DOI 10.1378/chest.06-1829
2007;131;705-710
Chest
Guo and Fung-Chang Sung
Yu-Chun Wang, Jia-Ming Lin, Chung-Yi Li, Long-Teng Lee, Yue-Liang
*
Obstruction
Prevalence and Risks of Chronic Airway
http://www.chestjournal.org/content/131/3/705.full.html
and services can be found online on the World Wide Web at:
The online version of this article, along with updated information
) ISSN:0012-3692
http://www.chestjournal.org/site/misc/reprints.xhtml
(
of the copyright holder.
may be reproduced or distributed without the prior written permission
Northbrook IL 60062. All rights reserved. No part of this article or PDF
by the American College of Chest Physicians, 3300 Dundee Road,
2007
Physicians. It has been published monthly since 1935. Copyright
CHEST is the official journal of the American College of Chest
Prevalence and Risks of Chronic Airway
Obstruction*
A Population Cohort Study in Taiwan
Yu-Chun Wang, MPH; Jia-Ming Lin, PhD; Chung-Yi Li, PhD; Long-Teng Lee, PhD;
Yue-Liang Guo, MD, PhD; and Fung-Chang Sung, PhD, MDH
Background: This study investigated the prevalence, incidence, and hospitalization for chronic
airway obstruction (CAO) in a population cohort.
Methods: Medical reimbursement claims from 1996 to 2002 based on a 1996 insured cohort of
167,372 persons from National Health Insurance, Taiwan, were used. We presented the
chronological trends of CAO (International Classification of Diseases, Ninth Revision code 496)
and the relationships between the CAO severity and age, sex, urbanization, and hospitalization
and comorbidity for the population > 40 years old.
Results: The overall average annual prevalence and incidence rates were 2.48/100 and 0.66/100,
respectively, for the population, among 4,568 patients with CAO cared during the study period.
For the population aged > 70 years, the prevalence rates had a peak of 8.83/100 in 1998 and
afterward remained a plateau until 2002. The corresponding incidence decreased from 2.48/100
to 1.62/100, and the hospitalization rate for them had a peak of 2.22/100 in 1999. The multivariate
logistic regression analysis showed that the risk of hospitalization for CAO was higher for patients
with the comorbidity of renal failure, coronary artery disease, and pneumonia and influenza, but
lower with skin and joint disorders.
Conclusions: The national insurance program promotes patient care and provides a proper
pathway for surveillance and identification of CAO.
(CHEST 2007; 131:705–710)
Key words: chronic airway obstructions; comorbidity; population cohort; universal health insurance
Abbreviations: BPH⫽ benign prostate hypertrophy; CAD ⫽ coronary artery disease; CAO ⫽ chronic airway
obstruc-tion; CI⫽ confidence interval; CPD ⫽ chronic pulmonary disease; ICD-9 ⫽ International Classification of Diseases, Ninth Revision; NHI⫽ National Health Insurance; OR ⫽ odds ratio; P&I ⫽ pneumonia and influenza
C
hronic pulmonary diseases (CPDs) are
hetero-geneous disorders of acute/chronic bronchitis,
emphysema, asthma, COPD, and chronic airway
obstruction (CAO) not elsewhere classified. These
diseases have attracted increasing attention because
of the augmentation of prevalence and mortality, and
the economic cost worldwide.
1Community-based
investigation has been considered as a reliable
ap-proach for the occurrence estimation of and etiologic
studies for these diseases, although the results may
*From the Institute of Environmental Health (Drs. Wang and Lin), National Taiwan University College of Public Health, Taipai; Department of Public Health (Dr. Li), Fu Jen Catholic University College of Medicine, Taipei Hsien; Department of Family Medicine (Dr. Lee), National Taiwan University Hospital, Taipai; Environmental and Occupational Medicine (Dr. Guo), National Taiwan University Medical Center, Taipai; and Institute of Environmental Health (Dr. Sung), China Medical University College of Public Health, Taichung, Taiwan.
This work was performed at Institute of Environmental Health, National Taiwan University College of Public Health. Dr. Wang and Dr. Sung contributed equally to this article.
This study was supported in part by Executive Yuan Department of Health grant DOH94-DC-1107, 2005, and National Science Council grant EPA-Z-039 – 001, 2005.
All authors have no conflicts of interest to disclose.
Manuscript received July 24, 2006; revision accepted November 1, 2006.
Reproduction of this article is prohibited without written permission from the American College of Chest Physicians (www.chestjournal. org/misc/reprints.shtml).
Correspondence to: Fung-Chang Sung, PhD, MPH, Professor and Dean, Institute of Environmental Health, China Medical Univer-sity College of Public Health, 91 Hsueh-Shih Rd, Taichung 404, Taiwan; e-mail: [email protected]
DOI: 10.1378/chest.06-1829
Original Research
differ from diagnosis criteria.
2,3Population-based
insurance data provide an opportunity to observe the
epidemiologic patterns of the diseases and factors
associated with the patterns.
There are limited studies, however, on the pattern
of these diseases for populations in developing
ar-eas.
4It has been inspirited to investigate the
effec-tive prevention strategy, including the reduction of
associated risk factors and comorbidities for these
diseases.
5–7Older age and comorbidities such as
hypertension and other cardiac conditions, diabetes
mellitus, and chronic renal failure have been
associ-ated with CPDs.
8 –16Smoking prevention, other risk
factors control, screening, and early treatment of the
diseases may reduce both the incidence and
preva-lence of the diseases.
17–19This study estimated annual trends of prevalence,
incidence, and hospitalization for CAO for the
pop-ulation
ⱖ 40 years old using cohort data generated
from population-based Taiwan National Health
In-surance (NHI) claims. Patient demographic
charac-teristics and comorbidity associated with the disease
severity were investigated as well.
Materials and Methods
A representative sample of 200,000 people was randomly selected from all beneficiaries ever enrolled in the Taiwan NHI program between March 1995 and December 2000. Among them, 167,372 were actively enrolled at the first day of 1996 and were considered as the study cohort in the current analysis. We linked, through individual’s personal identification number, to both the ambulatory care and inpatient claims (from 1996 to 2002) to identify all episodes of CAO encountered by the study subjects. Attrition of the study cohort was observed due to various reasons such as mortality and emigration. The NHI program had an initial coverage rate of 90.0% population in 1995, and later increased to 96.2% in 2000.20
The NHI electronic data files provided scrambled patient
identification numbers, gender, birthday, and the classification code of disease, dates of admission, and discharge, and medical institutions providing the services. The NHI has not yet required physicians to use the tenth revision of International Classification of Diseases, and cases of CPD were therefore coded using the International Classification of Diseases, Ninth Revision (ICD-9) clinical modification codes 490 – 496. The claims of CAO (ICD-9 code 496) not elsewhere classified were what we focused on in this study.
Patients who made only one ambulatory visit for the purpose of screening for CPD instead of receiving care for the disease during the study period (ie, 1996 to 2002) were not considered for this study. The incidence cases, however, were patients with the new claims for the care of the disease identified starting in 1997. Prevalence and hospitalization rates from 1996 to 2002 and incidence rates from 1997 to 2002 for population agedⱖ 40 years were estimated and adjusted by annual age-specific population. Average annual prevalence and incidence rates were calculated at intervals of 10 years (Table 1). Chronological trends of CAO were grouped for population aged 40 to 59 years, 60 to 69 years, and ⱖ 70 years due to very low prevalence in the younger groups and similar pattern in the oldest groups.
Except for lung function measurement,21,22exacerbations and
clinic visit frequency were adopted from methods developed by Donaldson et al23,24to determine the severity of CPDs. Based on
numbers of clinic visit, cases were categorized into four severity levels: level zero for patients with only 1 visit in the 7 years (⬍ 42.2 percentile), level one for 2 to 5 visits (42.2 to 81.1 percentiles), level two for 6 to 24 visits (81.1 to 95.0 percentiles), and level three forⱖ 25 visits (ⱖ 95 percentile). In addition to estimating trends of prevalence and incidence rates for the study population, we calculated annual visits-to-cases ratios to evaluate the impact of health insurance on CAO. These ratios were determined using annual total clinic visits divided by total cases claimed for CAO and categorized by the disease severity for comparison.
All cases of CAO identified during the entire study period were pooled for further analysis for the association between disease severity and associated comorbidities, controlling for sex, age, and urbanization level (Taipei, Kaohsiung, and Taichung vs the rest areas). We used the polytomous logistic regression (Proc catmod; SAS Institute; Cary, NC) to compute odds ratios (ORs) and confidence intervals (CIs) for factors associated with the disease severity. We further used multiple logistic regression to estimate the relationships between hospitalization and sex, age,
Table 1—Distribution of Patients Aged > 40 Years, Average Annual Rates of Primary Diagnosed CAOs, and
Male-to-Female Case Ratios by Age and Severity Level in Taiwan, 1996 –2002
Variables Cases in 7 Years, No. (%)* Male/Female Gender, No. Prevalence Rate, /100 Incidence Rate, /100 Age, yr 40–49 533 (11.7) 317/216 0.40 0.17 50–59 833 (18.2) 497/336 1.53 0.49 60–69 1,523 (33.3) 1,093/429 4.48 1.19 70–79 1,286 (28.2) 935/351 7.94 2.02 ⱖ 80 393 (8.6) 249/144 8.36 2.54 Severity† 0 1,847 (40.4) 1,148/698 1 1,624 (35.6) 1,088/536 2 832 (18.2) 639/193 3 265 (5.80) 216/49 All 4,568 (100) 3,091/1,476 2.48 0.66
*CAO cases were grouped based on the age of first clinic visit and primary diagnostic code.
urbanization level, occupation (blue collar, others, vs white collar), disease severity and comorbidities. The potential comor-bidities included in this study were hypertensive disease (ICD-9 codes 401.9 – 405.9), benign prostate hypertrophy (BPH) [ICD-9 code 600], diabetes mellitus (ICD-9 code 250), skin disorders (ICD-9 codes 690 –709), cervical and back disorders (ICD-9 codes 721–725), joint disorders (ICD-9 codes 710 –719.99), renal failure (ICD-9 codes 585–586), coronary artery disease (CAD) [ICD-9 codes 410 – 414.9], and pneumonia and influenza (P&I) [ICD-9 codes 480 – 487]. These were the most frequently diag-nosed diseases among the insurance claims. We used the stepwise analysis for model selection, and tested using the likelihood ratio test. Statistical analysis was performed using statistical software (SAS version 8.2; SAS Institute; Cary, NC).
Results
Case Characteristics
In this study cohort, there were 4,568 patients
with at least one claim of primary diagnosis as CAO
services during the 7-year study period for the
population aged
ⱖ 40 years (Table 1). The overall
average annual prevalence and incidence rates of
CAO were 2.48% and 0.66%, respectively, for these
age groups. There were more male than female
patients, with male-to-female ratios ranged from
1.47 to 2.66 by age. Cases claimed for CAO were in
patients predominantly aged 60 to 79 years (61.5%).
The pattern of severity revealed that 5.80% of CAO
patients had
ⱖ 25 clinic visits with primary diagnosis
of CAO in 7 years.
Among all (n
⫽ 745) hospitalized cases of CAO
during the study period, 64.4% of inpatients had only
CAO, and 32.0% (239 cases) were both CAO and
bronchitis/emphysema/asthma (data not shown).
These cases included 1,346 hospitalization events
with predominantly men (80.3%), rural residents
(54.7%), and those aged 60 to 79 years (70.4%).
Incidence, Prevalence, and Hospitalization Rates
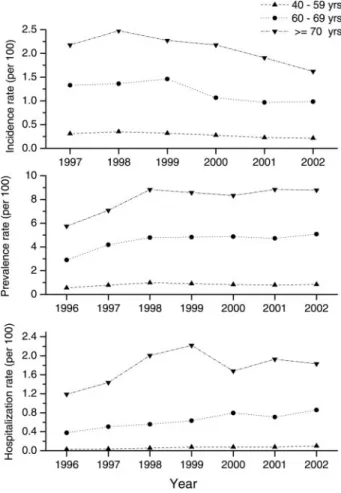
Figure 1 shows that the population aged
ⱖ 60
years dominated chronological changes in incidence,
prevalence, and hospitalization of CAO. The
preva-lence of ambulatory visits for the population aged
ⱖ 70 years increased from 5.75 per 100 in 1996 to
8.83 per 100 in 1998 and afterward became a plateau
until 8.79 per 100 in 2002. In the meanwhile, the
incidence among this age group decreased annually
to 1.62 per 100 in 2002, with an apparent peak rate
of 2.48 per 100 in 1998. The hospitalization rates of
CAO were also the highest in those aged
ⱖ 70 years
with a peak in 1999 (2.22 per 100) and declining to
1.83 per 100 in 2002.
Risk Factors Related to CAO
Table 2 shows the risks for the severity of CAO
associated with covariates estimated using
polyto-mous logistic regression analysis. The ORs of CAO
were higher for males, inpatients, and patients
re-ceiving care in 1996, and increased as the severity
increased. The OR of hospitalization for patients
with the level 3 severity of the disease was 8.01 (95%
CI, 5.92 to 10.8) higher than patients with the level
zero severity. Compared with population aged 40 to
49 years, the OR for the oldest group with the level
3 severity was 20.6 (95% CI, 2.77 to 152). Elevated
severity was also associated with several
comorbidi-ties, including BPH, CADS, and the highest with
P&I.
The estimated risk of hospitalization for CAO was
also evaluated and found to be greater for men and
patients identified in 1996 (Table 3). The OR
in-creased as age inin-creased, with the highest OR of 14.2
(95% CI, 6.54 to 30.9) for patients
ⱖ 80 years old.
The comorbidities of CAD, P&I, and renal failure
were significant factors to predict the hospitalization
of CAO. However, patients with comorbidities of
skin and joint disorders were at less risk for
hospi-talization.
Figure 1. Chronological trends in incidence, prevalence, and hospitalization rates of CAO in Taiwan from 1996 to 2002.
Discussion
The annual prevalence and incidence of CAO for
population in the study cohort increased to the peak
in 2 to 3 years after the launch of the universal health
insurance, and the incidence declined afterward. But
the prevalence of the disease was flattened on
approximately similar level after the peak year. A
study
25in the Netherlands for asthma prevalence in
children also reported a reversing trend in the period
of from 1996 to 2002, similar to our study period. In
this study, we believe the trends for CAO events are
not resulted from the disease coding because the
chronological incidence and prevalence of CAO
were opposite to the increasing trend of
cardiovas-cular diseases for the same time period in Taiwan
(data not shown).
CAO diseases are progressive diseases with mild
manifestation at the undiagnosed early stage.
26Both
the incidence and prevalence of the diseases may be
thus underestimated prior to the launch of the health
insurance. Because of the affordable NHI insurance
premium and low copayments for medical services,
the general population, particularly those with
med-ical indigence, were encouraged to seek services
during the earlier period of the insurance program.
Cheng and Chiang
27found that, compared with the
noninsured, the newly insured had more than twice
greater outpatient physician visits (0.48 vs 0.21,
p
⬍ 0.05) and hospital admissions (0.11 vs 0.04) in a
2-week period prior to the study interview. In this
study cohort, the increased CAO cases reaching the
peak level in 1998 are likely in response to the
increased physician visits in all levels of medical
facilities. The increased detections from 1997 to
1999 are likely the undiagnosed cases existing prior
to 1997.
Moreover, the impact from the epidemic of P&I in
Table 2—Disease Severity of CAO in Polytomous Logistic Regression Analysis
Variables
Severity Level§, OR (95% CI)
1 2 3 Male gender 0.97 (0.68–1.38) 1.63† (1.16–2.28) 1.81† (1.29–2.55) Hospitalization 2.41* (1.84–3.16) 4.08* (3.12–5.33) 8.01* (5.92–10.8) Clinic visit in 1996 1.96* (1.45–2.65) 3.55* (2.65–4.75) 4.50* (3.36–6.04) Urban 1.14 (0.89–1.46) 1.19 (0.94–1.51) 1.21 (0.95–1.55) Age, yr 40–49 1.0 1.0 1.0 50–59 9.14‡ (1.21–69.3) 12.5‡ (1.68–93.3) 14.9† (2.01–111) 60–69 10.4‡ (1.41–76.7) 18.4† (2.53–133) 24.3† (3.34–176) 70–79 12.3‡ (1.67–90.7) 23.6† (3.25–171) 33.3† (4.55–239) ⱖ 80 7.86‡ (1.04–59.2) 13.8‡ (1.87–102) 20.6† (2.77–152) Comorbidity Hypertensive disease 0.86 (0.64–1.16) 1.00 (0.75–1.34) 0.99 (0.74–1.32) BPH 1.29 (0.96–1.72) 1.27 (0.96–1.69) 1.49† (1.11–1.99) Diabetes mellitus 0.82 (0.61–1.10) 0.88 (0.66–1.17) 0.75 (0.56–1.01) Renal failure 1.00 (0.74–1.35) 0.76 (0.57–1.02) 0.84 (0.62–1.13) CAD 1.14 (0.87–1.50) 1.09 (0.84–1.42) 1.42† (1.09–1.86) P&I 1.25 (0.92–1.70) 1.51† (1.12–2.04) 1.91* (1.42–2.58) *p⬍ 0.0001. †pⱖ 0.0001 to ⱕ 0.01. ‡pⱖ 0.01 to ⬍ 0.05
§Based on clinic visit frequency in 7 years, zero for only 1 visit, one for 2 to 5 visits, two for 6 to 24 visits, and three forⱖ 25 visits.
Table 3—Hospitalization of CAO by Selected
Covariates in Multiple Logistic Regression, Stepwise Selection Variables OR 95% CI p Value Male gender 1.51 1.23–1.84 ⬍ 0.0001 Clinic visit in 1996 2.06 1.71–2.48 ⬍ 0.0001 Disease severity* 0 1.00 1 1.95 1.53–2.48 ⬍ 0.0001 2 3.39 2.64–4.34 ⬍ 0.0001 3 8.11 6.01–10.6 ⬍ 0.0001 Age, yr 40–49 1.00 50–59 2.72 1.20–6.15 0.0164 60–69 6.22 2.88–13.4 ⬍ 0.0001 70–79 8.18 3.81–17.6 ⬍ 0.0001 ⱖ 80 14.2 6.54–30.9 ⬍ 0.0001 Comorbidity Skin disorder 0.80 0.67–0.97 0.00215 Joint disorder 0.60 0.48–0.75 ⬍ 0.0001 Renal failure 1.36 1.12–1.65 0.0023 CAD 1.38 1.15–1.66 0.0005 P&I 1.87 1.52–2.29 ⬍ 0.0001
*Based on clinic visit frequency in 7 years: zero for only 1 visit, one for 2 to 5 visits, two for 6 to 24 visits, three forⱖ 25 visits.
the study period is another factor for consideration.
The temporal trend of mortality from P&I in Taiwan
also demonstrated an increasing trend in 1994 with a
peak in 1998 and flattening out afterward.
28A similar
pattern of CAO prevalence appeared in this study.
The influenza vaccination campaign has been
initi-ated since October 1998 for the elderly in Taiwan.
29Several studies
28 –30have evaluated the efficacy of
this flu prevention campaign in morbidity of and
mortality from CPDs, showing that the mortality
from CPDs significantly decreased after the
imple-mentation of vaccine program. However, there was
no benefit for hospitalization for CPD.
29,30Overall, age was found to be an important factor
associated with the severity of CPDs in this study.
The risk of CAO severity is the highest in the oldest
population. Hypertension, CAD, and P&I are the
most common comorbidities in patients receiving
health-care
attention
among
people
with
CPDs.
11,12,16These diseases are prevalent in the
elderly and are also associated with age,
comorbidi-ties, and other host health conditions. Incalzi et al
11found that renal failure may predict COPD
mortal-ity, but it was observed as a risk only for hospitalized
patients of CAO in this study. It is interesting to note
that the protective effect of skin and joint disorders
in reducing hospitalization for CAO may be due to
the activities acceptable for healthier patients. Rana
et al
14also reported COPD as a potential risk factor
for developing type 2 diabetes in women. In contrast,
diabetes mellitus was a weak protective comorbidity
associated with disease severity but with
hospitaliza-tion of CAO in the present study. The difference
could be resulted from the CAO patients being
predominant with men in this study. We believed the
risk for disease severities associated with covariates
were underestimated in this study because the
con-trol group was based on severity level zero, which
consisted of cases with one medical visit for CAO.
The above observed similarities and differences
are obviously important to note. Disease etiology and
care across nations and social status display a good
amount of variety deserving further study, along with
the gender difference in the comorbidity associated
with CPDs.
The primary limitation of this study is the nature
of disease diagnosis, which precludes the diagnosis
variation among physicians. In general, it is difficult
to clearly distinguish CAO or asthma from other
CPDs, not only about the terminology
31but also the
overlapped symptoms among diseases.
32Approxi-mately 69% of CAO patients in this cohort have
received medical attention for
bronchitis/emphy-sema and/or asthma as well (data not shown),
reflect-ing the coexistence of these diseases,
33,34introducing
the difficulty in taking apart the differences among
CPDs.
We were unable to analyze the patterns of asthma
separately because of the disease coding in this data
set. Claims of bronchitis, emphysema, and asthma
were grouped as one code by the Bureau of NHI
before 2000. Therefore, we evaluate the patterns of
CAO not elsewhere classified with consistent and
unique coding during 1996 –2002.
In conclusion, the chronological analysis for this
population-based cohort has demonstrated the
changing patterns of prevalence, incidence, and
hos-pitalization of CAOs for the population in Taiwan
after the launch of the nationwide health insurance.
The change reflects that this universal health
insur-ance plan provides convenient access to medical care
for the insured, particularly for the elderly. CAD and
P&I are comorbidities that not only can predict the
disease severity of CAO but also can enhance the
power of identifying the diseases.
ACKNOWLEDGMENT: We thank National Health Research Institutes for providing the insurance data and Dr. Sow-Hsing Kuo for his comments for this study. The interpretation and conclusions contained herein do not represent those of the Bureau of National Health Insurance, Department of Health, or National Health Research Institutes.
References
1 Burney P, Suissa S, Soriano JB, et al. The pharmacoepidemi-ology of COPD: recent advances and methodological discus-sion. Eur Respir J Suppl 2003; 43:1s– 44s
2 Viegi G, Pedreschi M, Pistelli F, et al. Prevalence of airways obstruction in a general population: European Respiratory Society vs American Thoracic Society definition. Chest 2000; 117:339S–345S
3 Plan and operation of Third National Health and Nutrition Examination Survey, 1988 –94: Series 1; Programs and col-lection procedures. Vital Health Stat 1 1994; 32:1– 407 4 McKnight J, Scott A, Menzies D, et al. A cohort study showed
that health insurance databases were accurate to distinguish chronic obstructive pulmonary disease from asthma and classify disease severity. J Clin Epidemiol 2005; 58:206 –208 5 Cerveri I, Accordini S, Verlato G, et al. Variations in the prevalence across countries of chronic bronchitis and smoking habits in young adults. Eur Respir J 2001; 18:85–92 6 Mannino DM, Gagnon RC, Petty TL, et al. Obstructive lung
disease and low lung function in adults in the United States: data from the National Health and Nutrition Examination Survey, 1988 –1994. Arch Intern Med 2000; 160:1683–1689 7 Zielinski J, Bednarek M. Early detection of COPD in a
high-risk population using spirometric screening. Chest 2001; 119:731–736
8 Burrows B, Earle RH. Prediction of survival in patients with chronic airway obstruction. Am Rev Respir Dis 1969; 99:865– 871
9 Connors AF Jr, Dawson NV, Thomas C, et al. Outcomes following acute exacerbation of severe chronic obstructive lung disease: the SUPPORT investigators (Study to Under-stand Prognoses and Preferences for Outcomes and Risks of Treatments). Am J Respir Crit Care Med 1996; 154:959 –967
10 Fuso L, Incalzi RA, Pistelli R, et al. Predicting mortality of patients hospitalized for acutely exacerbated chronic obstruc-tive pulmonary disease. Am J Med 1995; 98:272–277 11 Incalzi R, Fuso L, De Rosa M, et al. Co-morbidity contributes
to predict mortality of patients with chronic obstructive pulmonary disease. Eur Respir J 1997; 10:2794 –2800 12 Traver GA, Cline MG, Burrows B. Predictors of mortality in
chronic obstructive pulmonary disease: a 15-year follow-up study. Am Rev Respir Dis 1979; 119:895–902
13 Anthonisen NR. Prognosis in chronic obstructive pulmonary disease: results from multicenter clinical trials. Am Rev Respir Dis 1989; 140:S95–S99
14 Rana JS, Mittleman MA, Sheikh J, et al. Chronic obstructive pulmonary disease, asthma, and risk of type 2 diabetes in women. Diabetes Care 2004; 27:2478 –2484
15 Fouad MN, Mayo CP, Funkhouser EM, et al. Comorbidity independently predicted death in older prostate cancer pa-tients, more of whom died with than from their disease. J Clin Epidemiol 2004; 57:721–729
16 Tan WC, Xiang X, Qiu D, et al. Epidemiology of respiratory viruses in patients hospitalized with near-fatal asthma, acute exacerbations of asthma, or chronic obstructive pulmonary disease. Am J Med 2003; 115:272–277
17 Summaries for patients. The effects of a smoking cessation program on long-term survival in smokers with mild lung disease. Ann Intern Med 2005; 142:I12
18 Schoening SK. Issues and challenges for managed care in the treatment of asthma and chronic obstructive pulmonary disease. Am J Manag Care 2004; 10:S158 –163
19 Weng HC. Impacts of a government-sponsored outpatient-based disease management program for patients with asthma: a preliminary analysis of national data from Taiwan. Dis Manag 2005; 8:48 –58
20 National Health Insurance Profile [in Chinese]: Taipei: Bu-reau of National Health Insurance, Department of Health, Executive Yuan, Republic of China, 2001
21 Lundback B, Gulsvik A, Albers M, et al. Epidemiological aspects and early detection of chronic obstructive airway diseases in the elderly. Eur Respir J Suppl 2003; 40:3s–9s 22 Mannino DM. Chronic obstructive pulmonary disease:
defi-nition and epidemiology. Respir Care 2003; 48:1185–1191; discussion 1191–1193
23 Donaldson GC, Seemungal TA, Bhowmik A, et al. Relation-ship between exacerbation frequency and lung function de-cline in chronic obstructive pulmonary disease. Thorax 2002; 57:847– 852
24 Donaldson GC, Seemungal TA, Patel IS, et al. Longitudinal changes in the nature, severity and frequency of COPD exacerbations. Eur Respir J 2003; 22:931–936
25 van Schayck CP, Smit HA. The prevalence of asthma in children: a reversing trend. Eur Respir J 2005; 26:647– 650 26 Rennard S, Decramer M, Calverley PM, et al. Impact of
COPD in North America and Europe in 2000: subjects’ perspective of Confronting COPD International Survey. Eur Respir J 2002; 20:799 – 805
27 Cheng SH, Chiang TL. The effect of universal health insur-ance on health care utilization in Taiwan: results from a natural experiment. JAMA 1997; 278:89 –93
28 Chen YH, Liou SH, Chou CC, et al. Influenza and pneumo-coccal vaccination of the elderly in Taiwan. Vaccine 2004; 22:2806 –2811
29 Wang CS, Wang ST, Lai CT, et al. Reducing major cause-specific hospitalization rates and shortening hospital stays after influenza vaccination. Clin Infect Dis 2004; 39:1604 – 1610
30 Wang ST, Lee LT, Chen LS, et al. Economic evaluation of vaccination against influenza in the elderly: an experience from a population-based influenza vaccination program in Taiwan. Vaccine 2005; 23:1973–1980
31 Bhattarai MD. Asthma mistaken for chronic obstructive pulmonary disease. Lancet 2003; 361:1914 –1915
32 Bleecker ER. Similarities and differences in asthma and COPD: the Dutch hypothesis. Chest 2004; 126:93S–95S; discussion 159S–161S
33 Hansen EF, Phanareth K, Laursen LC, et al. Reversible and irreversible airflow obstruction as predictor of overall mortal-ity in asthma and chronic obstructive pulmonary disease. Am J Respir Crit Care Med 1999; 159:1267–1271
34 Ulrik CS, Backer V. Nonreversible airflow obstruction in life-long nonsmokers with moderate to severe asthma. Eur Respir J 1999; 14:892– 896