Title: Socio-demographic and meteorological correlates of sudden infant death in Taiwan
Type of manuscript: Original Article Running head: sudden infant death Authors’ names and affiliations:
Hua-Pin Chang
1,2, Chung-Yi Li
3,4, Ya-Hui Chang
3, Shiow-Li Hwang
5, Ying-Hwa Su
6*, Chi-Wen Chen
7*
1 Department of Nursing, Chung Shan Medical University Hospital, Taichung, Taiwan.
2 Institute of Medicine, Chung Shan Medical University, Taichung, Taiwan
3 Department of Public Health, College of Medicine, National Cheng Kung University, Tainan, Taiwan
4 Department of Public Health, College of Public Health, China Medical University , Taichung, Taiwan
5 School of Nursing, National Taipei University of Nursing and Health Sciences, Taipei, Taiwan.
6 Department of Nursing, College of Medicine, National Cheng Kung University, Tainan, Taiwan
7 Department of Nursing, College of Medicine, Fu-Jen Catholic University, Taipei, Taiwan
* Ying-Hwa Su and Chi-Wen Chen contribute to this study equally.
The author to whom correspondence should be sent:
Chi-Wen Chen, PhD, RN Associate professor
Department of Nursing, College of Medicine, Fu-Jen Catholic University No. 510, Chung-Cheng Rd., Hsin-Chuan
Taipei 24205, Taiwan
Tel: 886-2-29053459; Fax: 886-2-29052093 e-mail: [email protected]
Number of text pages: 20
Number of reference pages: 5 Number of tables: 2
Abstract
Background: This study was designed, using three national datasets including the
Taiwan Death Registry, Taiwan Birth Registry, and National Meteorological Dataset, to examine the socio-demographic, geographic and meteorological correlates of sudden
infant death syndrome (SIDS).
Methods: One thousand six hundred seventy-one cases of SIDS occurring between
1994 and 2003 and 8,355 matched controls were included in this nested case-control
study.
Results: Over the study period, the annual rate of SIDS declined only slightly, with an
averaged annual rate of 57.9/10
5. Male infants [AOR, 1.19; 95% CI-1.06, 1.33], preterm births [AOR, 1.69; 95% CI-1.33, 2.13], low birth weight [AOR, 2.87; 95% CI- 2.30, 3.59], and birth order > =3 [AOR, 1.62; 95% CI-1.37, 1.92] were the demographic risk factors for SIDS. Additionally, parental age with < 25 years [AOR, 1.37; 95% CI- 1.09, 1.71], urbanization [AOR, 1.46; 95% CI-1.20, .78], lower paternal education
(elementary and less) [AOR, 1.28; 95% CI-1.01, 1.64], and parental age difference less
than 10 years [AOR, 1.28; 95% CI-1.01, .64] were also associated with increased risks
of SIDS. We also noted that daily average temperature ranging from 9.2 to 14.2℃
26.4℃ or higher [AOR 0.60, 0.61] was significantly associated with the most reduced
risk.
Conclusion: Socio-demographic, geographic and meteorological data can be used to
identify families in greater need of early guidance and to promote various prevention
measures to avoid the occurrence of SIDS.
Key words nested case-control studies, sudden infant death, temperature,
socioeconomic factors, demographic factors
Sudden infant death syndrome (SIDS) is a common cause-of-death for infants between 28 days and 1 year of age. Causes of SIDS are largely unexplained even with a comprehensive case investigation, including autopsy examination and review of clinical histories.
1Despite that, several studies have pointed to an association of socio-
demographic, preterm, low birth weight,
2-7tobacco exposure,
8-10and prone sleeping position
11-13with SIDS. Additionally, some previous investigations reported that certain meteorological factors such as temperature might also play a role in the cause of
SIDS.
14-17Lee & Chung reported that the incidence rate of SIDS in Taiwan increased from 0.25 per 1,000 live births in 1984 to 0.56 per 1,000 live births in 1993.
18The
population-based study by Wu et al. noted the incidence rate of SIDS was 0.36 per 1,000 live births between 2000 and 2006 in Taiwan. The reported incidence of SIDS was slightly lower than that in the United States (0.58/1,000 live births) and close to that of United Kingdom (0.34/1,000 live births) but slightly higher than that in Japan
(0.26/1,000 live births). They also indicated male predominance and the more
frequently occurrence in the winter season (from December to March).
19In this national
study, we sought to further realize the potential influences of socio-demographic and
factors can be of great help in design preventive measures that can effectively further reduce the occurrence of SIDS.
Methods
Source of data
This study used data from three national datasets including Taiwan Death Registry (TDR), Taiwan Birth Registry (TBR), and Meteorological Data Set (MDS). Data for nearly 3 million live births (n =2,899,443) between 1994 and 2003 were collected from the TBR. The birth certificate contains information on gender, birth order, and number of birth, gestational age, birth weight, maternal age at delivery, paternal age at delivery, and place of birth, urbanization level of living area, marital status of parents, maternal education, paternal education, and age difference of parental ages. The TBR has been evaluated and is considered valid and complete.
20Data on mortality were obtained from the TDR, which is considered accurate and
complete because it is mandatory to register all deaths in Taiwan and for physicians to
complete all death certificates.
21We retrieved information on the date of death and
underlying-cause-of-death (UCOD) for each deceased individual.
Meteorological data were provided by the Central Weather Bureau of Taiwan. A total of 46 meteorological monitoring stations (MMS) were installed around the nation.
Each MMS recorded every hour a variety of meteorological data including temperature, relative humidity, wind velocity, precipitation etc.
Study design
We used a case-control study design nested within 10 birth cohorts (i.e., live births in 1994–2003) of Taiwan. A total of 1,730 cases of SIDS (International Classification of Disease 9
thversion (ICD-9) Code: 798) were identified from TDR between 1994 and 2004. Linkage, using scrambled personal identification number, of the 1,730 cases of SIDS to the TBR was unsuccessful for 59 cases, with 1,671 left in the analysis. For each case, we randomly selected 5 control subjects who were born in the same year as the case and were alive on the day of case’s death. If a subject was selected more than once, only the first-time selection was retained, and the subsequent selections were replaced with another random selection.
The information concerning birth characteristics including sex, birth order (1, 2,
>=3), number of birth (singleton, twin, triplet or more), gestational age (<37 wks, >=37
wks), birth weight (<2500 g, >=2500 g), parental age at the time of delivery (<25 yrs,
of urbanization (1 (most urbanized) to 4 (least urbanized)), was retrieved from TBR.
Urbanization for the living (birth) area of each study subject was grouped according to the National Statistics of Regional Standard Classification.
22The TBR also provided information on socioeconomic factors including marital status of parents (marriage, otherwise), parental educational levels (elementary or less, junior high, senior high, college or higher) and difference of parental ages (0-5 yrs, 6-10 yrs, >10 yrs).
Statistical analysis
We first compared the distributions of the selected socio-demographic variables between cases and controls. A logistic regression model was employed to calculate the crude odds ratio (COR) and adjusted odds ratio (AOR) and their corresponding 95%
confidence intervals (CIs) of SIDS in relation to the selected socio-demographic variables.
We further performed log-linear model to assess the associations of daily
maximum temperature (DMT) with rate of SIDS. The model, with the natural logarithm of rates of SIDS as the dependent variable, was used to fit grouped data from 11
temperature categories determined by percentiles, i.e., <5
th, 5
th-<15
th, 15
th-<25
th, …..85
th-
<95
th, and >=95
th. The rate of SIDS for each temperature category was calculated as
follows. First, we averaged the DMT recorded by all 46 MMS to come up with the
DMT of Taiwan between 1994 and 2003. The DMT of Taiwan was further categorized into percentiles using 45
thto <55
thpercentiles (i.e., 21.9
oC to 23.3
oC) as reference category. Second, the daily rate of SIDS was calculated as a ratio of number of SIDS to the number of live birth in that particular year. Third, we averaged the daily rate of SIDS for all days in the same DMT category to represent the rate of SIDS for that particular temperature category. The rate ratio (RR) of SIDS was estimated using the formula e
m, where m is the regression coefficient. All statistical analyses were
performed with SAS (version 9.1; SAS Institute, Cary, NC). A P value <0.05 was considered statistically significant.
Results
The annual number of live births gradually decreased from 322,938 in 1994 to 227,070 in 2003. The annual rate of SIDS was 58.5/10
5in 1994, and thereafter
increased to 63.8/10
5in 1996; it was then steadily downward to 52.7/10
5. The average annual rate of SIDS over the study period was estimated at 57.9/10
5.
Table 1 shows that distributions of most selected birth characteristics for cases and
controls were significantly different. Cases tended to be births with higher birth-order
weight (LBW, <2500 g) births were higher in cases than in controls. While there was no significant difference in maternal age between the groups, cases’ fathers were relatively young. In addition, significantly more cases than controls were delivered outside
hospitals or clinics; and 32.77% of cases lived in the most urbanized areas of Taiwan, compared to only 29.48% of controls living in such urban areas.
Socioeconomic status of cases and controls are presented in Table 1. Significantly less cases’ parents were married (95.92%), as compared with controls’ parents
(97.65%). Cases’ parents tended to be less educated. Fathers of some 52% cases and 48% controls had an educational level of junior high or less, while the corresponding figures for cases’ mothers were 48% and 43%. Additionally, the difference in paternal ages was significantly higher in cases than in controls.
Table 1 shows odd ratio of SIDS in relation to birth characteristics and
socioeconomic factors. Male were at an increased risk as compared to females [AOR,
1.19; 95% CI-1.06, 1.33]. The higher birth order also had greater risks [second birth,
AOR, 1.39; 95% CI-1.21, 1.58; > =3 birth, AOR, 1.62, 95% CI-1.37, 1.92]. Infants with
a shorter gestation age [AOR, 1.69; 95% CI-1.33, 2.13] or LBW [AOR, 2.87; 95% CI-
2.30, 3.59] were both at elevated risks. While maternal age at delivery was not to be
associated with the risk of SIDS, younger fathers (< 25 years) was associated with a
significantly increased risk of SIDS [AOR, 1.37; 95% CI-1.09, 1.71]. There was also geographic variation in risk of SIDS. Infants living in the most urbanized areas had the most increased risk SIDS [AOR, 1.46; 95% CI-1.20, 1.78]. With respect to the influence of socioeconomic factors on SIDS, we noted that compared to infants with higher educated fathers the subjects with a paternal educational level of elementary and less had a significantly increased risk of SIDS [AOR, 1.28; 95% CI-1.01, 1.64]. We also noted that age difference of parental ages greater than 10 years was associated with an significantly increased risk of SIDS [AOR, 1.28; 95% CI-1.01, 1.64].
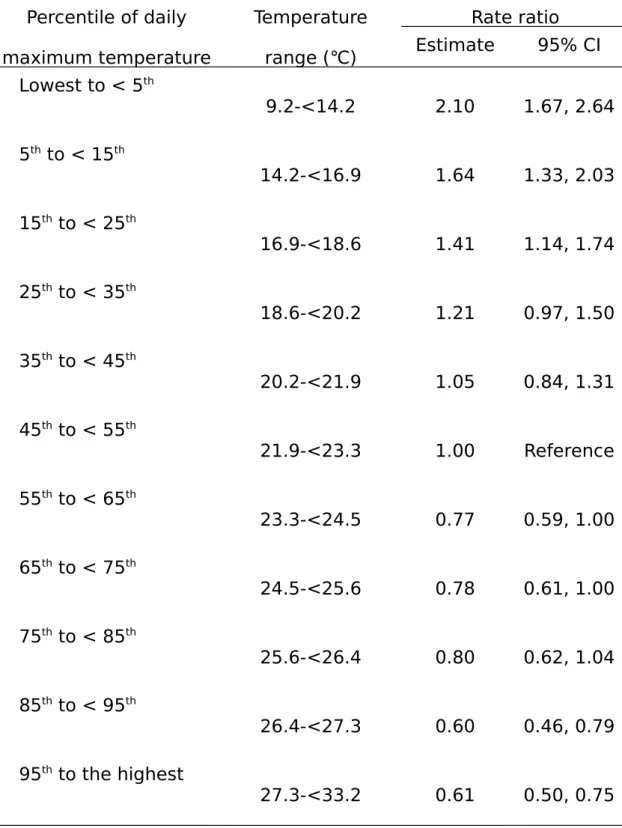
Risk of SIDS in relation to the averaged daily maximum temperature was shown in
Table 2. The DMT at the 5
thpercentile or lower (9.2 to <14.2℃) was associated with
the most elevated RR of SIDS [RR=2.10; 95% CI-1.67, 2.64]. The RR declined, but
was still significant, to 1.64 [95% CI-1.33, 2.03] and 1.41 [95% CI-1.14, 1.74] for a
DMT of 14.2 to <16.9℃ and 16.9 to <18.6℃, respectively. On the other hand, a higher
DMT posed favorable influences on the risk of SIDS. Compared to the reference DMT,
DMTs at 85
thto <95
thpercentile (i.e., 26.4-<27.3℃) and at the 95
thpercentile or higher
(i.e., 27.3-<33.2℃) were both associated with a significantly reduced RR, with a figure
of 0.60 [95% CI-0.46, 0.79] and 0.61 [95% CI-0.50, 0.75], respectively.
Discussion
The SIDS is currently defined as "the sudden unexpected death of an infant less
than 1 year of age with onset of the fatal episode apparently occurring during sleep, that
remains unexplained after thorough investigations. SIDS, whose etiology remains rather
vague, is still the major cause of death among infants between 1 month and 1 year of
age in industrialized countries with varying incidences in different populations.
23We
found that the incidence rate of SIDS in Taiwan is quite stable (52.763.8 per 10
5live
births) from 1994 to 2003, but such a incidence rate was lower than the figures reported
from most Western studies (around 100 per 10
5live births), and the socio-demographic
risk factors for SIDS identified from our analysis were essentially the same as findings
from previous studies.
2-7, 14-17However, unlike data from most Western nations showing
that the SIDS incidence rate has been dropped more than 50% over the past decade,
3the
incidence rate of SIDS declined only slightly during the study period in Taiwan (58.5 to
52.7 per 10
5live births). The following three reasons might account for such mild
decline. First, the rate of autopsy and scene investigation among deceased infants of
Taiwan was not possible due to culture reasons unless the possibility of non-natural
infant death cannot be ruled out.
16, 17This was particularly true in earlier years than in
recent years. Thus, the incidence rate of SIDS noted in earlier years could be
underestimated due to under-diagnosis of SIDS. Second, information on UCOD analyzed in this study was entirely retrieved from death certificate. Previous studies have indicated that some cases of accidental asphyxia or suffocation may be
misdiagnosed or misclassified as SIDS, so that the true incidence rate of SIDS may be
falsely elevated.
16, 21Whether more cases of accidental asphyxia or suffocation were misclassified as SIDS in recent years in Taiwan needs to be further clarified. Third, the campaign of safe sleeping position from prone to supine has significantly dropped the incidence of SIDS in many Western countries in recent years.
3, 5, 7, 11-13However, there was no such campaign had been advocated in Taiwan, which might also be responsible, at least to some extent, for the difference in declining trend of SIDS incidence between Taiwan and Western nations.
Previous studies frequently reported that male infants, shorter gestational week, lower birth weight, and higher birth order were the risk factors for SIDS.
2-7, 19With such a large sample size, our study also provided empirical support for the previous findings.
Prior studies noted that the SIDS has a distinctive characteristic male fraction of
approximately 0.61 for all postneonatal SIDS in the US.
24In the study by Wu et al., the
male dominance in sudden death was even noted in all age groups by age of 18.
19Future
studies will be required to elucidate the respiratory or cardiac mechanisms underlying
the male predominance for SIDS. Little is known regarding the influence of birth order on risk of SIDS. Blake took a critical stance highlighting the dilution of family
resources allocated to children. Blake argued that the first-born usually got much of the parental resources and attentions.
25As more children were born to the families, parents might have lesser resources and attentions personally devoted to children, which might be associated with an increased risk of SIDS for those high birth order infants who usually receive lesser attention and care provided by their parents.
As for the socioeconomic factors, previous studies suggested that teen mothers and less educated mother were at high risks of SIDS,
3, 5, 7mainly because young mothers and the those with lower educational level are likely to have inadequate baby care skills and information concerning baby caring. Our study did note significant associations of SIDS with maternal age and maternal educational level in bivariate analyses. However, such associations disappeared in the multivariate analysis in which many socio-demographic factors, including paternal age and educational level, were considered simultaneously.
Maternal age <25 years was significantly associated with an increased risk of SIDS
[OR=1.15; 95% CI-1.10, 1.31], but such elevated risk reduced and become insignificant
in the multivariate model [OR=1.09, 95% CI-0.92, 1.30]. Because the correlation
between maternal age and paternal age was considered high (Spearman’s rho=0.61), the
insignificant results associated with maternal age <25 years could be due to
simultaneous inclusion of paternal age in the model. As we simultaneously included paternal and maternal ages in the multivariate regression model, a potential numerical problem concerned collinearity between covariates, rendering estimated regression coefficients invalid. To investigate this potential statistical problem, we examined the size of standard error (SE) of regression coefficient, and found that collinearity should not be a concern in our data as the multivariate logistic regression model that included both variables had no large estimated slope coefficients or estimated SEs.
26On the other hand, maternal education level was not significantly associated with risk of SIDS in both simple and multivariate logistic regression models. Previous studies indicated that paternal education may predict, independent of maternal education, certain birth outcomes including preterm birth and lower birth weight.
27-29A higher level of paternal education may be associated with higher family income and better nutrition of children, which may lead to improvement in infant’s birth outcome and health. Further
exploration of the paternal role in the causation of SIDS would facilitate more specific interpretations on the independent effect of paternal education on SIDS.
In addition to individuals’ socio-demographic characteristics, our study also
disclosed a geographic variation of SIDS in Taiwan, which was also found in prior
studies. In an earlier study, Knobel et al. noted a geographic variation in infant mortality rate in Taiwan, but they argued that such urban and rural-difference in infant mortality rate could be due to under-registration of infant death in rural areas, which was also empirically supported by a subsequent study by Chen et al.
30The higher SIDS in urban areas was also previously reported in an earlier study by Spiers and Schlesselman.
31Whether the urban-rural difference in SIDS noted in our study was artificially
attributable to the under-registration of infant deaths in rural areas needs to be further investigated.
Apart from the above artifact, a range of environmental factors may interact to contribute to the adverse health conditions conducive to SIDS. Among them,
temperature and air pollutants were the two factors frequently considered to be related
to risk of SIDS.
16, 32Knöbel et al examined the possible roles of weather and air
pollution in SIDS and suffocation, and found that mortality from SIDS per 1000 live
births was 3.3 times greater in the lowest category of visibility on the day of death than
in the highest category; this rate ratio was 3.4 for the average visibility during the 9 days
before death. Adjustment for various potential clinical and meteorological confounders
increased these rate ratios to 3.8 and 5.1, respectively.
16A literature review conducted
by Tong and Colditz concluded that although the body of evidence appears to suggest
that air pollution (especially particles and some gaseous pollutants) may play a certain role in the occurrence of SIDS, the available evidence is inadequate to come to any conclusion about a causal relationship between air pollution and SIDS.
32The potential relationship between urbanization and risk of SIDS noted in our study might be explained by an increased prevalence of respiratory causes, that are also positively associated with air pollution, in urban areas.
33In addition to air pollution, infections may also be involved in some cases of SIDS suggested by numerous previous studies.
Bentham explored the influences of population mixing on SIDS in England and Wales, and concluded that geographical variations in SIDS deaths in England and Wales are strongly associated with differences in rates of population mixing as measured by migration. The findings support the hypothesis that population mixing, by its influence on exposure to infections, may be a significant factor in the aetiology of SIDS.
34A previous Taiwanese study also noted a positive relationship between urbanization and population migration.
35Due to unavailability of data on air pollutants and population mixing, we were unable to draw specific conclusion concerning the roles of these environmental factors in causing the urban-rural difference in SIDS.
Low temperature has been consistently found to be associated with SIDS
mortality.
15, 16, 19, 36However, with a very few exception,
36most of the previous studies
were conducted in nations located in high latitude. Knöbel et al. investigated the influence of Taiwan’s temperature on risk of SIDS, and found that an averaged daily temperature in a range of of 7-13℃ may increase the risk of SIDS by 3.3 times,
16which was somewhat higher than the risk estimate noted in our study. Leiss and Suchindran used 25 percentile as the cutoff point to associate low temperature with SIDS in North Carolina, and found that temperature lower than 12℃ was associated with an increased risk of SIDS by 1.8 times.
15When the daily temperature was decreased to 9.2℃
(Lowest to < 5
th%), the relative risk estimate increased to 2.1. These results suggested the influences of low temperature on SIDS mortality can persist even in a country where there is no appreciable seasonal variation of temperature like Taiwan. Mage conducted an analysis in Hawaii, which has a semi-tropical climate with warm temperature throughout the year, and still noted peak SIDS mortality in winter.
35Our study had several strengths. First, this study was population-based, and
employed a nationally representative sample, which leads to little room for selection
bias. Additionally, information analyzed in this study was registry-based and didn’t rely
on recall, which also reduce the likelihood of information bias. Second, the statistical
power was more than enough given a very large sample size of subjects with SIDS
being included in the analysis. Third, we simultaneously analyzed the influences of
socio-demographic, geographic, and meteorological factors associated with SIDS mortality, which provided comprehensive understanding of the roles of these factors in the causation of SIDS.
The major limitation of the study is the information of infant residential and sleeping environment is unable to retrieve, such as parents’ smoking and bottle or breast feeding behavior, sleeping position, co-sleeping, indoor temperature and clothes
thickness, etc., are all unable to be controlled and analyzed in the study. Taking all 46 MMS around Taiwan as a whole may not be able to accurately reflect the real
temperature experiences by each study subject in this study. Unfortunately, there is no detail address registered in both the Birth and Death registries. Specifically, we know only the city/county (totally 20 cities/counties in Taiwan) where the study subject lived.
It would not be correct to simply use the temperature data of MMSs located in a certain city/county to represent the exposure of all study subjects living in the same city/county.
A study subject’s residence could be closer to an MMS in the neighboring city/county than to an MMS located in the same city/county as his/her household. Because of the limited information on each study subject’s residential address, we tend to retain our analysis that considered Taiwan as a whole.
In conclusion, this analysis indicated that socio-demographic, geographic and
meteorological data can be used to identify families in greater need of early guidance and to promote various prevention measures to avoid the occurrence of SIDS.
Additionally, future studies should further analyze the impact of other risk factors for SIDS mortality such as parents’ behavior and living environment, etc., in order to obtain an even more complete profile of risk factors for SIDS mortality in Taiwan.
Acknowledgements
This study was supported by a grant from National Scientific Council, Taiwan
(NSC98-2314-B-227-001-MY2).
References
1. Krous HF, Beckwith JB, Byard RW et al. Sudden infant death syndrome and unclassified sudden infant deaths: A definitional and diagnostic approach.
Pediatrics 2004; 114: 234-238.
2. Knöbel HH, Yang WS, Ho MS. Urban-rural and regional differences in infant
mortality in Taiwan. Soc. Sci. Med. 1994; 39: 815-822.
3. Sullivan FM, Barlow SM. Review of risk factors for sudden infant death syndrome.
Paediatr. Perinat. Epidemiol. 2001; 15: 144-200.
4. Thompson JMD, Mitchell EA. Are the risk factors for SIDS different for preterm
and term infants? Arch. Dis. Child. 2006; 91: 107-111.
5. Ostfeld BM, Perl H, Esposito L et al. Sleep environment, positional, lifestyle, and demographic characteristics associated with bed sharing in sudden infant death
syndrome cases: A population-based study. Pediatrics 2006; 118: 2051-2059.
6. Malloy MH. Size for gestational age at birth: impact on risk for sudden infant death and other causes of death, USA 2002. Arch. Dis. Child. Fetal. Neonatal. Ed. 2007;
92: F473-F478.
7. Fu LY, Colson ER, Corwin MJ, Moon RY. Infant sleep location: associated
maternal and infant characteristics with sudden infant death syndrome prevention
recommendations. J. Pediatr. 2008; 153: 503-508.
8. McMartin KI, Platt MS, Hackman R et al. Lung tissue concentrations of nicotine in
sudden infant death syndrome (SIDS). J. Pediatr. 2002; 140: 205-209.
9. Cutz E, Perrin DG, Hackman R, Czegledy-Nagy EN. Maternal smoking and
pulmonary neuroendocrine cells in sudden infant death syndrome. Pediatrics 1996;
98: 668-672.
10. Glinianaia SV, Rankin J, Bell R, Pless-Mulloli T, Howel D. Does particulate air pollution contribute to infant death? A systematic review. Environ. Health Perspect.
2004; 112: 1365-1370.
11. Colson ER, Rybin D, Smith LA, Colton T, Lister G, Corwin MJ. Trends and factors associated with infant sleeping position: The national infant sleep position study,
1993-2007. Arch. Pediatr. Adolesc. Med. 2009; 163: 1122-1128.
12. Vennemann MM, Bajanowski T, Brinkmann B et al. Sleep environment risk factors for sudden infant death syndrome: The German sudden infant death syndrome
study. Pediatrics 2009; 123: 1162-1170.
13. Mitchell EA, Thach BT, Thompson JM, Williams S. Changing infants’ sleep position increases risk of sudden infant death syndrome. Arch. Pediatr. Adolesc.
Med. 1999; 153: 1136-1141.
14. Schluter PJ, Ford RP, Brown J, Ruan AP. Weather temperatures and sudden infant
death syndrome: a regional study over 22 years in New Zealand. J. Epidemiol.
Community. Health. 1998; 52: 27-33.
15. Leiss JK, Suchindran CM. Sudden infant death syndrome and local meteorologic
temperature in North Carolina. Am. J. Epidemiol. 1996; 144: 111-115.
16. Knöbel HH, Chen CJ, Liang KY. Sudden infant death syndrome in relation to weather and optimetrically measured air pollution in Taiwan. Pediatrics 1995; 96:
1106-1110.
17. Knöbel HH, Yang WS, Chen CJ. Risk factors of sudden infant death in Chinese
babies. Am. J. Epidemiol. 1996; 144: 1070-1073.
18. Lee CL, Chung TL. The trend of sudden infant death syndrome in Taiwan from
1984 to 1993. Acta. Paediatrica. Sinica. 1995; 36: 431-433.
19. Wu MH, Chen HC, Wang JK, Chiu HH, Huang SC, Huang SK. Population-based
study of pediatric sudden death in Taiwan. J Pediatr. 2009; 155: 870-874.
20. Lin CM, Lee PC, Teng SW, Lu TH, Mao IF, Li CY. Validation of the Taiwan birth
registry using obstetric records. J. Formos. Med. Assoc. 2004; 103: 297-301.
21. Lu TH, Lee MC, Chou MC. Accuracy of cause-of-death coding in Taiwan: Types of
miscoding and effects on mortality statistics. Int. J. Epidemiol. 2000; 29: 336-343.
22. National Statistics of Regional Standard Classification Data. Directorate-General Budget, Accounting and Statistics. 1993; Taipei, Taiwan.
23. Courts C, Madea B. Genetics of the sudden infant death syndrome. Forensic. Sci.
24. Mage DT, Donner M. A unifying theory for SIDS. Int. J. Pediatr. 2009; 2009:
368270.
25. Blake J. Family size and the quality of children. Demography 1981; 18: 421-442.
26. Hosmer DW, Lemeshow S. Applied logistic regression. 1989; New York: John
Wiley.
27. Chen CW, Tsai CY, Sung FC et al. Adverse birth outcomes among pregnancies of teen mothers: age-specific analysis of national data in Taiwan. Child. Care. Health.
Dev. 2010; 36: 232-240.
28. Parker JD, Schoendorf KC, Kiely JL. Associations between measures of
socioeconomic status and low birth weight, small for gestational age, and premature
delivery in the United States. Ann. Epidemiol. 1994; 4: 271–278.
29. Jacobsen G, Schei B, Hoffman HJ. Psychosocial factors and small-for-gestational- age infants among parous Scandinavian women. Acta. Obstet. Gynecol. Scand.
1997; 165(Suppl.): 14–18.
30. Chen LM, Sun CA, Wu DM, Shen MH, Lee WC. Underregistration of neonatal deaths: An empirical study of the accuracy of infantile vital statistics in Taiwan. J.
Epidemiol. Community. Health. 1998; 52: 289-292.
31. Spiers PS, Schlesselman JJ, Wright SG. Sudden infant death syndrome in the United
States: a study of geographic and other variables. Am. J. Epidemiol. 1974; 100: 380-
389.
32. Tong S, Colditz P. Air pollution and sudden infant death syndrome: a literature
review. Paediatr. Perinat. Epidemiol. 2004; 18: 327-335.
33. Marchal F. Sudden infant death syndrome: respiratory causes. Bull. Eur.
Physiopathol. Respir. 1983; 19: 411-421.
34. Bentham G. Population mixing and sudden infant death syndrome in England and
Wales. Int. J. Epidemiol. 1994; 23: 540-544.
35. Li CY, Lin RS, Lin CH. Urbanization and childhood leukemia in Taiwan. Int. J.
Epidemiol. 1998; 27: 587-591.
36. Mage DT. Seasonal variation of sudden infant death syndrome in Hawaii. J.
Epidemiol. Community. Health. 2004; 58: 912-916.
Variables Case (n=1,671)
Control (n=8,355)
p Crude estimate Adjusted
estimate
n % n % COR
c
95% CI
cAOR 95% CI
Birth characteristics Sex
0.00 2 Female
729 43.6
3
3997 47.8
4
1.00 1.00
Male
942 56.3
7
4358 52.1
6
1.19 1.07,
1.33
1.19 1.06,
1.33 Birth order
<0.0
1
601 35.9
9
3581 42.9
1
1.00 1.00
2
661 39.5
2
3078 36.8
8
1.28 1.13,
1.44
1.39 1.21,
1.58
>=3
409 24.4
9
1687 20.2
1
1.45 1.26,
1.66
1.62 1.37,
1.92 No. of birth
<0.0 01 Singleton
1602 96.0
4
8209 98.2
5
1.00 1.00
3.12 1.63 Gestational age (wks)
<0.0 01
<37
242 14.6
9
398 4.80 3.42 2.88,
4.05
1.69 1.33, 2.13
>=37
1405 85.3
1
7890 95.2
0
1.00 1.00
¯x±SD
38.29±2.92 39.15±1.66 <0.0 01 Birth weight (g)
<0.0
>=2500
1367 83.3
6
7895 95.0
7
1.00 1.00
<2500
278 16.6
4
412 4.93 3.90 3.31,
4.59
2.87 2.30,
3.59
¯x±SD
2962.18±656 .94
3206.01±47 1.33
<0.0 01 Maternal age at
delivery (yrs) 0.09
3
<25
475 28.7
0
2169 26.2
4
1.15 1.01,
1.31
1.09 0.92,
1.30
7 0
>=30
497 30.0
3
2501 30.2
6
1.05 0.92,
1.19
0.89 0.76,
1.03
¯x±SD
27.1±4.9 27.4±4.5 0.01
7 Paternal age at delivery
(yrs) <0.0
01
<25
164 10.2
1
700 8.55 1.34 1.10,
1.63
1.37 1.09,
1.71 25-29
510 31.7 2916 35.6 1.00 1.00
>=30
932 58.0
3
4571 55.8
3
1.17 1.04,
1.31
1.06 0.90,
1.24
¯x±SD
30.7±5.2 30.5±4.7 0.22
8 Place of birth
0.01 3 Hospital/Clinics
1578 94.5
5
7968 95.6
3
1.00 1.00
Midwife
center/Home 91 5.45 364 4.37 1.26 1.00,
1.60
1.09 0.80,
1.48
1 4 (least urbanized)
212 12.9
3
1200 15.9
2
1.00 1.00
3
474 28.9
2
2387 29.2
3
1.22 1.02,
1.45
1.21 1.00,
1.46 2
416 25.3
8
2072 25.3
7
1.23 1.03,
1.47
1.22 1.00,
1.50 1 (most urbanized)
637 32.7
7
2408 29.4
8
1.50 1.26, 1.78
1.46 1.20,
1.78
Marital status of
parents <0.0
01 Married
1600 95.9
2
8101 97.6
5
1.00 1.00
Otherwise
68 4.08 195 2.35 1.77 1.33,
2.34
0.93 0.51,
1.71 Maternal education
0.00 6 College or higher
215 13.0
1
1086 13.1
6
1.00 1.00
8 1 1.08 1.03 Junior high
661 39.9
9
2980 36.1
1
1.12 0.95,
1.33
0.90 0.73,
1.12 Elementary or less
202 12.2
2
992 12.0
2
1.03 0.83,
1.27
0.74 0.57,
1.00 Paternal education
<0.0 01 College or higher
315 19.5
9
1696 20.6
6
1.00 1.00
Senior high
522 32.4 2956 36.0 0.95 0.82, 0.99 0.83,
Junior high
578 35.8
9
2842 34.6
2
1.10 0.94,
1.27
1.07 0.88,
1.29 Elementary or less
195 12.1
1
715 8.71 1.47 1.20,
1.79
1.28 1.01,
1.64 Age difference of
parental ages (yrs) <0.0
01 0-5
1233 77.1
6
6561 80.6
0
1.00 1.00
6-10
291 18.2
1
1367 16.8
0
1.13 0.98,
1.30
1.13 0.96,
1.34
2.44 2.39
COR=crude odds ratio; CI=confidence interval; AOR=adjusted odds ratio
a
Inconsistency between total sample size and the sample size summed for individual variables was due to missing information
b
Based on
2test or Student t-test
c