis B and hepatitis C.