Contrasting Effects of Aqueous Tissue Extracts from an Invasive Plant,
Bidens pilosa L. var. radiata, on the Performance of Its Sympatric Plant
Species
Hsiao-Mei Hsu(1) and Wen-Yuan Kao(1,2*)
1. Institute of Ecology and Evolutionary Biology, National Taiwan University, 1, Roosevelt Rd., Sec. 4, Taipei 106, Taiwan. 2. Department of Life Science, National Taiwan University, 1, Roosevelt Rd., Sec. 4, Taipei 106, Taiwan.
* Corresponding author. Tel: +886-2-33662511; Fax: +886-2-2367-3374; Email: wykao@ntu.edu.tw (Manuscript received 8 April 2009; accepted 16 June 2009)
ABSTRACT: Bidens pilosa L. var. radiata Sch. Bip., a common weed in lowland Taiwan, is listed as one of the twenty most noxious invasive plants in Taiwan. In this study, we examined the effect of aqueous extracts of leaves, stems and roots of the invasive plant on germination and growth of seedlings (estimated by measuring the elongation of hypocotyls and radicals) of the same species and two other sympatric species, B. bipinnata and Ageraturem conyzoides. The objective of this study was to understand whether the aqueous tissue extracts affected the performance of the target species and whether these effects varied among tissue types and among target species. We found that the germination percentage of seeds of B. bipinnata was significantly reduced by root and leaf extracts, that of B. pilosa var. radiata was also significantly reduced by the application of root extract, while that of A. conyzoides was not affected by any of the three tissue extracts. The application of stem and leaf extracts inhibited the elongation of radicals of B. pilosa var. radiata, consequently, the growth of seedlings of this species was decreased in these two treatments. Though the elongation of hypocotyls was stimulated by leaf extract, the overall growth of seedlings of B. bipinnata was not affected by any tissue extract. In contrast, all three extracts stimulated the elongation of hypocotyls and radicals of A. conyzoides, consequently, the overall growth of seedlings of this plant was promoted by all three extracts. These results revealed that aqueous extracts from tissue of B. pilosa var. radiata had differential effect on the emergence and seedling growth of the three target species. The inhibition effect of its root and leaf extracts on the germination of B. bipinnata may partially explain the overwhelming dominance of B. pilosa var. radiata over B. bipinnata when they are sympatric.
KEY WORDS: Ageraturem conyzoides, aqueous tissue extracts, Bidens pilosa L. var. radiata Sch. Bip., B. bipinnata, invasive plants.
INTRODUCTION
Two exotic Bidens species, belonging to Asteraceae,
were reported in Taiwan (Peng et al., 1998). Bidens
pilosa L. var. radiata Sch. Bip., a native plant of North
America (Peng et al., 1998), was first recorded in Taiwan in 1984 (Wu et al., 2004). It is now widely distributed and listed as one of the twenty most noxious invasive plants in Taiwan (Chang et al., 2003). Bidens bipinnata, the other exotic Bidens species in Taiwan, is also a native of North America and was first recorded in Taiwan in 1904 (Wu et al., 2004). In contrast to B. pilosa var.
radiata, B. bipinnata has become a naturalized plant and
mainly distributed in Southern Taiwan (Peng et al., 1998). Probably due to its less competitive than B. pilosa var. radiata, very few populations of B. bipinnata can be found in Taiwan now (personal obs). During a field survey of the distribution of Bidens species in Taiwan, we found both species growing sympatrically inside an abandoned farm where B. pilosa outnumbered B.
bipinnata. According to observations made by the owner
of the orchard, B. bipinnata used to dominate the farm, however, it declined rapidly during B. pilosa var. radiata invasion. By the time we started the experiment, about
99 % of the ground inside the farm was covered by B.
pilosa L. var. radiata (ca. 40 plants m-2), while only about
20 plants of B. bipinnata (covered less than 10 m2) were
found. The question occurred to us, what makes B. pilosa L. var. radiata outcompete B. bipinnata?
Not only the mechanisms by which B. pilosa var.
radiata displaces B. bipinnata are unexplored, factors
contributing to the success of B. pilosa var. radiata in Taiwan are poorly understood. Allelopathy, the plant-plant interaction mediated by chemicals released from the plant, is known to play a significant role in affecting community structure, including plant dominance and succession (Muller, 1966). Recently, more and more studies have reported that part of the success of invasive plants may be their novel allelopathic effects (Bais et al., 2003; Hierro and Gallaway, 2003; Prati and Bossdorf, 2004; Dorning and Cipollini, 2006; Cipollini et al., 2008). Stevens and Tang (1985) reported that root exudates of B. pilosa inhibited seedlings growth of some crop species. Accordingly, is it possible that B. pilosa var. radiata also exerts allelopathic effects on B. bipinnata? Because of difficulties in separating allelochemical effects from resource competition, it is extremely difficult to demonstrate allelopathy effect in the field (Bais et al., 2003). Instead, many researches have successfully shown
the potential for allelopathy in invasive plants using tissue extracts (Lawrence et al., 1991; Roberts and Anderson, 2001; Bais et al., 2003; Hierro and Callaway, 2003; Doring and Cipollini, 2006; Cipollini et al., 2008). A similar approach was also applied in this research to study the effects of the invasive plant on its sympatric species.
Ageraturem conyzoides, an annual herb originally
from tropical America, is also a common weed in Taiwan and is often found growing sympatically with the invasive plant in Northern Taiwan (pers. obs.). Is the performance of this species also affected by the invasive plant? This is the second question we would like to answer in this study. If the invasive plant exerts differential effects on its sympatric plants, it will have significant potential in altering community structure in Taiwan.
The objective of this study was to understand whether the aqueous tissue extracts from B. pilosa var.
radiata affected the performance of its sympatric plants
and whether the effects varied among tissue types and among target species. To achieve the objective, we applied aqueous tissue extracts from the invasive plant to seeds of target species and then measured seed germination and seedling growth of these species.
MATERIALS AND METHODS
Methods used by San Emeterio et al. (2004) were adopted to mimic the leachates of B. pilosa var. radiata produced in the field and to test its allelopathic potential on target species.
B. pilosa var. radiatawere harvested in Juy of 2005
from an abandoned farm in Madou Town (120°15’ E, 23°10’ N), Tainan County, SouthernTaiwan. The farm, ca. 1500 m2, was originally established for the
cultivation of shaddock, while had been abandoned for at least one year before we stated conducting the experiment. At the time of the plant harvesting, no more shaddock could be seen, and the farm was densely covered by other plant species, mainly Basella alba L.,
B. bipinnata, B. pilosa var. radiata, Chloris barbata Sw., Ipomoea cairica (L.) Sweet, and Passiflora suberosa
Linn. Among them, B. pilosa var. radiata was the most dominant species covering about 99 % of the ground inside the farm (ca. 40 plants m-2).
Plants of B. pilosa var. radiata harvested from the
farm were separated into leaf, stem, and root components. Each component was first cleaned to remove the dirt, air-dried and then ground into powder by a mechanical grinder. Five gram of each ground component was dissolved into a 5 L of distilled water by 12 hours of continuous blending. The resulting solution was then subjected to centrifugation (3000 rpm for 15
min for root, 3500 rpm 20 min for stem and leaf components) for the separation of supernatant and residues. The supernatant was collected and its water potential and pH value determined with a osmometer (WP4-T, Decagon Devices, Inc., USA) and a pH meter (TS-2, syntex), respectively. The effect of extract from each component, prepared as above, of B. pilosa var.
radiata on seed germination rate and the growth of
seedlings were examined.
Seeds of B. pilosa var. radiata and B. bipinnata were also collected from the abandoned farm on July 2nd, 2005. Though Ageraturem conyzoides was not found in the farm, it is often found growing sympatrically with B. pilosa var.
radiata in Northern Taiwan (pers. obs.). Accordingly, we
also examined the effect of extracs of B. pilosa var. radiata on this species. Seeds of A. conyzoides were collected on the campus of National Taiwan University, Taipei, Taiwan, 2005.
In a pioneer experiment, we found that seeds of B.
pilosa var. radiata and B. bipinnata were easy to be
infected by fungus. To prevent fungus infection, seeds of these two species were sterilized in NaOCl (5% v/v), and then washed with distilled water before being sowed. Twenty five seeds of each tested species were placed on filter paper in a petri dish (diameter × height = 90 mm × 15 mm). A 4 ml of extract from each component was added to the filter paper in each Petri dish. Distilled water was used as control. There were four replicates for each treatment and resulting in 48 Petri dishes in total. These Petri dishes were then transferred to a growth chamber setting at 25 ℃/20 ℃ for day/night temperature, a relative humidity of 70%, and a 12/12 hr light/dark regime. The photosynthetic photon flux density (wavelengths at 400- 700 nm) at the light period was about 100 μmol m-2 s-1.
During the experiment, the solutions in each Petri dish evaporated gradually. To avoid possible effect caused by changes in solution concentration, we replaced the filter papers, added solution and carefully transferred seeds (or seedlings) onto the new plates on day 10 of the experiment.
Seed germination was recorded everyday for 17 days. Seeds were considered germinated when radicles could be observed by naked eyes (according to Reddy and Singh, 1992). All three species are epigeous and on the seedlings regions for radical and hypocotyl can be easily identified (Fig. 1). The effect of extract on the growth of seedlings was assayed by measuring the length of radicle and hypocotyl of the germinating seeds. For each tested species, lengths of radicle and hypocotyl of seedlings were measured on a single day when its seeds had reached the maximal germination rate, i.e., on day 8 for B. pilosa var.
radiata, day 15 for B. bipinnata, and day 7 for A. conyzoides. These two parameters were not measured on
Days after sowing 0 2 4 6 8 10 12 14 16 18 G erminati on p ercentage (% ) Root Stem Leaf Control 100 80 60 40 20 0 (a) * * * * * * * * * * * *
Days after sowing
0 2 4 6 8 10 12 14 16 18 Germ in ati on pe rc en tag e (% ) Root Stem Leaf Control 100 80 60 40 20 0 (b) * * * * * * *
Days after sowing
0 2 4 6 8 10 12 14 16 18 Ger m in atio n p e rcentag e (% ) Root Stem Leaf Control 100 80 60 40 20 0 (c)
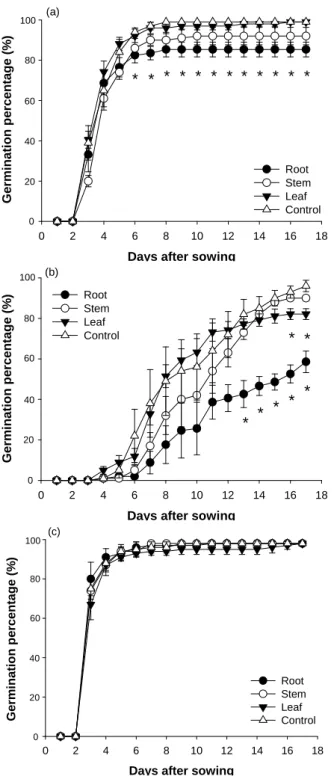
Fig. 1. Effect of aqueous extracts of roots, stems, and leaves of Bidens pilosa var. radiata on the germination of (a)
B. pilosa var. radiata, (b) B. bipinnata, and (c) Ageratum conyzoides. A star sign indicates a significant difference
from control (p <0.05, t-test, n = 4).
Means were analyzed by unpaired t-test (double-tailed, alpha = 0.05) to identify significant difference from control.
RESULTS
The water potential of extract from each component was close to 0 MPa (data not presented), which was greater than the threshold value believed to affect seed germination (approximate -0.25 MPa, L. San Emeterio et al., 2004). The pH value was 7.3 of root extract, 5.6 of stem extract, 5.3 of leaf extract, and 6.8 of control. Almost all of the seeds (99 %) of B. pilosa var. radiata germinated after being incubated in distilled water for 8 days (Fig. 1a). The final germination percentage of seeds of B. pilosa var. radiata was significantly reduced by application of root extracts, 39 % reduction, in contrast, that was neither affected by stem extract nor by leaf extract.
After being incubated in distilled water for 17 days, 97% of seeds of B. bipinnata germinated (Fig. 1b). In compared to control, the final germination percentage was significantly reduced by root and leaf extracts, 36 and 15% reduction, respectively. Application of stem extract also reduced seed germination percentage, however, the effect was not significant.
In contrast to those of B. pilosa var. radiata and B.
bipinnata, the percentage of seed germination of Ageratum conyzoides was not affected by any of the three tissue
extracts (Fig. 1c).
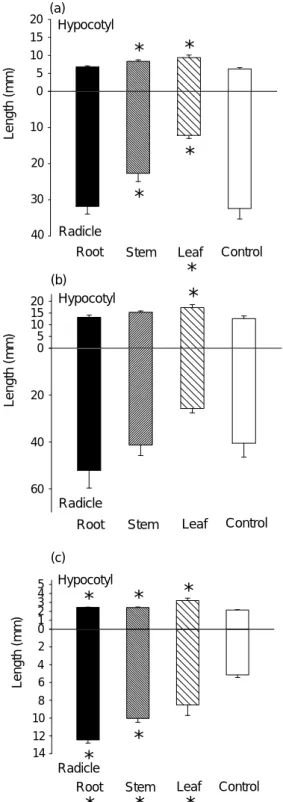
In B. pilosa var. radiata, the overall growth of new tissue was reduced by stem and leaf extracts (Fig. 2a). However, these extracts exerted differently on the elongation of hypocotyls and radicals. Both stem and leaf extracts significantly stimulated the elongation of hypocotyl while inhibited that of radical. In contrast, root extract neither affected the elongation of hypocotyls nor that of radicals.
Though the overall growth of seedlings of B. bipinnata was not affected by any tissue extract, the elongation of hypocotyls was stimulated by leaf extract (Fig. 2b). In contrast to that of B. pilosa var. radiata and B.
bipinnata, the growth of hypocotyls and radicals of A. conyzoides was significantly stimulated by all three tissue
extracts. Consequently, the overall growth of seedlings of this species was significantly increased by all three tissue extracts of B. pilosa var. radiata (Fig. 2c).
DISCUSSION
Results from this study demonstrated that the aqueous extracts from tissues of B. pilosa var. radiata had differential effect on the emergence and seedling growth of its same species, B. bipinnata and A. conyzoides. The responses of seed germination and seedling growth to the extracts were not consistent. Tissue extracts showing significant influence on seed germination (Fig. 1)
Len gth (mm) 0 5 10 15 20
Root Stem Leaf Control 20 40
*
*
*
*
*
10 30 (a) Hypocotyl Radicle Le ngth (mm) 0 5 10 15 20Root Stem Leaf Control 20 40 60
*
(b) Hypocotyl Radicle Len gth (mm) 0 1 2 3 4 5 2 4 6 8 10 12 14Root Stem Leaf Control
*
*
*
*
*
*
*
*
(c) Hypocotyl RadicleFig. 2. Effect of aqueous extract of roots, stems, and leaves of Bidens pilosa var. radiata on the seedling growth of (a) B.
pilosa var. radiata, (b) B. bipinnata, and (c) Ageratum conyzoides. Bars stand for standard errors. The star signs
above x axis, below x axis, and right beside the names of the treatments indicate significant differences of hypocotyl, radicle, and total seedling length from control, respectively (p <0.05, t-test, n = 4).
did not necessarily exhibit the same level of effect on seedling growth (Fig. 2). For instance, seed germination percentage of B. bipinnata was significantly reduced (Fig. 1b), whereas the growth of hypocotyl, radicle and total length of seedling of the same species were not affected (Fig. 2b) by the extracts of root of B. pilosa var. radiata. In
A. conyzoides, tissue extracts significantly enhanced the
growth of its seedlings (Fig. 2c), however, germination percentage was not affected (Fig. 1c). These results suggested the existence of allelopathic effect, however, the influence of allelochemicals on seed germination and seedling growth may work through different mechanisms. The differential response of germination and seedling growth to tissue extracts was also found in other studies. A study has shown that B. bipinnata whose radicle length seemed to be more sensitive to pH value than seed germination (Reddy et al., 1992). In addition, San Emeterio et al. (2004) also found that seedling growth of three pasture species was more sensitive to allelochemiclas than seed germination.
In general, hypocotyls displayed more consistent response to tissue extracts than radicals (Fig. 2). The growth of hypocotyls was mostly stimulated by tissue extracts. In contrast, the response of radicals to tissue extracts varied among species. The elongation of radicals of B. pilosa var. radiata was inhibited by leaf and stem extracts, that of B. bipinnata was not affected, while that of
A. conyzoides was enhanced by all three extracts. The
study by San Emeterio et al. (2004) also showed that shoot extract from Lolium rigidum stimulated aerial growth of seedlings of three pasture species. In that study, the authors also demonstrated that allelochemicals displaying a stimulatory effect at low concentrations may exhibit an inhibitory effect at higher concentrations (San Emeterio et al., 2004).
Self-inhibiton was observed in B. pilosa var. radiata. Seed germination percentate of B. pilosa var. radiata was lowered by its own extract of leaves. Radicle growth, which was crucial to early establishment, was also inhibited by the extracts of stems and leaves. The influence may be even intensified under restricted water conditions (Kobayashi, 2004; San Emeterio et al., 2004). This self-inhibition potential could reduce possible future intra-species competitions by interfere with the growth of other individuals at an early stage.
Allelochemicals produced in the tissues of plants may enter soils as leachates, root exudates or during tissue decomposition (Inderjit and Duke, 2003). Though only aqueous tissue extracts were used in this study, the results reveal the potential of inhibition effect of the invasive plant B. pilosa var. radiata on the congeneric species B.
bippinata. However, it has been found that both
hydrophobic and hydrophilic root exudates of B. pilosa inhibited seedling growth of some crop species (Stevens
and Tang, 1985). Accordingly, further studies need to be conducted to examine the effect of hydrophobic component of tissue extracts and to further verify the alleochemical effect of this species on the same species and other species in field. In addition, the concentration of tissue extracts used in this study was referenced the study of Lolium rigidum (a plant with a productivity of 425 kg ha-1) by San Emeterio et al. (2004). A survey on
the residue of B. pilosa var. radiata produced in field and therefore the actual concentration of leachate released into the soil is necessary to evaluate the dosage effect of tissue extracts on its neighbours. In addition, microbes and other compositions in soil may change the way an allelochemical works (El-Khatib et al., 2004; Vivanco et al., 2004). Environment conditions, for example, the precipitation and nutrient level, may also play a role in allelopathy by amplifying of reducing the effect (Kobayashi, 2004; San Emeterio et al., 2004; Cipollini et al., 2008). How these factors interact with allelochemicals released by B. pilosa var. radiata deserve further investigation.
In conclusion, A. conyzoides, B. bipinnata and B.
pilosa var. radiata showed differential response to
aqueous tissue extracts of B. pilosa var. radiata. The aqueous tissue extracts inhibited seedling growth of B.
pilosa var. radiata while enhanced that of A. conyzoides.
Root and leaf extracts significantly inhibited seed germination of B. bipinnata. Similar taxa would have similar ecological niche. Resuls of this study revealed that the effect of aqueous tissue extracts on seed germination and seedling development was more deterious to B. pilosa and B. bipinnata than to A.
conyzoides. The pattern implys a high ability of B. pilosa
to compete against similar taxa. A high allelopathic inhibiton of related neighbours in early stages of development would reduce the threat that they become competitiors later on. In addition, the inhibition effect of
B. pilosa on B. radiatioa may partially explain the
overwhelming dominance of B. pilosa var. radiata over
B. bipinnata when they are growing sympatrically.
ACKNOWLEDGEMENTS
We thank Mr. Pao-Yuan Chen and his family for allowing us to get access to the orchard farm and for their hospitality. This study was supported by a grant from the National Science Council (NSC 94-2621-B-002-010) of Republic of China.
LITERATURE CITED
Bais, H. P., R. Vepachedu, S. Gilroy, R. M. Gallaway and J. M. Vivanco. 2003. Allelopathy and exotic plant invasion: from molecules and genes to species interactions. Science 301: 1377-1380.
Chang, M., L.-M. Hsu, C.-I. Yuan, F.-Y. Chen and Y.-C. Chang. 2002. The harmful effect and ecology of invasive plants in Taiwan. In: The Harmful Effect and Field Management of Mikania micrantha, Hualien District Agricultural Research and Externsion, Agricultural Council, Taiwan. pp. 97-109.
Cipollini, D., R. Stevenson and K. Cipollini. 2008. Contrasting effects of allelochemicals from two invasive plants on the performance of a nonmycorrhizal plant. Int. J. Plant Sci. 169: 371-375.
Dorning, M. and D. Cipollini. 2006. Leaf and root extracts of the invasive shrub, Lonicera maackii, inhibit seed germination of three herbs with no autotoxic effects. Plant Ecol. 184: 287-296.
El-Khatib, A. A., A. K. Hegazy and H. K. Galal. 2004. Allelopathy in the rhizosphere and amended soil of Chenopodium murale L. Weed Biol. Manag. 4: 35-42.
Hierro, J. L. and R. M. Callaway. 2003. Allelopathy and exotic plant invasion. Plant Soil 256: 29-39.
Kobayashi, K. 2004. Factors affecting phytotoxic activity of allelochemicals in soil. Weed Biol. Manag. 4: 1-7.
Lawrence, J. G., A. Colwell and O. J. Sexton. 1991. The ecological impact of allelopathy in Ailanthus altissima (Simaroubaceae). Am. J. Bot. 78: 948-958.
Muller, C. H. 1966. The role of chemical inhibition (allelopathy) in vegetational composition. Bull. Torrey Bot. Club 93: 332-351.
Prati, D. and O. Bossdorf. 2004. Allelopathic inhibition of germination by Alliaria petiolata (Brassicaceae). Am. J. Bot. 91: 285-288.
Peng, C.-I, K.-F. Chung and H-L. Li. 1998. Compositae. In: Huang, T.-C. et al., (eds.), Flora of Taiwan. 2nd ed. 4: 807-1101. Editorial Committee, Dept. Bot., NTU, Taipei, Taiwan.
Reddy, K. N. and M. Singh. 1992. Germination and emergence of hairy beggarticks (Bidens pilosa). Weed Sci. 40: 195-199. Roberts, K. J. and R. C. Anderson. 2001. Effect of garlic
mustard [Alliaria petiolata (Beib. Cavara & Grande)] extracts on plants and arbuscular mycorrhizal (AM) fungi. Am. Midl. Nat. 146: 146-152.
San Emeterio, L., A. Arroyo and R. M. Canals. 2004. Allelopathic potential of Lolium rigidum Gaud. on the early growth of three associated pasture species. Grass Forage Sci. 59: 107-112.
Stevens, G. A. and C.-S. Tang. 1985. Inhibition of seedling growth of crop species by recirculating root exudates of Bidens pilosa L. J. Chem. Ecol. 11: 1411-1425.
Vivanco, J. M., H. P. Bais, F. R. Stermitz, G. C. Thelen and R. M. Callaway. 2004. Biogeographical variation in community response to root allelochemistry: Novel weapons and exotic invasion. Ecol. Lett. 7: 285-292.
Wu, S-H, C-F. Hsieh and M. Rejmanek. 2004. Catalogue of the naturalized flora of Taiwan. Taiwania 49: 16-31.
比較入侵植物大花咸豐草的組織水溶液萃取液對其共域植物的影響
徐曉玫(1)、高文媛(1,2*)
1. 國立臺灣大學生態學與演化生物學研究所,106 臺北市羅斯福路四段 1 號,臺灣。 2. 國立臺灣大學生命科學系,106 臺北市羅斯福路四段 1 號,臺灣。
* 通信作者。Tel: +886-2-33662511; Fax: +886-2-2367-3374; Email: wykao@ntu.edu.tw
(收稿日期:2009 年 4 月 8 日;接受日期:2009 年 6 月 16 日)